Grantee Research Project Results
2012 Progress Report: Toxicity Screening with Zebrafish Assay
EPA Grant Number: R835168Title: Toxicity Screening with Zebrafish Assay
Investigators: Tanguay, Robyn L.
Institution: Oregon State University
EPA Project Officer: Aja, Hayley
Project Period: May 1, 2012 through April 30, 2016
Project Period Covered by this Report: May 1, 2012 through April 30,2013
Project Amount: $1,199,999
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The overall objectives of our project have not changed and are to:
- Expose zebrafish embryos to at least 1,000 chemicals.
- Study the morphology and behavior of the embryos to look for evidence of developmental toxicity.
- When an adverse effect occurs, conduct whole-transcriptome RNA sequencing studies to observe gene expression patterns, identify the initial biochemical target, and begin to elucidate the toxicity pathway.
- Conduct statistical modeling to compare the results of the zebrafish assay with the results of in vitro assays and other in vivo assays where they exist.
- Share our data with EPA and the broader research community.
Progress Summary:
In the first year of the grant we focused on improving the statistical analysis of our high throughput data. Important collaborations with David Reif (NC State, at no cost to grant) were established.
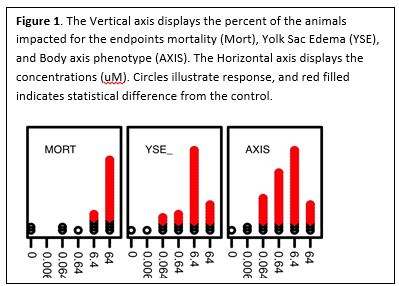
- We collaboratively developed novel statistical and data approaches to quantify and visualize toxicity data. For the first 100 chemicals screened we observed varied concentration response curves. In some cases, we had stereotypical dose response relationships; however, many of the compounds produced very steep concentration responses. This made it impossible to calculate EC50 values. Working with Dr. Reif at NC State, we instead decided to calculate the lowest effect level concentration for each compound and for each endpoint. An example of the data analysis is provided in Figure 1 below.
- We focused the remainder of the year on conducting the world’s largest in vivo toxicity screen covering broad chemical classes and across broad concentration ranges (0-64 µm). The chemicals were provided by the U.S. Environmental Protection Agency and the ToxCast program. This was an enormous task that leveraged heavily on the instrument development that was just accomplished through NIH funding. We evaluated all 1060 unique U.S. EPA ToxCast Phase 1 and 2 compounds using the embryonic zebrafish and found that 487 induced significant adverse biological responses. The utilization of 18 simultaneously measured endpoints means that the entire system serves as a robust biological sensor for chemical hazards. The experimental design enabled us to describe global patterns of variation across tested compounds, evaluate the concordance of the available in vitro and in vivo Phase 1 data with this study, highlight specific mechanisms/value-added/novel biology related to notochord development, and demonstrate that the developmental zebrafish detects adverse responses that would be missed by less comprehensive testing strategies. In total, we screened more than 400,000 embryos.
- As promised, all the ToxCast raw morphology phenotype data has been shared with the U.S. Environmental Protection Agency (EPA).
- In addition to the morphology phenotypic data, we also collected 24 hour behavioral (spontaneous motion) data for all of the embryos. At this point, we have not determined the appropriate statistical analysis tools that will be a focus in the next year. A manuscript reporting this research is in the final stage of preparation. An exciting outcome is that this assay has proven predictive for numerous later stage toxic responses. This is particularly important as the throughput of the assay is very high.
- David Reif is proposing a web-based interface for investigators using our testing platform. A version 1.0 has been developed and piloted by our group. These tools will make this model more accessible to the broader research community.
- We have contacted the National Toxicology Program and Ray Rice, who will provide a large set of chemicals for additional testing in Year 2 of the grant.
- The PI has presented this approach at several meetings in the past few months.
Future Activities:
- Complete analysis of the 24 hpf behavioral data and finish the manuscript.
- Prepare the benchmark dose manuscript.
- Present results to further disseminate the approach and the results to the broader research community.
- Complete the screening of the NTP compounds.
- Identify the compounds from our hits for deep sequencing.
Journal Articles:
No journal articles submitted with this report: View all 27 publications for this projectProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.