Grantee Research Project Results
Final Report: Uptake of Specific Engineered Nanoparticles (ENP) by Sludge Particulates as Affected by the Presence of Dissolved Organic Matters (DOM)
EPA Grant Number: R834859Title: Uptake of Specific Engineered Nanoparticles (ENP) by Sludge Particulates as Affected by the Presence of Dissolved Organic Matters (DOM)
Investigators: Huang, C. P. , Johnston, Murray V.
Institution: University of Delaware
EPA Project Officer: Aja, Hayley
Project Period: February 1, 2011 through January 31, 2014
Project Amount: $599,678
RFA: Increasing Scientific Data on the Fate, Transport and Behavior of Engineered Nanomaterials in Selected Environmental and Biological Matrices (2010) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The goal of this project was to test the hypothesis that engineered nanoparticles (ENP) tend to be collected at solid particulates in wastewater treatment and that physico-chemical properties of the dissolved organic matter (DOM) and solids (both ENP and sludge) play important roles in the distribution of ENP in wastewater treatment plants (WWTP). To achieve this goal, the following three specific objectives will be pursued: 1) to study the distribution of ENP in wastewater treatment plants, 2) to study the uptake of ENP by wastewater particulates, and 3) to predict the distribution of ENP in wastewater treatment plants.
Summary/Accomplishments (Outputs/Outcomes):
1. Methodology for the identification and quantification of engineered nanoparticles in wastewater.
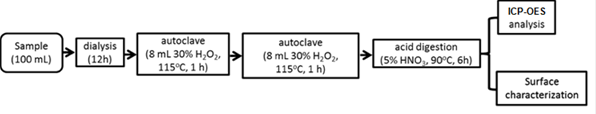
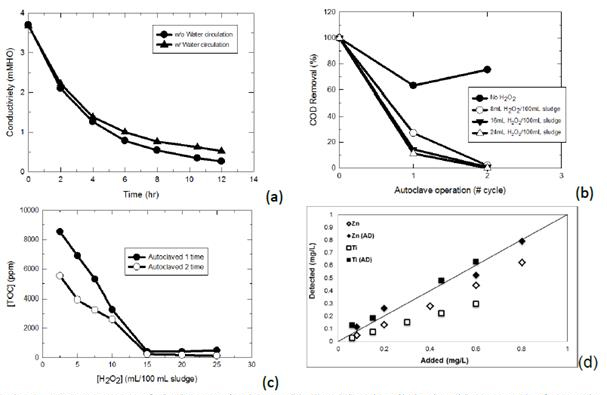
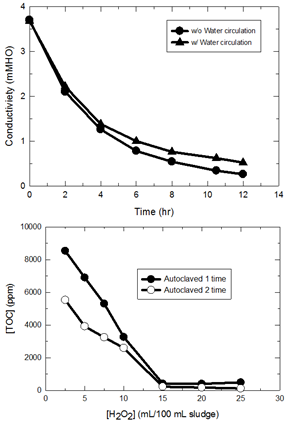
It is believed that engineering nanoparticles in a wastewater treatment system can be present in the bulk phase as free particles or become associated with the sludge particulates. Currently, two general methods have been used to detect nanoparticles in wastewater: by direct digestion followed by total metal content analysis using induction coupled plasma spectroscopy or by infinite dilution of the sludge mass. The former method overestimates the concentration of nanoparticles, whereas the second method, is thought to be more reliable than the first method with regard to particle content; nonetheless, the second methods is extremely time-consuming. In this research, we have developed a rational method for the identification and quantification of engineered nanoparticles in wastewater. Sludge or wastewater samples were pre-treated with dialysis to remove inorganic salts. The desalted sludge mass then is treated with thermal oxidation in the presence of an appropriate amount of H2O2. Results indicated that for every 100 mL of sludge/wastewater sample, desalting was completed in 10-12 hours in static or dynamic flow mode. For every 100 mL of sludge/wastewater sample (MLSS 2,000 mg/L), 15 mL of 30% H2O2 was able to remove 99.9% of organic matter by thermal treatment using an autoclave operated at 115°C for 1 hour. The final solution was analyzed by inductively coupled plasma atomic emission spectroscopy (ICP-OES) for total metal concentration with and without acid digestion (Standard Method 3030G using 5% HNO3). Results indicated that acid digestion before ICP-OES is necessary to account for the total nanoparticles presented.
The method so developed is described by the following flow chart:
Figure 1. Pretreatment of sludge particulates: (a) desalting by dialysis; (b) removal of organic matter
by autoclave; (c) effect of H2O2 dosage on organic removal; (d) effect of acid digestion of solution before
ICP-OES analysis
The particles so collected were analyzed further for particle size and for the identification of the chemical state using transmission microscopy and elemental mapping technique. The particle size was analyzed using an aerosol atomizer incorporated with a mass spectrometer (NAMS). Nanoparticles in the size range of 15-30 nm were detected.
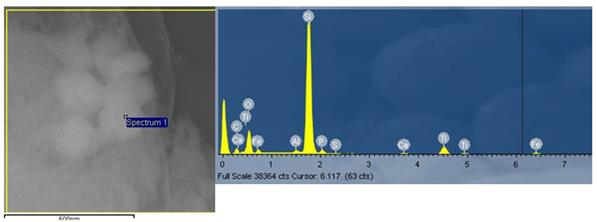
The sludge particulates collected at the end of the pretreatment process were dispersed using ultrasound and the particles were examined using a scanning electron microscope (Fig. 2) and the chemical composition of the particles was analyzed using energy dispersive x-ray spectroscopy (EDX) as shown in Figure 2. The identification of engineered nanoparticles was done by mapping the elements of the SEM images as shown in Figure 3. Results showed that TiO2 and ZnO are the major engineered nanoparticles present in the wastewater treatment systems studied. Other nanoparticles such as Fe2O3 and SiO2 also were present. However, due to the abundance of iron and silicon in the natural environment, it is assumed that these particles were of natural origin rather than from anthropogenic contributions. The particle size of engineered nanoparticles as seen from SEM is shown in Figure 4. Generally, the particle size is in the range of 100-300 nm.
Figure 2. SEM image and EDX spectra of sludge particles of primary sludte a W WWTP.
Figure 3. Mapping of Ti and O elements confirming presence of nano-TiO2 particles.
Figure 4. SEM images of sludge particulates
2. The distribution of engineered nanoparticles in wastewater treatment plants.
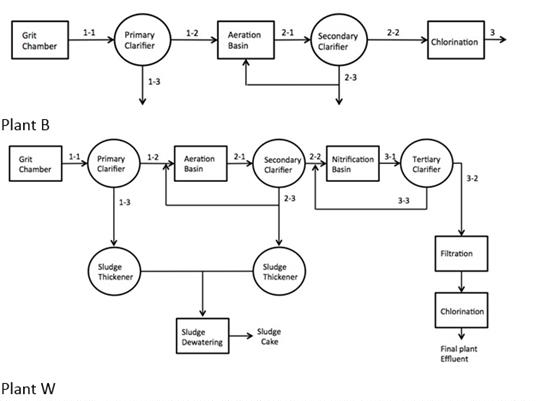
The distribution of engineered nanoparticles, namely, TiO2 and ZnO, were investigated focusing on two municipal wastewater treatment plants, namely, Plant B and Plant W. Fig. 5 shows the plant flow systems. Plant W has a daily treatment capacity of 370 MGD and Plant B has an average treatment capacity of 180 MGD.
Figure 5. Flow diagram of two munincipal wastewater treatment plants, B and W.
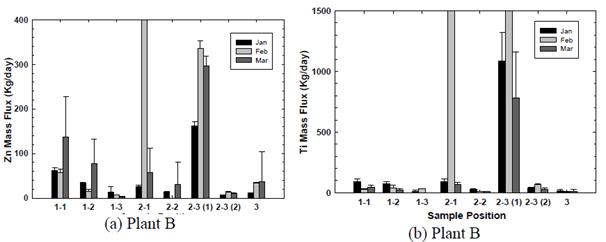
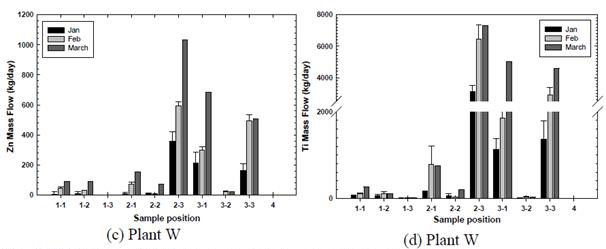
The distribution of TiO2 and ZnO nanoparticles in the above WWTPs was shown in the following figures (Fig. 6 a-d). Results showed a remarkably high content of ENP in sludge particulates. Clearly, the secondary sedimentation basin becomes an important sink for ENP, both TiO2 and ZnO. The other point of ENP concentration was the return sludge of the tertiary sedimentation basin. Results demonstrated that there was significant accumulation of ENP in the secondary and the tertiary sedimentation basins. Results also showed that at Plant B, the flux of ENP, both ZnO and TiO2, were at the peak values during February; whereas the peak ENP flux at Plant W was slightly increased in March. It is interesting to note that Plant B is located a few miles north of Plant W. Furthermore, the plant capacity of Plant W was greater than that of Plant B.
Figure 6 Distribution of ENP and two municipal wastewater treatment plants.
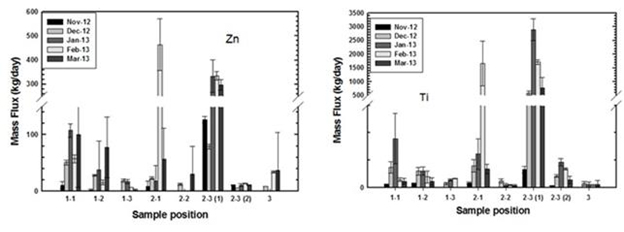
The seasonable distribution of ENP in municipal wastewater treatment plants was further studied at Plant B. As shown in Figure 7, results clearly indicated there was a surge of ENP flux during the months of January and February.
Figure 7. Seasonable distribution of nano-TiO2 and nano-AnO at Plant B
3. Interactions between engineered nanoparticles and sludge particulates: aggregation mode.
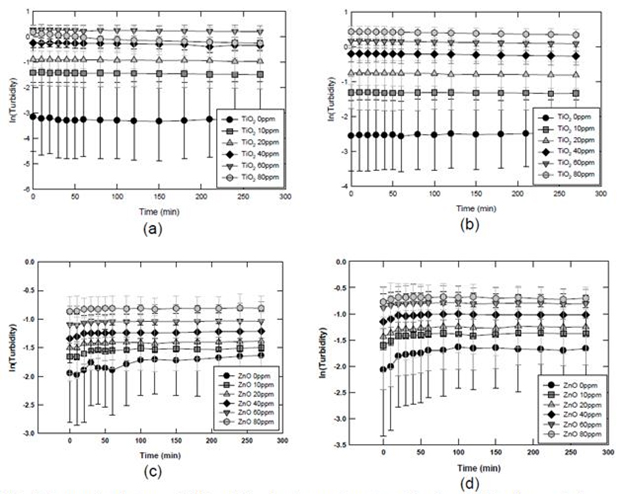
It is hypothesized that the interaction between ENP and sludge particulates is dependent on their relative particle size. That is, the mode of particle interaction will be dominated by particle aggregation (of ENP) when the particle size of the sludge particulates is smaller than that of the ENP. Likewise, the mode of particle-particle interactions will be dominated by uptake or adsorption of ENP onto sludge particulates, when the sludge particulates are greater in size than that of ENP. To test this hypothesis, the sludge particulates were classified into four fractions: (1) fraction 1 of DOM (~1000 Da), fraction 2 of 1000 Da 1000 nm, (3) fraction 3 of >1000 nm, and (4) fraction 4 of the total sludge (all the fractions above). Fraction 1 was obtained by centrifuging (at 10000 g) of the supernatant of sludge after sedimentation (Fraction 2); Fraction 2 was obtained from the supernatant of sludge sedimentation (for 12 hrs); Fraction 3 was obtained by sludge sedimentation (for 6 hrs), and Fraction 4 was the whole sludge mass. As indicated above, when the sludge particulates are smaller or in the same size range as that of ENP, the interaction between sludge particulate and ENP will undergo the particle aggregation process; that is, interactions such as adsorption of DOM and mutual capture of sludge particulates and ENP will result in the growth of the ENP. The following results show that the interaction between the DOM of primary and secondary sludge with nano- ZnO and nano-TiO2. It is interesting to note that there was little change of particle size upon exposing ENP to the DOM for TiO2 and a slight increase in particle size of ZnO upon contact with DOM. The DOM from the secondary sludge appeared to be more reactive with nano-ZnO.
Figure 8. Interaction between ENP and dissolved organic matters (franction 1). (a) primary sludge DOM and TiO2; (b) secondary sludge DOM and TiO2; (c) primary sludge DOM and ZnO; (d) secondary sludge DOM and ZnO.
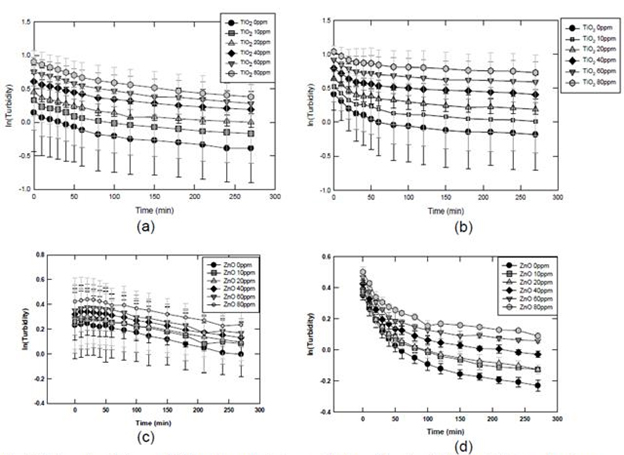
The degree of interaction between fraction-2 sludge particulates, i.e.,<1000 nm was significant, more so for the secondary sludge than the primary as shown in Figure 9. Results show that the turbidity decreased as the contact time increased. This can be attributed to the increase in aggregate size due to mutual capture of sludge particulates and ENP, which resulted in settling of the aggregates formed over time.
Figure 9. Interaction between ENP and small sludge particulates (franction 2, 100nm). (a) small primary
sludge particulates and TiO2; (b) small secondary sludge particulate and TiO2; (c) small primary
sludge particulates and ZnO; (d) small secondary sludge particulates and ZnO.
4. Interactions between engineered nanoparticles and sludge particulates: uptake mode.
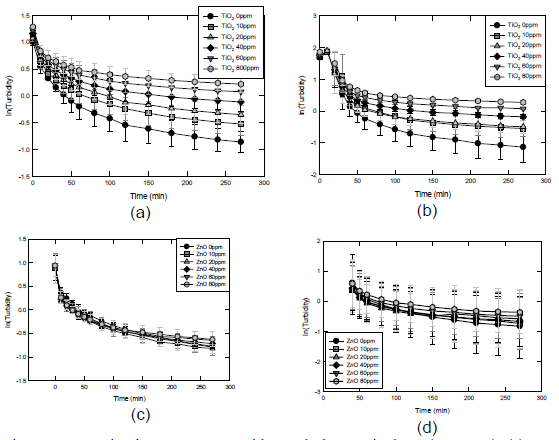
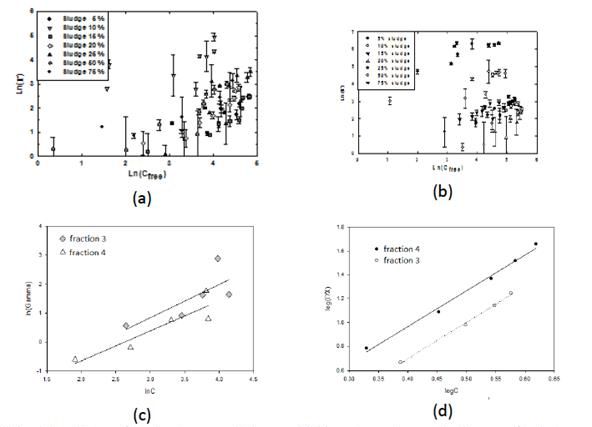
The interaction between Fractions 3 and 4 of sludge particulates with nano-TiO2 and nano-ZnO was studied. As shown in Figure 10, the interactions between the particles, i.e., sludge and engineered nanoparticles, resulted in preferential sedimentation of the larger sludge particulates leading to the capture of ENP by the settling sludge particulates. A preferential sedimentation technique developed in our laboratory enabled the determination of the mass of the free nanoparticles and the associates including the sludge particulates and the ENP. Results in Figures 10 and 11 can be further analyzed to obtain the adsorption isotherms. The uptake of ENP by the sludge particulates can be described by the Fruendlich adsorption isotherms: q = kC1/n, where q, k, C and n are the adsorption density, constant, equilibrium free concentration of ENP, and exponential constant, respectively. The linearized Fruendlich adsorption is shown in Figure 12. Table 1 summarizes the equilibrium constants of the uptake of ENP onto sludge particulates (Fractions 1 and 4).
| Table 1. Summary of Fruendlish Adsorption Parameters | |||
|---|---|---|---|
| TiO2 | |||
| Sludge particulates | n | K | R2 |
| Primary f4 | 0.23 | 4.12 X 10-6 | 0.87 |
| Primary f3 | 0.5 | 6.74 X 10-3 | 0.98 |
| Secondary f4 | 1.0 | 6.72 X 10-2 | 0.58 |
| Secondary f3 | 0.91 | 7.43 X 10-2 | |
| ZnO | |||
| Primary f4 | 0.93 | 0.47 | 0.7 |
| Primary f3 | 1.53 | 1.55 | 0.96 |
| Secondary f4 | 3.01 | 0.58 | 0.99 |
| Secondary f3 | 3.03 | 0.31 | 0.99 |
Figure 10. Interaction between ENP and large sludge particulates (1000 nm). (a) largeprimary sludge particulates and TiO2;
(b) large secondary sludge particulate and TiO2; (c) large primary sludge particulates ZnO; (d) large secondary sludge particulates
and ZnO.
Figure 12. Adsorption isotherms. (a) nano-TiO2 onto primary sludge particulates (fraction 4);
(b) nano-ZnO onto primary sludge particulates (fraction 4); (c) TiO2 on secondary sludge particulates;
(d) ZnO onto secondary sludge particulates.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 6 publications | 2 publications in selected types | All 2 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Fan R, Huang YC, Grusak MA, Huang CP, Sherrier DJ. Effects of nano-TiO2 on the agronomically-relevant Rhizobium-legume symbiosis. Science of the Total Environment 2014;466-467:503-512. |
R834859 (Final) |
Exit Exit Exit |
|
|
Huang YC, Fan R, Grusak MA, Sherrier JD, Huang CP. Effects of nano-ZnO on the agronomically relevant Rhizobium-legume symbiosis. Science of the Total Environment 2014;497-498:78-90. |
R834859 (Final) |
Exit Exit Exit |
Supplemental Keywords:
engineered nanoparticles, titanium dioxide, zinc oxide, nanoparticle adsorption, wastewater treatment, nanoparticle distribution;Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.