Grantee Research Project Results
2011 Progress Report: Uptake of Specific Engineered Nanoparticles (ENP) by Sludge Particulates as Affected by the Presence of Dissolved Organic Matters (DOM)
EPA Grant Number: R834859Title: Uptake of Specific Engineered Nanoparticles (ENP) by Sludge Particulates as Affected by the Presence of Dissolved Organic Matters (DOM)
Investigators: Huang, C. P. , Johnston, Murray V.
Institution: University of Delaware
EPA Project Officer: Aja, Hayley
Project Period: February 1, 2011 through January 31, 2014
Project Period Covered by this Report: February 1, 2011 through January 31,2012
Project Amount: $599,678
RFA: Increasing Scientific Data on the Fate, Transport and Behavior of Engineered Nanomaterials in Selected Environmental and Biological Matrices (2010) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Progress Summary:
We have successfully developed procedures for the purification and isolation of engineered nanoparticles from wastewater-sludge matrix. Dialysis was used to remove salts in the matrix. Dialysis for 2 hours was adequate to eliminate > 90% of salts in sludge mass. Further purification for the separation of ENP from the salt-free sludge mass was achieved by advanced oxidation process. The salt-free sludge mass was treated with hydrogen peroxide in autoclave at 105oC for 1 h. Results showed that oxidation achieved TOC removal at approximately 99 percent. Electrophoretic process was used to classify the inorganic particles in the purified sludge mass. Particles so collected covered the size range of 8 to 250 nm observed under SEM. Identification of individual nanoparticles was carried out by finger-printing the chemical composition of the particle-suspensions using ICP-OES technique. Results showed that titanium, zinc, iron, and aluminum nanoparticles dominated the particle population. Measurements indicated a high concentration of ENP in the return sludge and aeration basin indicating that ENP has a strong affinity toward sludge particulates, namely biological and organic matters. Concentration of NP was detected at a level of 600 mg-NP/kg-died sludge in a Wilmington wastewater treatment plant. Results also showed that nanoparticles were aggregated in the sludge mass.
An international collaboration program was initiated to gain broader information on the distribution and fate of engineered nanoparticles in the water environment. Our collaborator was Professor Gen-shuh Wang of the Department of Public Health, National Taiwan University. There are many high-tech industries in Taiwan. We hypothesized that the nature and the distribution of engineered nanoparticles in a more industrial environment would be different from those of a municipal scenario. The particles were classified using super-speed centrifugation and membrane filtration. The concentration of metallic element in the solution was detected with ICP-MS. Results showed that the concentration of NP increased as the water traveled downstream. Results showed that aluminum, titanium and zinc were the major components of nanoparticles in the size range of nano-meter; iron was found in particles in the submicron and nanometer range. There was a general decrease of particle concentration in the sub-micro- and the nanometer range. The concentration of nano-sized particles in the river waters was in the range of 80 to 180 ppb of iron. The concentration of other elements was low in the range of 10 to 45 ppb. Further classification of nanoparticles collected from a municipal wastewater treatment plant in Wilmington, DE, was conducted using a scanning mobility particle sizer (SMPS) coupled with a nanoparticle nebulizer (LiquiScan). Results showed that the combination of SMPS and LiquiScan was able to detect particles in the size range of 10-20 nm. Preliminary experiments were conducted to study the interactions between nanoparticles and simple electrolytes.
Supporting Information
Brief Statement
The first-year annual report includes results of the research activities conducted from March 1, 2011, to February 29, 2012. The main focus of this years' research was on studying the distribution of engineered nanoparticles (ENPs) in wastewater sludge. We were able to successfully separate the nano-size inorganic materials from the sludge for a series of physical and chemical characterization. A variety of metallic ENPs were detected in the sludge samples and characterized. The samples were treated by various methods as to isolate ENPs for the analysis of element composition, total particle quantity, particle size, and number concentration. Results showed the presence of titanium, zinc, iron and aluminum nanoparticles throughout the wastewater treatment process, including the water that was discharged from the wastewater treatment plants (WWTP) to the water environment. Concentration of metallic ENPs up to a level of 600 mg-NP/kg-dried sludge was detected. Results also showed that the ENPs present in sludge were in the form of aggregates. The stability of nanoparticle stability in water was studied, preliminarily.
The ENPs in the sludge were separated first by dialysis as to remove the inorganic salts. The salt-free sludge was then treated with advanced chemical oxidation to remove the organic fraction such as biological and organic materials using wet peroxide oxidation method. Hydrogen peroxide was added to the sludge samples, and the mixture was autoclaved for 1 hr at 105°C. Dialysis was able to remove > 90 percent of salts over a period of 2 h. Advanced chemical oxidation achieved a TOC removal of approximately 99 percent.
Energy-dispersive X-ray spectroscopy (EDX) and scanning electron microscopy (SEM) were used to analyze the size and element composition of ENPs. Titanium, zinc, iron and aluminum nanoparticles were detected. SEM images revealed the size of ENPs to be 80~250 nm. Results obtained from the EDX were used as a guideline to design procedures for ICP-OES (Inductively Coupled Plasma-Optical Emission Spectrometer) measurements. ICP-OES was able to detect elements at the ppb levels. Results indicated a high concentration of ENPs in the return sludge and aeration basin, which was indicative of a strong affinity between ENPs and sludge particulates. It can also be inferred that ENPs will accumulate within the WWTP system due to continuous circulation of the sludge. Results showed that the amount of ENP in the effluent was similar to that of the inflow to the WWTP. This indicates that, even with the attachment of ENPs to sludge particulates, most of the ENP discharged into the WWTP is not captured by the wastewater treatment process.
To evaluate the amount of nanoparticles in the receiving water from industrial parks, three sites in Taiwan were selected for ENP assessment. Samples were collected from two rivers (Siao Li River and Ke Ya River) and analyzed with an ICP-MS. Results showed high concentrations of iron up to 250 ppb and zinc of 40 ppb with various amounts of titanium as well. Results showed increasing amounts of ENP toward the downstream of both rivers. There was also a spike in the ENP concentration near the wastewater discharge point of the Hsinchu Science Park.
In addition to ICP-OES, Inductively Coupled Plasma-Mass Spectroscopy (ICP-MS) and SEM, a scanning mobility particle sizer (SMPS), were used to classify the particle size and condensation particle counter (CPC) of the ENP of municipal wastewater using a LiquiScan NP nebulizer and SMPS detection system. Results showed that the ENP in the sludge had a diameter of 10~20 nm. Results of LiquiScan NP nebulizer showed that the particle size decreased with dilution indicating aggregation of the particles. The number concentration also increased as the dilution factor increased, which confirmed that dilution destabilizes aggregates. The size distribution of the most dilute solutions suggested that the individual nanoparticle size was on the order of 10 nm or less.
The stability of ENP in aqueous medium was also studied using titanium dioxide (TiO2) as model ENP. The critical coagulation concentration (CCC) was measured in the presence of mono-valent chloride (NaCl) and divalent sulfate (Na2SO4) anions at a pH of 3. Results showed an increase in the hydrodynamic radius with time at different electrolyte concentrations. The CCC was established at lower salt concentrations. The ratio of CCC was 1:33.3 for NaCl: Na2SO4. The difference between the experimental and the theoretical CCC values might be attributed to the asymmetrical nature of the electrolyte in question. It must be noted that the ShulzeHardy rule was applicable only to symmetrical electrolytes, such as NaCl, and may not be applicable precisely to asymmetrical electrolytes such as Na2SO4.
In summary, we have successfully qualified and quantified the amount of ENP in a municipal wastewater treatment plant. The size, element composition and coagulation characteristics were studied using a host of instrumentations. Ongoing research activities will focus on studying the interactions between model ENP and sludge particles. Moreover, with the successful development of isolation and separation procedures for ENPs, we will expand our study sites to cover all four municipal wastewater treatments in Philadelphia, PA; Baltimore, MD; Washington, DC; and Wilmington, DE. Results obtained so far have been reported preliminarily at an on-campus conference organized by the Delaware Institute of Environment (DENIN).
Results to Date
Separation of Engineered Nano Particles (ENP) from Wastewater Sludge
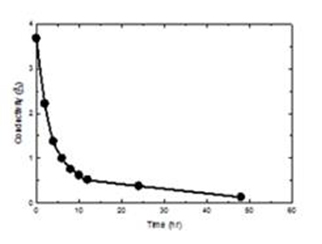
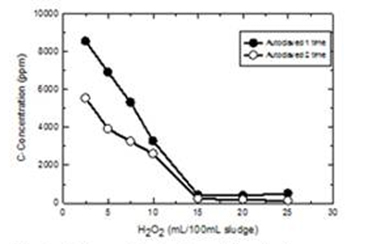
The separation of ENP within the sludge was achieved in two steps: dialysis and advanced oxidation process. Dialysis was able to remove the salts present in sludge. The salt-free sludge that still contains a good fraction of biological and organic materials was treated with an advanced oxidation process using hydrogen peroxide and autoclave. Dialysis was applied to the sludge samples before advanced oxidation to remove existing slats and metal ions. A given amount of sludge was placed in a seamless cellulose dialysis bag with a molecular weight cut-off of 1 kDa. The dialysis tubes were submerged in a water tank of 2 L for total of 48 hr during which the water in the tank was changed every 24 hr. Results shown in Figure 1 indicate a removal of salts, measured in terms of conductivity, from 3.7 to 0.3 mΩ in 48 hr, 92 percent removal. The wet peroxide method was used to oxidize the organic and biological material in the salt-free sludge. Hydrogen peroxide was added to the sludge and autoclaved for 1 hr at 105°C. The degree of organic removal, measured in term of TOC (total organic carbon), was dependent on the hydrogen peroxide dosage. Figure 2 shows typical results of TOC removal as a function of hydrogen peroxide dosage and treatment time. Results showed that 15 mL of 30% H2O2 and 1 hr autoclaving was able to remove 99 percent TOC.
Figure 1. Dalysis result of activated sludge. The dialysis
tube was submerged in a tank for 48 hr and changed
every 24 hr.
Figure 2. Sludge oxidation by adding hudrogen peroxide and
autoclaving. Amount of hudrogen peroxide was changed
additional to the number o autoclave operations - 10 mL.
H2O2 (30%), Temperature 1050C, Autoclaved once (●),
Autoclaved twice (O)
Qualifying and Quantifying the Amount of ENP in the Sludge
Energy-dispersive X-ray spectroscopy (EDX) and scanning electron microscopy (SEM) were used to analyze the size and elemental composition of the ENP. Based on the results obtained with the EDX, aluminum, silver, titanium, zinc and iron were chosen as the standard elements for inspection (Figure 3). Figure 4 show typical SEM images of City of Wilmington municipal sludge; the size of the ENP in the sludge was 80~250 nm. Results obtained from the EDX analysis were used as a guideline to design procedures for elemental quantification using inductively coupled plasma optical emission spectrometry (ICP-OES) and inductively coupled plasma mass spectrometry (ICP-MS) techniques.
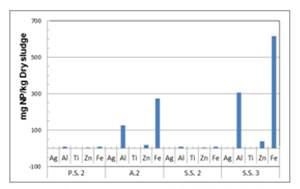
ICP-OES technique was capable of detecting inorganic faction of nanoparticles at concentrations in the ppm levels. Figure 5 shows typical results of major inorganic elements deriving from various types of ENP in the sludge from the wastewater treatment plant in the City of Wilmington, DE. Results showed high concentration of ENP in the return sludge and the aeration basin, indicating that ENP has a strong affinity to sludge particulates. Results showed high concentrations of iron, aluminum and zinc within the return sludge (S.S.3), whereas the primary sedimentation outflow (P.S.2) exhibited the lowest concentration of all elements. Considering the fact that the return sludge is continuously circulated within the WWTP, the high concentrations of metallic NP can be expected. The high concentration of the ENP in the aeration basin can be explained by the addition of return sludge with the primary sedimentation outflow. However, considering the normal mixing ratio of the P.S.2 and the return sludge, the ENP concentration in the aeration basin was higher than expected indicating a certain amount of circulation of the sludge within the basin. Trace amounts of iron and aluminum were still detected in the outflow of the secondary sedimentation basin (S.S.2). This indicates the fact that conventional WWTP cannot separate nanoparticles that are discharged into the wastewater treatment facility.
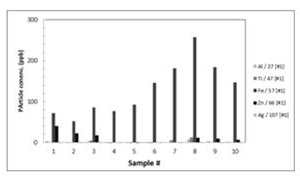
Sixteen raw water samples from Taiwan were collected and analyzed with the ICP-MS. The samples were from various areas of Taiwan, which were adjacent to science parks and highly populated areas. Figure 6 shows typical results of element profile of the water samples. The samples came from three major areas where samples #1~5 were sampled from the Siao Li River, and samples #6~10 were sampled from the Ke Ya River. Results indicated an increasing amount of iron and titanium NP among samples #3 and #4 compared with samples #1 and #2. It can be assumed that there was an additional source of pollution entering along the stream. However, it can be observed that the amount of zinc is decreasing throughout the flow of the river. With samples #6~10, there was a definitive increase in the iron composition as the water travels downstream. A distinct spike in all the tested elements can be observed with sample #8 (Hsinchu Science Park wastewater discharge point). However, samples taken downstream of this point showed a lower amount of NP, suggesting the adsorption of NP onto organic or particular silica in the environment.
Figure 3. Typical EDX results of major elements in the oxidized sludge
from the wastewater treatment plant in the City of Wilmington, DE.
Figure 4. Typical SEM images of oxidezed sludge from
the wastewater treatment plat of the City of Wilmington, DE.
Figure 5. Element profile of oxidized sludge from
Wilmington WWTP.
Figure 5. Typical element profile of water samples at
He Sing upstream of Siao LI R. and KeYaR., Hsin Chu
Tiwan.
Detection and Determination of Physical Properties of Nanoparticles From WWTP
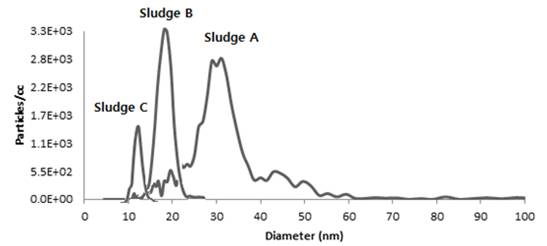
Electrospray aerosol analysis (EAA) technique was used to further characterize the ENP particles collected from the wastewater treatment plant of the city of Wilmington, DE. The physical properties (size distribution, number concentration) were determined with a scanning mobility particle sizer (SMPS) that consists of a differential mobility analyzer (DMA) to classify the particle size and a condensation particle counter (CPC) to quantify the size distribution of nonvolatile material that was originally in the liquid sample. Wastewater samples were collected at three points at Wilmington WWTP. One sample was retrieved from the returned sludge from the secondary sedimentation basin (Sludge B). A sample was taken from the effluent of the primary sedimentation (Sludge A) and from the effluent of the secondary sedimentation (Sludge C).
Sludge C (Figure 7) exhibited a slightly smaller median diameter, whereas Sludge A showed the largest diameter. The number of particles was significantly greater for sludge B, A and C by approximately a factor of 10.
Figure 7. Typical size distribution of undiluted sludge samples. A~C using the EAA technique.
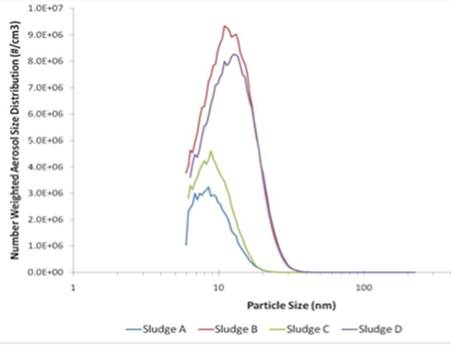
To further investigate whether the EAA size distributions reflect individual nanoparticles or nanoparticle aggregates, the samples were analyzed using the LiquiScan NP nebulizer. This allowed an internal dilution of the samples in order to detect loose agglomerates of the nanoparticles. Figure 8 shows the typical size distribution for each sample at different dilution factors. The distribution for a dilution factor of about 30 gave a smaller diameter for each sample than the EAA results. An even larger dilution factor of about 400 was measured to give an even smaller diameter (< 10nm) for each wastewater sample. The number concentration of each sample also increased as the dilution factor increased, which confirms the hypothesis that dilution destabilizes aggregates. The size distributions of the most dilute solutions suggested that the individual nanoparticle sizes were on the order of 10 nm or less.
Figure 8. Number weighted aerosol size distribution of wastewater samples using LiquiScan technique
5.4 Stability of ENP
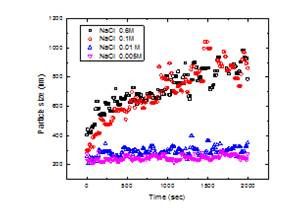
A model ENP, TiO2, was used to study the stability of nanoparticles in the water environment. Dynamic light scattering (DLS) (Zetasizer nano, Malvern, UK) was used to determine the hydrodynamic radius of the TiO2 particles and a value of 200 nm (±20 nm) was obtained. Figure 9 shows the increase of TiO2 particle size as a function of time.
Figure 9. Dynamic light scattering (DLS) measurements
of TiO2 aggregation. The aggregation profile was measured
as a function of salt concentration. Conditions: TiO2 =
10mg/L, pH = NaCI used as an electrolyte.
The experiments were performed in duplicates at each electrolyte concentration showing good reproductivity. Results showed an increase in the hydrodynamic radius with time for different electrolyte concentrations. The slope of the hydrodynamic radius versus time curve increased dramatically when the electrolyte concentration was increased from 0.01 M to 0.1 M. Any increase of the electrolyte concentration showed good reproductivity. Results showed an increase in the hydrodynamic radius with time for different electrolyte concentrations. The slope of the hydrodynamic radius versus time curve increased dramatically when the electrolyte concentration was increased from 0.01 M to 0.1 M. Any increase of the electrolyte concentration did not result in an additional increase of the particle size, indicating that NaCl concentration of 0.1 M was in the fast aggregation regime. This is in good agreement with the characteristics of the fast regime where the coagulation rate constant is independent of the electrolyte concentrations. It can also be observed that at higher electrolyte concentrations, the particle size distribution increases with time. This is due to multiple scattering, which leads to a diffuse halo around the primary laser beam inside the cell and a reduced intercept of the autocorrelation function, resulting in a wider spread in particle size measurement. Figure 10 shows the aggregation profiles with different divalent anion concentrations at pH 3.
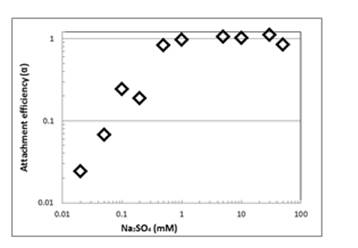
Figure 10. Attachment efficiency as a function of Na2O4
concentration obtained at a TiO2 concentration of 10 mg/L
and at pH 3.
Results showed a rapid increase in the slope at a Na2SO4 concentration of 0.5 mM. The slopes of the aggregation profile obtained with the two different electrolytes showed the same average rate constant (k) at concentration higher than the CCC, confirming that aggregation kinetics are diffusion-controlled under such conditions. Compared with the results in Figure 9 (which has the same pH and different electrolytes), the CCC of NaCl : Na2SO4 is 1:33.3. This is different from 1: 64 as predicted by the Schulze-Hardy rule. The difference between the experimental and the theoretical CCC values might be attributed to the asymmetrical nature of the electrolyte in question.
Future Activities:
Journal Articles:
No journal articles submitted with this report: View all 6 publications for this projectProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.