Grantee Research Project Results
2013 Progress Report: Center for Integrative Research on Childhood Leukemia and the Environment (CIRCLE)
EPA Grant Number: R834511Center: Center for Integrative Research on Childhood Leukemia and the Environment - 2015
Center Director: Metayer, Catherine
Title: Center for Integrative Research on Childhood Leukemia and the Environment (CIRCLE)
Investigators: Buffler, Patricia , Rappaport, Stephen M. , Kyle, Amy , Chokkalingam, Anand , Metayer, Catherine , Dahl, Gary , Wiemels, Joe , Barcellos, Lisa , Zhang, Luoping , Miller, Mark , Selvin, Steve
Current Investigators: Metayer, Catherine
Institution: University of California - Berkeley , University of California - San Francisco , Stanford University
Current Institution: University of California - Berkeley
EPA Project Officer: Hahn, Intaek
Project Period: September 25, 2009 through September 24, 2015
Project Period Covered by this Report: September 25, 2012 through August 24,2013
Project Amount: $3,704,598
RFA: Children's Environmental Health and Disease Prevention Research Centers (with NIEHS) (2009) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
The Children’s Environmental Health Center based at the University of California, Berkeley is designed to examine the effects of in utero and early life exposure to potentially carcinogenic chemicals present in homes (i.e., pesticides, tobacco-related contaminants, polychlorinated biphenyls (PCBs), and polybrominated diphenyl ethers (PBDEs)), genetic and epigenetic factors and their interplay in the development of childhood leukemia. The Center, referred to as Center for Interdisciplinary Research on Childhood Leukemia and the Environment (CIRCLE), includes three Research Projects and two Cores.
R834511C001: Childhood Leukemia International Consortium Studies
Childhood Leukemia International Consortium Studies will identify the exposures to the most relevant time periods and childhood leukemia subtypes and identify important genetic polymorphisms that can modify the association between childhood leukemia and parental tobacco smoking or home pesticide exposure by pooling data from 19 studies worldwide.
R834511C002: Exposure Assessment for Childhood Leukemia
Exposure Assessment for Childhood Leukemia will assess carcinogen exposures, based upon analysis of house dust and blood specimens, with special interest in tobacco-related contaminants, PCBs, and PBDEs.
R834511C003: Prenatal Exposures, DNA Methylation & Childhood Leukemia
Prenatal Exposures, DNA Methylation, & Childhood Leukemia will provide a clearer understanding of the association between parental smoking, pesticides, PCBs, PBDEs exposures and DNA methylation patterns in childhood leukemia, using neonatal bloods.
Core A: Administrative Core
Core A will provide: (1) oversight, coordination, and integration of Center activities; (2) scientific leadership; and (3) centralized data management support. Core A will establish and manage Internal and External Advisory Committees; appoint and collaborate with two Pediatric Health Specialists; and support the research career development of new, junior faculty-level investigators within the structure of the Center.
Core B: Research Translation and Outreach Core
Core B, the Research Translation and Community Outreach Core (RTCO Core), will disseminate findings from each Project to various audiences with common interest in the etiology of childhood leukemia. Community engagement will be coordinated through the RTCO Core, with the assistance of the Pediatric Health Specialists.
Progress Summary:
R834511C001: Childhood Leukemia International Consortium Studies
Specific Aim 1: Pool data elements collected from 14 CLIC case-control studies in 10 countries.
General update: More than 40 participants attended the 2012 CLIC Annual Meeting held in Berkeley on October 1-3, in conjunction with the CIRCLE External Scientific Advisory Committee Meeting on September 28-29 (minutes of the CLIC meeting are available at clic_logo.gif Childhood Leukemia International Consortium Exit). The CLIC investigators leading pooled analyses presented updates and preliminary results during special workshops to enhance scientific interaction between investigators of participating studies. New childhood leukemia studies from Mexico, Denmark, Brazil, France, and the United Stated applied for CLIC membership and were granted Associate study level. They will be eligible to become Active studies once they contribute to pooled analyses.
The CLIC Management Group (chaired by Prof. Buffler, Director of CIRCLE) continues to hold regular conference calls to discuss issues related to data pooling, progress on proposed pooled analyses, CLIC guidelines, and organization of the CLIC annual meeting. Importantly, the CLIC Management Group recognizes the need to establish a Data Coordinating Center to support CLIC’s expanding research portfolio and infrastructure, and to secure funding for the Data Coordinating Center. As a first step toward this goal, the Data Coordinating Center will be hosted by the Section of Environment and Radiation at IARC in Lyon, France, under the direct supervision of the Section Head, Dr. Joachim Schüz (CLIC Member). The Data Coordinating Center proposes to (a) collect individual and pooled available CLIC data for centralized storage, (b) prepare datasets by harmonizing across study centers, (c) maintain records of study-specific knowledge important for future pooled analyses, and (d) distribute data to CLIC Principal Investigators for future projects.
A manuscript describing the history, composition, and objectives of CLIC was published in the journal Cancer Epidemiology (Metayer et al., Jun 2013; 37(3):336-347).
Update on pooled analyses: Progress on data harmonization and analyses of proposed pooled analyses is being made by lead investigators, as summarized in Table 1.
|
Table 1 |
|||
|
Pooled analyses |
Lead Investigator |
Institution |
Status since last report |
|
(1) Parental smoking, metabolizing genes, and risk of ALL |
Infante-Rivard C. |
McGill University, Montreal, Quebec, Canada |
Data from 13 studies were harmonized including ~ 9,100 cases and 14,860 controls and are currently being analyzed (see preliminary results below, Aim 3). |
|
(2) Parental smoking, metabolizing genes and risk of AML and APL |
Metayer C. |
UC Berkeley, USA |
Data from 10 studies were harmonized including ~ 967 cases and 11,530 controls and are currently being analyzed (see preliminary results below, Aim 3). |
|
(3) Home pesticide use and risk of ALL & AML |
Bailey H., Joachim Schüz |
IARC, France |
Data from 10 studies were harmonized including ~ 8,900 ALL cases, 1,400 AML cases, and 16,440 controls. |
|
(4) Geographical distribution of AML, APL and other cytogenetic subtypes |
Zhang L. |
UC Berkeley, USA |
Expression of Interest (EOI) was submitted in June 2011 - Data delivery delayed into Year 4 due to competing requests listed above. |
|
(5) Geographical distribution of ALL cytogenetic subtypes |
Pombo-de-Oliveira M. (to be confirmed) |
National Cancer Institute, Rio de Janeiro, Brazil |
EOI to be developed |
Specific Aim 2: Conduct descriptive analyses to assess geographical differences in the frequency of leukemia subtypes defined by age, immunophenotype and cytogenetics, and assess possible sources for geographical differences.
Dr. Luoping Zhang is conducting a literature review on the incidence of acute promyelocytic leukemia (a rare type of acute myeloid leukemia) worldwide.
Specific Aim 3: Assess the association between maternal/paternal smoking or home pesticide exposures and childhood leukemia during different time periods (prenatal, during pregnancy, and postnatal) stratified by histologic, immunophenotypic, and cytogenetic subtypes.
Parental smoking, metabolizing genes, and risk of childhood ALL: Logistic regression analyses adjusted for age, sex, and study center showed no associations with maternal smoking before and during pregnancy. Statistically significant increased risks of small magnitude were observed with paternal smoking before conception (pooled OR=1.13; 95% CI: 1.04-1.22) and during pregnancy (pooled OR=1.15; 95% CI: 1.05-1.27). These associations were no longer significant in models adjusting for parental education, whereas there was a suggestion that both maternal and paternal smoking after birth slightly increased the risk of childhood ALL (ORs=1.11, 95% CI:1.03-1.20), with risk increasing with larger number of cigarettes smoked per day. Analyses for metabolizing genes are under way.
Parental smoking, metabolizing genes, and risk of childhood AML: Logistic regression analyses adjusted for age, sex, race/ethnicity, education, and study center showed no associations with AML and maternal smoking at any time, and lifetime paternal smoking increased the risk of childhood AML (ORs=1.35, 95% CI:1.11-1.63). Fathers' smoking habits did not vary substantially over time (before conception, during and after pregnancy), limiting our ability to assess the separate contribution of specific window(s) of exposure. In-depth evaluation of the impact of study heterogeneity was conducted by partitioning of within/between variability, and assessing the goodness of fit of the analytic models.
Home pesticide use, metabolizing genes, and risk of childhood ALL and AML: Analyses will start shortly.
Specific Aim 4: Examine the influence of genetic variation on the association between parental smoking or home pesticide exposures and childhood leukemia by histologic, immunophenotypic and cytogenetic subtypes.
Statistical analyses are under way for parental smoking, xenobiotic genes and risk of ALL. The SNPs relevant to the other planned GxE analyses will be selected.
Specific Aim 5: Maintain the CLIC website Childhood Leukemia International Consortium Exit to facilitate communication among CLIC members and outside communities.
The CLIC website is being maintained and updated and used regularly by CLIC collaborators.
Significance
Leukemia is the most common type of childhood cancer. About 2,400 cases of childhood leukemia (ages 0-14 years) are diagnosed annually in the United States. The etiology of childhood leukemia is complex; confirmed clinical and epidemiologic associations explain less than 10% of childhood leukemia incidence. Project 1 is the first epidemiologic study that proposes to collaborate with a large international group of investigators to examine ubiquitous environmental exposures (i.e., tobacco smoking and residential pesticides) that may be causally associated with the most frequent cancer in children. Pooling data from 14 case-control studies presents a unique opportunity to fully investigate the critical periods of exposures to these contaminants and the possible modifying effects of metabolizing genes in the etiology of childhood, and to examine rare and less-studied childhood leukemia types like acute myeloid leukemia and other cytogenetic subgroups.
R834511C002: Exposure Assessment for Childhood Leukemia
Specific Aim 1: Measure cotinine, polychlorinated biphenyls (PCBs), and polybrominated diphenylethers (PBDEs) in serum samples obtained from 250 childhood leukemia cases at diagnosis. Estimate correlations in analyte levels between serum and house dust samples.
During Year 3, we developed a method for extracting PCBs and PBDEs from 100 mL of whole blood, consistent with available quantities of blood from the childhood leukemia cases. The current analytical scheme consists of a liquid:liquid extraction followed by silica column clean-up, and analysis via high resolution gas chromatography-mass spectrometry. In Year 4, we validated the method using whole blood samples from anonymized adults, spiked bovine serum samples, and standard reference serum from the National Institute of Standards and Technology (NIST SRM 1957 and NIST SRM 1958). To date, we have extracted 64 of 250 whole blood samples for PCB and PBDE analysis. We have analyzed 10 whole blood samples for PBDEs via high resolution gas chromatography-mass spectrometry.
Specific Aim 2: Investigate effects of time and seasonality on house dust levels of PBDEs.
- Measure levels of PBDEs in the 500 household-dust samples (original and repeat dust samples from each of 250 households. Repeat dust sampling and measurements of nicotine, PAHs and, PCBs will be conducted under NIEHS grant 1R01-ES015899-01A2).
- Use mixed-effects models of levels of PBDEs in house dust to evaluate trends, seasonality, and within-household variability.
During Years 3 and 4, we used the analytical method developed in Years 1 and 2 to measure PAHs (12 analytes), PCBs (6 congeners) and PBDEs (22 congeners) in repeat dust samples from 204 households that had been measured at baseline (giving a total of 408 baseline and repeat samples available for statistical analyses). At present, all PAH measurements have been completed while PCBs and PBDEs are at the final stage of mass spectrometric analysis.
We used mixed-effects models of house-dust PAH concentrations to estimate variance components and identify sources of variability. For some PAHs [benzo(b)fluoranthene, benzo(k)fluoranthene, benzo(a)pyrene, indeno(1,2,3-c,d)pyrene, dibenzo(a,h)anthracene, benzo(g,h,i)perylene], we observed a decreasing trend in PAH concentrations in house dust over time. Although house dust concentrations at baseline (2001-2007) were significantly correlated with concentrations from repeat samples (2010) for each PAH, substantial within-household variability was observed. Indeed, the variability in concentrations within a household (over time) was generally greater than the variability between households (across the population). We were able to identify several factors that explained some of the variability between households, including residents' cigarette smoking habits, urban density, residence construction date, and residence type. A manuscript describing the mixed-effects modeling of house-dust PAH concentrations has been published in Environmental Health Perspectives.
In Year 4, we used mixed-effects models of house-dust PBDE concentrations to estimate variance components and identify sources of variability. For some PBDEs (BDE-183, BDE-196) we observed a decreasing trend in PBDE concentrations in house dust over time. Although house dust concentrations at baseline (2001-2007) were significantly correlated with concentrations from repeat samples (2010) for each PBDE, substantial within-household variability was observed. Indeed, the variability in concentrations within a household (over time) was generally greater than the variability between households (across the population). We were able to identify several factors that explained some of the variability between households, including the presence of furniture with exposed or crumbling foam, the recent installation of carpets in the residence, neighborhood urban density, racial and ethnic characteristics, and to a lesser extent, income. A manuscript describing the mixed-effects modeling of house-dust PBDE concentrations has been accepted for publication in Environmental International.
Specific Aim 3: Develop and apply methods for detecting and profiling human serum albumin (HAS) adducts in samples of dried blood spots (DBS) and serum.
- Develop methods for measuring HSA adducts in DBS.
- Measure profiles of HSA adducts in DBS from leukemia cases and matched controls. Compare profiles between childhood leukemia cases and controls.
- Measure profiles of HSA adducts in serum from leukemia cases at diagnosis. Compare profiles of HSA adducts from leukemia cases measured in DBS and serum.
During Year 4, we finalized an untargeted method for profiling HSA adducts at the Cys34 locus in proteins extracted from DBS. The method involves extraction of proteins from a 3-mm DBS punch (about 60 mg of HSA), followed by removal of hemoglobin, digestion of serum proteins with trypsin and purification of the Cys34 adducted peptides by off-line high performance reverse-phase liquid chromatography. The adducted peptides are then profiled by Triple-Quadrupole MS/MS using a technique called Fixed-step SRM (also developed in our laboratory) to detect all Cys34 adducts with added masses up to 384 Da represented by 86 adduct bins. In profiling experiments with 46 DBS from control children -- half from smoking mothers and half with nonsmoking mothers -- we detected an average of 67 adduct hits from the 86 bins (78% of all bins). We currently are conducting permutation tests to determine if any adducts detected in fetal DBS are associated with maternal smoking as well as the gender and race of the infants.
We have identified DBS from 100 childhood leukemia cases and 100 matched controls for performing the final profiling experiments under this aim. DBS samples were selected to provide substantial overlap with serum analyzed under Aim 1 and with DBS analyzed for epigenetic effects in Project 3. Adduct profiles will be investigated during Year 5.
Specific Aim 4: Identify HSA adducts observed in profiles, paying particular attention to adducts associated with leukemia status and changes from birth to diagnosis.
Selected adduct fractions from the fetal DBS described above are being analyzed by HRMS to obtain accurate masses and molecular compositions on the more abundant adducts detected in the profiling experiments.
Specific Aim 5: Develop and apply quantitative assays for HSA adducts identified in Aim 4.
- Develop assays for isotope-dilution mass spectrometry of identified adducts.
- Investigate the stability of these adducts in DBS from control children of smoking and nonsmoking mothers.
- Quantify levels of these adducts in archived profiling samples from Aim 3 and investigate possible associations with self-reported parental smoking and house dust levels of nicotine, polycyclic aromatic hydrocarbons (PAHs), PCBs, and PBDEs.
During Year 4, we developed a targeted Triple-Quadrupole SRM method for quantitating particular Cys34 adducts of interest via isotope-dilution mass spectrometry. We then used this targeted method to quantitate 19 specific Cys34 adducts suspected to be associated with cigarette smoking, due to reactions involving aldehydes, quinones and reactive oxygen species. These data also are being analyzed to determine whether there are statistical associations with smoking status. This targeted method will be applied to adducts identified in childhood leukemia cases and controls under Aim 4.
Significance
Although leukemia is the most common type of childhood cancer, the etiology of childhood leukemia is complex and confirmed clinical and epidemiologic associations explain less than 10% of childhood leukemia incidence. The results of this study are providing important information regarding the contribution of various environmental, infectious, immune, and genetic factors to the risk of childhood leukemia. This Center is conducting research to examine the interplay of environmental, genetic and epigenetic factors of childhood leukemia, with a focus on children's exposures to common chemicals with strong biologic plausibility, i.e., benzene, nicotine, PAHs, PCBs, and PBDEs. In particular, PBDEs are the most common brominated flame retardants in the United States; they are persistent and ubiquitous environmental contaminants that have been implicated with cancer in humans and animals. Contact with house dust is thought to account for 80-90% of the total PBDE exposure, in large part because PBDEs originate entirely from indoor consumer products. Work in Project 2 is quantifying PBDEs in both house dust and serum from childhood leukemia cases.
Preliminary work in Project 2 has shown that profiles of HSA adducts can be measured in 3-mm punch samples from fetal DBS of control children. We now will measure these adducts in DBS collected at birth in childhood leukemia cases and matched controls. Because HSA persists in human blood for a month, such measurements of adducts in fetal DBS will provide valuable information about fetal exposures during the final month of gestation.
R834511C003: Prenatal Exposures, DNA Methylation & Childhood Leukemia
Specific Aim 1: Characterize the DNA methylation pattern of normal B-cell differentiation as compared to their leukemic cell counterparts.
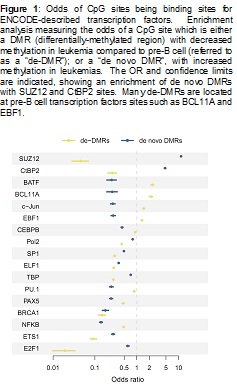
We have characterized the pre-B cell methylation pattern of normal B-cell differentiation, and published the results (Lee, et al., 2012). We also have characterized the leukemia methylome in relation to pre-B cells, with a manuscript in draft. We are replicating this latter analysis by performing a whole genome bisulfite sequencing for a pair of leukemia samples and also several stages of pre-B cells. This work will be submitted to a high profile journal later this year. A particularly interesting finding from the comparison of leukemia cells to normal cells is an observed association between SUZ12 and CtBP2 binding sites (both of which are described as transcription factors in the ENCODE Project) to CpG locations that had increased DNA methylation in the leukemias. An enrichment of CpG site DNA methylation changes is shown in Figure 1.
Specific Aim 2: Characterize DNA methylation pattern in 250 neonatal DBS cards from leukemia cases (derived from the same case samples as Aim 1) and 250 controls.
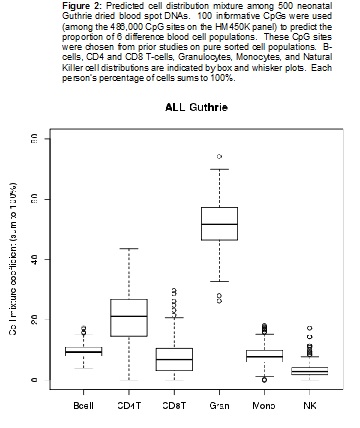
The laboratory component of this aim is completed, and we currently are performing extensive analysis. In the past year, we have worked to develop and execute new analytical strategies to improve analysis of newborn bloods, and remove bias or confounding due to differences in cell distribution. Every blood cell type has its own distinct DNA methylation pattern. Environmental factors, genetics, infections or other factors may influence the growth patterns of various blood cell types. If one were to attempt to make associations between such factors and DNA methylation at specific CpG sites, the result may be biased or confounded by differences in cell distributions between individuals. We now are correcting for these distribution differences in our analyses. In Figure 2, we demonstrate the overall distribution of cell types within our Guthrie card population. These cell distributions, determined using differentially methylated regions, are exactly what one would predict for actual cell counting (which is impossible on dried blood spots). In addition, the data have been normalized with linear mixed effect models, to minimize array, position, and plate differences in our experiments. Because of these extensive enhancements to our analysis, our “discovery” analysis has taken a few more months than expected; however, the results from these studies will be far more reliable and robust.
Specific Aim 3: Replicate and extend the findings of Aim 1 by characterizing DNA methylation in a set of disease- and exposure-relevant meta-stable CpG sites in DBS cards from select groups of DBS cards from California leukemia cases and controls.
We have constructed a sample set for replication, which includes 250 case and 250 gender-birthdate-ethnicity matched children, and have cut cards and isolated DNA. Depending on the final results from Aim 2, we will construct a replication panel of CpG sites that are associated with both environmental exposure variables as well as leukemia incidence. This biomarker panel will be subjected to a second replication analysis using neonatal blood spots that are from California births but outside of the California Childhood Leukemia Study (CCLS).
Significance
The etiology of childhood leukemia is complex; confirmed clinical and epidemiologic associations explain less than 10% of childhood leukemia incidence. Despite the recognition that certain genotoxic exposures such as diagnostic radiation during pregnancy cause leukemia in children, leukemia rates have not fallen; rather, there is an increased incidence over the past 50 years. Part of this increase in incidence is likely due to changes in patterns of exposure to manufactured and natural chemicals introduced into a child’s environment, and also changing patterns of infectious disease exposure and severity.
DNA methylation is a process of marking genes during development for the compression of chromatin and an “off” signal for expression. DNA methylation also is vital for suppressing recombinogenic regions of the genome such as retrotransposon fossils. Environmental exposures may impact the signal transduction pathways that lead to DNA methylation, and/or affect DNA methylation directly, causing aberrant DNA methylation patterns and changes in the methylation status of individual critical genes. Project 3 studies the association of precisely assessed chemical measurements in California Childhood Leukemia Study participant’s birth bloods in cases and controls with DNA methylation status in the same birth bloods as well as leukemic bone marrows (among cases only). Our comprehensive examination and integrated statistical analysis will reveal new relationships and pathways between environmental exposures and key epigenetic factors that impact the genesis of acute lymphocytic leukemia in children.
Core A: Administrative Core
Specific Aim 1: Assist the Center Director in the daily planning, execution, and coordination of the Center.
Aim 1 is being met. Members of the Internal Advisory Committee including the Project and Core Directors, Pediatric Health Specialists, and the Junior Faculty Investigator meet monthly to discuss progress on all three Projects and Cores, as well as any additional needs for administrative support. Decision and action items are summarized in detailed minutes, to ensure timely follow-up. CIRCLE members participate in monthly NIEHS/EPA conference calls with investigators from other Centers.
On September 28-29, 2012, members of the Internal Advisory Committee met with the External Advisory Committee (EAC): Andrea Hricko (USC/UCLA), David Martin (Children’s Hospital Oakland Research Institute), Rob McConnell (USC), Lauren Zeise (Cal EPA), Jorn Olsen (Danish National Birth Cohort), Elaine Faustman (University of Washington), Irva Hertz-Picciotto (University of California, Davis), and Paul Wise (Stanford). Terry Dwyer (Murdoch Children’s Research Institute), Elaine Faustman (University of Washington) and Kathleen Stewart (Region 9 EPA), were not be able to attend the EAC. The meeting was coordinated in the same venue with the Annual Meeting of the California Childhood Leukemia Study EAC (September 27-28) and the Annual Meeting of the Childhood Leukemia International Consortium Coordinating (October 1-2) to facilitate interactions and potential collaborations among childhood leukemia investigators. The EAC reported that it was impressed “with the accomplishments of the Center, and that there was clear evidence of extensive, ongoing analytic and methodological research that holds the promise of novel and important insights into the biology of childhood leukemia, risk factors, and environmental exposure assessment with broad implications for this disease and for the field of exposure assessment and environmental epidemiology more generally.”
The Center received a site visit from Rich Callan and Nica Louie on May 30, 2012. The site visit included (1) review and update of projects, cores; (2) presentations by the junior faculty development investigator and Pediatric Health Specialists; (3) discussions pertaining to financial status of the grant; and (4) visits to two labs (Rappaport and Barcellos.) The reviewers reported that, “the projects are making great progress and the researchers are continuing with their efforts under all three projects. Preliminary findings for the projects were shared and the next steps were outlined.
Specific Aim 2: Provide scientific leadership for the overall Center.
Core A coordinated the participation of Center members in the January 16-17, 2013 Symposium on Cumulative Impacts and Children's Environmental Health in Sacramento in conjunction with West Coast Centers and the Western PESHUs. The Symposium was organized by Core B and Pediatric Health Specialist, Dr. Mark Miller. The meeting was followed by presentations to the California Legislature. A meeting conference proceedings paper is being drafted and videos of the presentations will be posted on YouTube.
Core A coordinated the following presentations at the Center public webinar in October:
- Patricia A. Buffler. Improving Knowledge of the Environmental Causes of Leukemia in Children: the Center for Integrative Research on Childhood Leukemia and the Environment (CIRCLE)
- Catherine Metayer. Global Perspectives on Childhood Leukemia Research - the Childhood Leukemia International Consortium
- Joseph Wiemels. Building a Framework to Study Environmental Effects on DNA Methylation in Blood Cell Development -- and Links to Leukemia
- Todd Whitehead. Estimating Exposures to Indoor Contaminants (such as PAHs and PBDEs) Using House Dust
Core A also coordinated a review and response to a factsheet about childhood leukemia for parents and others interested in children's environmental health who lack any particular technical background, which was drafted by the Children’s Environmental Health Network, one of our Research Translation Core partners.
Center Director Dr. Patricia Buffler accepted an invitation to serve on the Steering Committee to assist with organizing the centers national meeting in Washington, D.C., in October 2013.
Pediatric Health Specialists
Center Pediatric Health Specialist, Dr. Gary Dahl, is collaborating with Center members Dr. Steve Rappaport, and Steve Francis (graduate student), and Erin Breese, a Fellow at Stanford, on a genomics/metabolomics pilot that grew out of discussions during Center IAC meetings. The aims of the pilot will be to compare protein adducts on the serum albumin between cases and controls and to compare identifiable and non-identifiable nucleic acids processed from total RNA between leukemia patients and controls.
Center Pediatric Health Specialists, Dr. Mark Miller and Dr. Gary Dahl are co-authors on an abstract submitted by Cristy Zachek MPH, entitled "Translating environmental health research into clinical practice: A survey of pediatric hematologists and oncologists" to the American Public Health Association Conference (APHA) in San Francisco (October 27 - October 31, 2012) to be held in San Francisco, CA. The abstract was selected for an oral presentation, and builds on the survey instrument work of our Pediatric Health Specialists. A paper will be coming out soon.
An online Continuing Education credit course will be translated into a simpler version for the general public. The focus will be on cumulative impacts and the complex interactions of environment and genetic factors pertaining to disease risk in five areas, the first being asthma and the second being childhood leukemia. This web-based program will be case-based or story-based. The home page will show a photo of a family reunion whose members represent different races. Users will be able to scroll over people and obtain their story (e.g., a five year old with leukemia and then follow branches to obtain more information). Center members will record 2-3 minute videos to be included in the project.
Junior Faculty Development
Dr. Morimoto (the Center’s Junior Faculty Development appointee) was awarded an Exploratory/ Developmental Research Grant Award (R21) from NIEHS/NIH in August 2012, titled “Pilot Study of Childhood Leukemia in Guatemala” (1R211ES021819-01) on her first submission, receiving a priority score of 18. The purpose of this pilot study is to assess the feasibility of conducting a full case-control study of the genetic and environmental epidemiology of childhood leukemia in this population. The specific aims of this pilot study are to: (1) identify and enroll 50 children with incident childhood leukemia and 50 control children; (2) develop and administer a survey instrument to record exposure to biomass fuels, as well as other suspected and/or known epidemiologic and environmental exposures for childhood leukemia; and (3) collect diagnostic bone marrow and remission peripheral blood on cases, and saliva on cases, controls, and parents of both cases and controls for future studies. An informal aim of this project is to establish collaborative relationships and train local Guatemalan health professionals in epidemiologic research methods, and to help build a local infrastructure that will support future international research collaborations and local self-initiated research endeavors.
Since receiving the award, Dr. Morimoto has made significant progress in achieving project aims. After identifying a Guatemalan institution with an internal review board with an FWA (federal wide assurance) to review and oversee the human subjects protections of this research protocol, Dr. Morimoto successfully submitted and received approval for this pilot study protocol at all foreign and domestic institutions involved. She guided the local Guatemalan hospital administrators through the process of registering and obtaining a DUNS number (Dun & Bradstreet), registering with the System for Award Management (SAM), and completing subaward research contracts, making them eligible to be the recipient of federal research grant monies. In consultation with UCB mentors with expertise in childhood leukemia, exposure measurement of woodsmoke from cook stoves, and Guatemalan urban and rural lifestyles, she developed an online questionnaire to elicit and store relevant epidemiologic and environmental exposure information. This questionnaire was translated into Spanish and pilot tested in a number of local Guatemalan women for comprehension, ease of administration, and duration. Over the course of two trips to Guatemala, Dr. Morimoto worked with in-country collaborators to develop workable protocols for case and control recruitment and enrollment and biospecimen collection. She trained local research and laboratory staff in case and control enrollment procedures (including case and control identification, assessment of eligibility, informed consent, and response rate tracking), biospecimen collection and processing, laboratory procedures, and interview administration.
Specific Aim 3: Provide and maintain access to research data and specimens.
Material Transfer Agreements continue to provide data and specimens from the CCLS to CIRCLE research Projects 1, 2 and 3. Core A also assists investigators in preparing IRB applications. Dr. Metayer aids in selecting cases and controls from the CCLS database and biorepository that Dr. Wiemels uses for the replication sets. Dr. Metayer also provides Dr. Rappaport (Project 2) with the same set of cases and controls that Project 3 will use in a replication study.
Significance
Leukemia is the most common type of childhood cancer. About 2,400 cases of childhood leukemia (ages 0-14 years) are diagnosed annually in the United States. The etiology of childhood leukemia is complex; confirmed clinical and epidemiologic associations explain less than 10% of childhood leukemia incidence. The results of this study are providing extremely important information regarding the contribution of various environmental, and genetic factors to the risk of childhood leukemia.
Core B: Research Translation and Outreach Core
Studies and Results
Specific Aim 1: Develop a narrative that communicates the key questions, findings, and trajectory of future research to find the causes and cures for leukemia in children, to be used as the basis for the research translation and community outreach program.
The RTOC continues to focus on topics identified in previous reports. The complexity of the research, with regard to subtypes of leukemia and the ways they can be characterized, presents challenges to audiences who are primarily interested in causes. We did not engage a communications consultant, as originally planned, due to changes in the consultant’s organizational affiliation; we are conducting all tasks internally.
Over the course of several meetings focused on the development of web content that represents the scientific findings, the center management team and IAC decided that the research translation activities need to reflect findings from the California Childhood Leukemia Study (CCLS) as well as those specifically funded by the CIRCLE grant. This is because the scientific work is well integrated between these two components. The findings produced by CIRCLE supplement those from CCLS, but both are needed to tell a coherent story. This will require a broadening of the focus for research translation in the coming year. This combined work then will be identified under the new branding of Children’s Cancer and the Environment.
In Year 3, we completed the first set of videos reflecting the work of CIRCLE from a symposium in January 2012. This format has proven to be successful and to offer an intermediate form of communication between a scientific publication and web content. In Year 4, we recorded additional footage to more fully represent the diverse work of the investigators. This now is in the editing and post-production process and will be completed by the end of Year 4. This will extend the video content available about CIRCLE and related areas of investigation. We also developed a modern e-mail program that we are using for regular updates to our audiences. We have a Facebook page as well, so we are moving into the social media using Facebook, in addition to our presence at YouTube. Dr. Kyle remains active in the NIEHS Partnership for Environmental Public Health and has been selected to present a workshop at a multi-agency meeting to be held during the last week of July at RTP. She also provided a talk at a national webinar organized by the U.S. EPA.
Specific Aim 2: Engage with two non-governmental organizations that serve parents, health/child care professionals, community leaders, policy leaders, and other advocates interested in children’s environmental health to develop relevant messages and materials about childhood leukemia and opportunities and challenges of research in this field and to convey and distribute this information to key audiences.
Partnerships with the Children’s Environmental Health Network (CEHN) and the Pediatric Environmental Health Specialty Unit (PEHSU) at UCSF continue to be highly successful. The PEHSU is presenting information to clinical audiences in collaboration with the pediatric oncology expert and completed a survey about physician practices in collecting information about environmental exposures. As noted in their report, CEHN is incorporating research findings into programs, communications, and educational resources including web pages, audience-specific fact sheets, posts on Twitter and Facebook, and newsletter articles.
Specific Aim 3: Reach out to targeted earned media outlets that report in depth on health, environmental or environmental health topics to generate interest in covering the trajectory of results from research on causes of childhood leukemia, and the promise of future research.
Discussions are under way for development of a piece on the Symposium from January 2013.
Specific Aim 4: Develop relationships with voluntary organizations, such as the Leukemia and Lymphoma Society, that serve those who have cancer and their families and allies, to increase their awareness of, and possible interest in, research on causes of cancer. Provide resources of value for one or more of these organizations.
Initial contact was made with the Leukemia and Lymphoma Society, and the relationship will be developed into Year 5. During the external review conducted in the fall of 2012, we discussed working with the healthy and smoke free housing communities and organizations to share findings particularly about relationships between smoking and leukemia and also about the contaminants found in dust and their potential health significance for childhood leukemia and potentially other diseases as well. Contact has been made with the Coalition for Healthy Homes, and future contacts and interactions will continue through Year 5.
Specific Aim 5: Develop and convey briefings and updates about advances in knowledge and potential future benefits of research on childhood leukemia to state and national policy and stakeholder audiences, including the California Department of Public Health and the California Environmental Protection Agency, using web-based communication strategies as well as directly targeted communications.
Based on the success of the first symposium held in January 2012, CIRCLE collaborated with the Office of Environmental Health Hazard Assessment of Cal-EPA and the PEHSU at UCSF on a second Pediatric Environmental Health Symposium in January 2013 for scientists and analysts at Cal EPA and CDPH, EPA Region 9, and non-governmental organizations. For this symposium we selected a specific theme, Cumulative Impacts and Children’s Environmental Health. This is a significant issue for OEHHA because the state is engaged in developing tools to better measure cumulative impacts and to identify highly impacted and disadvantaged communities.
Consideration of how best to integrate children’s environmental health and the significance of early life exposure are timely for this effort. Core B of CIRCLE coordinated the involvement of the children’s environmental health centers and the PEHSUs for the West Coast in this symposium, coordinating a series of conference calls among these entities to gain input for the symposium and to identify the topics that the various centers could address. All of the centers provided presentations. A pre-meeting packet and web resources were provided to support the learning process of the audiences. The meeting was distributed through webcast by Cal EPA. We recorded the presentations and will have them posted on YouTube for viewing by any interested party. This will be supported by web elements. We also are developing a manuscript summarizing the important themes of this meeting.
Dr. Kyle remains engaged in briefing government representatives and others about the implications of chemicals policy for children, for the California Green Chemistry Initiative and Chemical Information Clearinghouse. She provided comments on implications for children’s health of proposed regulations regarding use of flame retardants in furniture to the Department of Toxic Substances Control. She appeared at the Green Chemistry Conference in San Francisco in April 2013 to discuss progress on the development of the regulations and remaining obstacles. She also is part of a group advising EPA leaders on actions to implement the Toxic Substances Control Act from a children’s health perspective.
Specific Aim 6: Work with other children’s environmental health research centers to seek opportunities for combined efforts to enhance outreach and research translation for the program as a whole.
Core B has taken the lead on organizating interactions among six West Coast NIEHS/EPA funded Children’s Environmental Health Research Centers and the PEHSUs. Dr. Rob McConnell of USC also joined. The group supported the Symposium on Cumulative Impacts and Children’s Environmental Health. This prompted additional analyses at some centers, including CIRCLE, reflecting a two-way exchange. The Centers and PEHSUs also participated in a legislative briefing held at the California State Capitol on January 7, 2013. The event was attended by several legislators and many staffers and covered key issues including air pollution, autism and developmental effects, pesticides, childhood leukemia, and others. The investigators offered short presentations and responded to many questions. Feedback from the participants was very positive, and this has led to additional contacts and an invitation from the committees of jurisdiction to co-sponsor in future years. There is interest in further collaboration and coordination on research as well as on research translation. Areas of interest included possible sharing of samples, methods, policy, and synthesis of information. However, because all of the investigators are wholly occupied, strategic supplemental funding will be needed to achieve synergies.
Significance
Research translation and outreach can amplify the public benefits of the investment in research by making the results accessible to key audiences that can support both policy and behavioral changes. The health relevance comes from making both immediate results and broader understanding of results achieved through sustained research available to those who can act upon this knowledge for the public good. Our progress illustrates the potential in using a networked model of research translation and outreach to reach a more diverse audience in conjunction with partners.
Future Activities:
R834511C001: Childhood Leukemia International Consortium Studies
R834511C002: Exposure Assessment for Childhood LeukemiaSpecific Aim 1: Organize the 2013 CLIC meeting in at IARC, Lyon (October 1-3).
Specific Aim 2: Continue statistical analyses.
Specific Aim 3: Obtain CLIC cytogenetic data; compare ratios of childhood ALL/AML and APL/AML to those derived from population-based registries in corresponding countries.
Specific Aim 4: Continue statistical analyses.
Specific Aim 5: Continue website maintenance and development
Specific Aim 1: Perform measurements of cotinine, PCBs, and PBDEs in serum samples and nicotine in dust samples.
Specific Aim 2: Complete analyses of PBDEs in archived and repeated dust samples.
Specific Aim 3: Profile HSA adducts in 3-mm punches from DBS from 100 childhood leukemia cases and matched controls.
Specific Aim 4: Continue with identification of adducts detected in DBS samples.
Specific Aim 5: Continue with quantification of putative adducts in DBS samples from childhood leukemia cases and controls.
R834511C003: Prenatal Exposures, DNA Methylation & Childhood Leukemia
Specific Aim 1: We have completed this aim and have published part of the results (Lee, et al., 2012). We will publish the remainder of the results in the coming year after we have completed bisulfite sequencing of leukemia and normal pre-B cells.
Specific Aim 2: We will complete the analysis of 250 case and 250 matched control Guthrie cards for DNA methylation assessments by the Illumina Infinium method, and publish the results.
Specific Aim 3: We will perform a replication analysis this year on CCLS case/control cards. Additional cards from the California archive will be analyzed in the latter part of this year.
Core A: Administrative Core
Core B: Research Translation and Outreach CoreSpecific Aim 1: Continue administrative and research support. Pediatric Health Specialists, Drs. Mark Miller and Gary Dahl, will continue to develop short seminar series of environmental and genetic factors in childhood leukemia for clinicians. Dr. Morimoto will submit a R01 after obtaining sufficient preliminary data from her R21.
Specific Aim 2: Continue coordination of scientific agenda. Participate in NIEHS monthly conference calls with investigators from other Centers.
Specific Aim 3: Continue support for data sharing and data base maintenance
Specific Aim 1: We will continue to develop web content integrated with video content and plan to broaden the content and brand under Children’s Cancers and the Environment.
Specific Aim 2: UCB, CEHN, and PEHSU investigators will continue to roll out relevant messages.
Specific Aim 3: We will attempt to place one or articles in free media.
Specific Aim 4: We will work with our additional partners.
Specific Aim 5: We will continue to engage audiences and hope to contribute to a national briefing in Year 5.
Specific Aim 6: We will continue to support joint efforts among western centers and PEHSUs, and hope to hold a second California legislative briefing in 2014.
Journal Articles: 13 Displayed | Download in RIS Format
| Other center views: | All 52 publications | 13 publications in selected types | All 13 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Bailey HD, Fritschi L, Infante-Rivard C, Glass DC, Miligi L, Dockerty JD, Lightfoot T, Clavel J, Roman E, Spector LG, Kaatsch P, Metayer C, Magnani C, Milne E, Polychronopoulou S, Simpson J, Rudant J, Sidi V, Rondelli R, Orsi L, Kang AY, Petridou E, Schuz J. Parental occupational pesticide exposure and the risk of childhood leukemia in the offspring: findings from the Childhood Leukemia International Consortium. International Journal of Cancer 2014;135(9):2157-2172. |
R834511 (2014) R834511 (Final) |
Exit Exit |
|
|
de Smith AJ, Walsh KM, Ladner MB, Zhang S, Xiao C, Cohen F, Moore TB, Chokkalingam AP, Metayer C, Buffler PA, Trachtenberg EA, Wiemels JL. The role of KIR genes and their cognate HLA class I ligands in childhood acute lymphoblastic leukemia. Blood 2014;123(16):2497-2503. |
R834511 (2013) R834511 (Final) |
Exit Exit |
|
|
Gonseth S, Roy R, Houseman EA, de Smith AJ, Zhou M, Lee ST, Nussle S, Singer AW, Wrensch MR, Metayer C, Wiemels JL. Periconceptional folate consumption is associated with neonatal DNA methylation modifications in neural crest regulatory and cancer development genes. Epigenetics 2015;10(12):1166-1176. |
R834511 (2014) R836159 (2017) R836159 (2018) R836159 (2019) R836159C003 (2016) |
Exit Exit Exit |
|
|
Lee S-T, Xiao Y, Muench MO, Xiao J, Fomin ME, Wiencke JK, Zheng S, Dou X, de Smith A, Chokkalingam A, Buffler P, Ma X, Wiemels JL. A global DNA methylation and gene expression analysis of early human B-cell development reveals a demethylation signature and transcription factor network. Nucleic Acids Research 2012;40(22):11339-11351. |
R834511 (2012) R834511 (2013) R834511 (Final) |
Exit Exit |
|
|
Lee S-T, Muench MO, Fomin ME, Xiao J, Zhou M, de Smith A, Martin-Subero JI, Heath S, Houseman EA, Roy R, Wrensch M, Wiencke J, Metayer C, Wiemels JL. Epigenetic remodeling in B-cell acute lymphoblastic leukemia occurs in two tracks and employs embryonic stem cell-like signatures. Nucleic Acids Research 2015;43(5):2590-2602. |
R834511 (2014) R834511 (Final) |
Exit Exit |
|
|
Metayer C, Milne E, Dockerty JD, Clavel J, Pombo-de-Oliveira MS, Wesseling C, Spector LG, Schuz J, Petridou E, Ezzat S, Armstrong BK, Rudant J, Koifman S, Kaatsch P, Moschovi M, Rashed WM, Selvin S, McCauley K, Hung RJ, Kang AY, Infante-Rivard C. Maternal supplementation with folic acid and other vitamins before and during pregnancy and risk of leukemia in the offspring: a Childhood Leukemia International Consortium (CLIC) study. Epidemiology 2014;25(6):811-822. |
R834511 (2013) R834511 (2014) R834511 (Final) |
Exit Exit |
|
|
Milne E, Greenop KR, Metayer C, Schuz J, Pertridou E, Pombo-de-Oliveira MS, Infante-Rivard C, Roman E, Dockerty JD, Spector LG, Koifman S, Orsi L, Rudant J, Dessypris N, Simpson J, Lightfoot T, Kaatsch P, Baka M, Faro A, Armstrong BK, Clavel J, Buffler PA. Fetal growth and childhood acute lymphoblastic leukemia: findings from the Childhood Leukemia International Consortium (CLIC). International Journal of Cancer 2013;133(12):2968-2979. |
R834511 (2013) R834511 (Final) |
Exit Exit |
|
|
Whitehead TP, Brown FR, Metayer C, Park J-S, Does M, Petreas MX, Buffler PA, Rappaport SM. Polybrominated diphenyl ethers in residential dust: sources of variability. Environment International 2013;57-58:11-24. |
R834511 (2013) R834511 (Final) |
Exit Exit |
|
|
Whitehead TP, Crispo Smith S, Park JS, Petreas MX, Rappaport SM, Metayer C. Concentrations of persistent organic pollutants in California children’s whole blood and residential dust. Environmental Science & Technology 2015;49(15):9331-9340. |
R834511 (2014) R834511 (Final) |
Exit |
|
|
Xiao J, Lee S-T, Xiao Y, Ma X, Houseman EA, Hsu L-I, Roy R, Wrensch M, de Smith AJ, Chokkalingam A, Buffler P, Wiencke JK, Wiemels JL. PTPRG inhibition by DNA methylation and cooperation with RAS gene activation in childhood acute lymphoblastic leukemia. International Journal of Cancer 2014;135(5):1101-1109. |
R834511 (2014) R834511 (Final) |
Exit Exit Exit |
|
|
Li H, Grigoryan H, Funk WE, Lu SS, Rose S, Williams ER, Rappaport SM. Profiling Cys34 adducts of human serum albumin by fixed-step selected reaction monitoring. Molecular & Cellular Proteomics 2011;10(3):M110.004606 (13 pp.). |
R834511 (2013) R834511 (Final) |
Exit Exit |
|
|
Metayer C, Milne E, Clavel J, Infante-Rivard C, Petridou E, Taylor M, Schuz J, Spector LG, Dockerty JD, Magnani C, Pombo-de-Oliveira MS, Sinnett D, Murphy M, Roman E, Monge P, Ezzat S, Mueller BA, Scheurer ME, Armstrong BK, Birch J, Kaatsch P, Koifman S, Lightfoot T, Bhatti P, Bondy ML, Rudant J, O'Neill K, Miligi L, Dessypris N, Kang AY, Buffler PA. The Childhood Leukemia International Consortium. Cancer Epidemiology 2013;37(3):336-347. |
R834511 (2013) R834511 (Final) |
Exit |
|
|
Zhang L, Samad A, Pombo-de-Oliveira MS, Scelo G, Smith MT, Feusner J, Wiemels JL, Metayer C. Global characteristics of childhood acute promyelocytic leukemia. Blood Reviews 2015;29(2):101-125. |
R834511 (2014) R834511 (Final) |
Exit Exit |
Relevant Websites:
Center for Integrative Research on Childhood Leukemia: http://circle.berkeley.edu/ ExitChildhood Leukemia International Consortium: https://clic.brg.berkeley.edu/ Exit
Progress and Final Reports:
Original Abstract Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834511C001 Childhood Leukemia International Consortium Studies
R834511C002 Exposure Assessment for Childhood Leukemia
R834511C003 Prenatal Exposures, DNA Methylation & Childhood Leukemia
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2014 Progress Report
- 2012 Progress Report
- 2011 Progress Report
- 2010 Progress Report
- Original Abstract
13 journal articles for this center