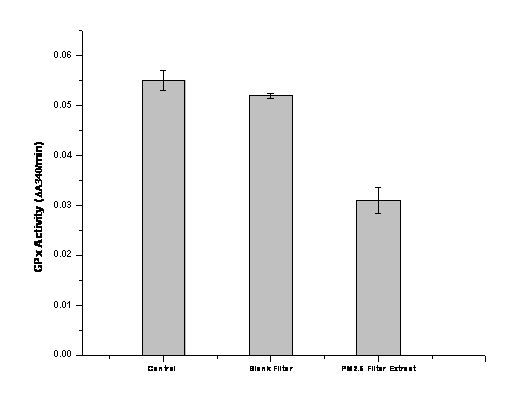Grantee Research Project Results
2009 Progress Report: Oxidative Stress Responses to PM Exposure in Elderly Individuals With Coronary Heart Disease
EPA Grant Number: R832413C004Subproject: this is subproject number 004 , established and managed by the Center Director under grant R832413
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Center for Comprehensive, optimaL, and Effective Abatement of Nutrients
Center Director: Arabi, Mazdak
Title: Oxidative Stress Responses to PM Exposure in Elderly Individuals With Coronary Heart Disease
Investigators: Delfino, Ralph , Vaziri, Nosratola D , Gillen, Dan , Staimer, Norbert , Neuhausen, Susan
Current Investigators: Delfino, Ralph , Vaziri, Nosratola D , Gillen, Dan , Staimer, Norbert , Neuhausen, Susan , Gastanaga, Victor
Institution: University of California - Irvine
EPA Project Officer: Chung, Serena
Project Period: October 1, 2005 through September 30, 2010 (Extended to September 30, 2012)
Project Period Covered by this Report: August 1, 2008 through July 31,2009
RFA: Particulate Matter Research Centers (2004) RFA Text | Recipients Lists
Research Category: Human Health , Air
Objective:
The overall goal of this study is to advance knowledge on the importance of particle size and composition to the induction of oxidative stress responses in a high-risk population of elderly people with coronary artery disease. We hypothesize that biomarkers of oxidative stress responses will be associated with indoor and outdoor home PM mass and total particle number concentration. Given the interplay between oxidative stress and inflammation, we anticipate this would support the view that PM leads to systemic inflammatory responses. We further hypothesize that biomarkers will be more strongly associated with predicted indoor exposure to PM of outdoor origin (from source tracer analyses). We also will evaluate effects of exposure to specific metals, elemental and organic carbon, and specific organic components used as source tracers. We further hypothesize that biomarker associations with ultrafine and fine PM will be better explained by chemical assays that measure reactive oxygen species and electrophilic activity. Individual susceptibility also will be assessed, including medication use and polymorphisms in genes coding for proteins involved in oxidative stress responses.
Progress Summary:
Over the period of 9 months of the fourth year of study, analysis tasks have been ongoing. Results from the complete 2 years of study panel follow-up were published this year (Delfino, et al., 2009). Below we summarized these results. This work represents cumulative progress over the 4 years of funding. Additional laboratory work for biomarkers of antioxidant activity is underway and we briefly describe methods and preliminary results below. Genotyping work and analyses of gene-environment interactions for GST M1 and T1 are complete (summarized below) and additional genetic work is planned.
Analysis of the relationship between biomarkers of antioxidant activity and air pollutants:
As proposed, the Cardiovascular Health and Air Pollution Study (CHAPS) is a panel study with daily repeated measurements of health outcomes and exposures in elderly individuals with a history of coronary artery disease. Study subjects ages 65 years or older lived in retirement communities prohibiting indoor tobacco smoke at shared locations and in buildings with common ventilation systems. As discussed in the previous report, we completed follow-up in four retirement communities in the Los Angeles air located in inland urban areas considered down-wind smog receptor sites with aged PM, but also affected by local traffic with freshly emitted PM. Subjects studied included 60 subjects ages 71-96 years with 5-12 weekly blood draws (N=578).
Over a 7-month period, each subject is followed in two 6-week blocks with blood draws for circulating biomarkers of inflammation, thrombosis, oxidative stress and antioxidant activity (described below), and measurements of exhaled nitric oxide (NO) at the end of each of 12 weeks. During the 12 weeks of panel follow-up, subjects complete daily personal digital assistant (PDA) or paper diaries that included reports of medication use.
In 2005-2006, subjects in two retirement communities were followed in four alternating 6-week phases (groups 1-2). Again in 2006-2007, subjects in another two retirement communities were followed in four alternating 6-week periods (groups 3-4). We collected 6 weeks of data in each community during a warmer period of higher photochemical activity (July-early Oct, phase 1), and 6 weeks of data in each community during a cooler period of higher air stagnation (late Oct-Feb, phase 2).
Air pollutant exposure measurements included hourly indoor (i) and outdoor (o) home pollutant gases, total particle number (PN), PM2.5 elemental carbon (EC), PM2.5 organic carbon (OC), and aethelometer black carbon (BC). We also measured size fractionated PM mass, condensation mode (quasi-ultrafine) particles, 0-0.25 mm in diameter (PM0.25), accumulation mode particles, 0.25-2.5 mm in diameter (PM0.25-2.5), and coarse mode particles, 2.5-10 mm in diameter (PM2.5-10) collected with impactor samplers.
We also estimated indoor and outdoor secondary organic carbon (SOC) from total OC and primary OC (OCpri) from total OC as described in our publication (Polidori, et al., 2007). Briefly, the contributions of primary and secondary OC to measured outdoor OC were estimated from collected OC and EC concentrations using EC as a tracer of primary combustion-generated OC (i.e., “EC tracer method”). The study average outdoor SOC accounted for 40 percent of outdoor particulate OC (40-45% in the summer and 32-40% in the winter). Air exchange rates and infiltration factors (Finf) at each site also were determined. Estimated Finf and measured particle concentrations then were used in a single compartment mass balance model to assess the contributions of indoor and outdoor sources to measured indoor OC, EC, PM2.5 and PN. We assume that indoor exposures to PM of outdoor origin are relevant to personal exposures given that people spend most of their time indoors. These data will be used in an analysis of biomarker associations to be completed this year.
Exposure-response data were analyzed with mixed linear models controlling for temperature and excluding weeks subjects had infections. As reported last year and now in press (Delfino, et al., 2009), we found many positive associations of EC, OC (mostly OCpri), BC, PN, CO and NO2 with IL-6, sTNF RII, sP-selectin, CRP and TNF-α. Generally, the strongest and most significant associations for particle mass were for quasi-ultrafine PM0.25. Most associations were strongest for longer-term averages out to the last 5-9 days. Associations of air pollutants with sP-selectin were stronger in subjects not taking platelet aggregation inhibitors while association with sTNF RII and CRP were stronger among subjects not taking statins.
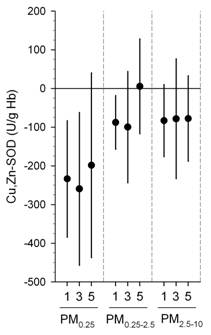
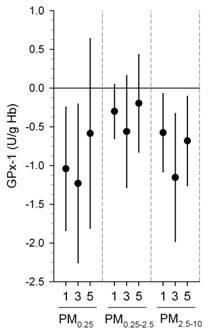
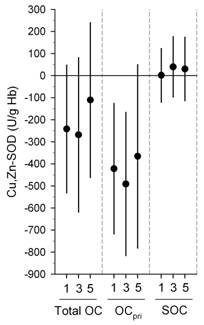
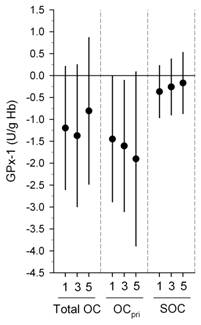
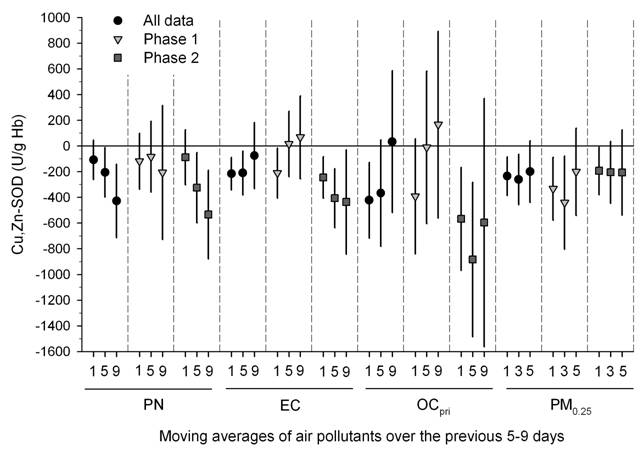
We also found significant inverse associations were for erythrocyte SOD and GPx with the same exposures above (Figures 1-3) (Delfino, et al., in press). Inverse associations were found for erythrocyte SOD and quasi-ultrafine PM0.25 plus PM0.25‑2.5 and PM2.5‑10, although associations were stronger overall for SOD with quasi-ultrafine PM0.25 (Figure 1). Inverse associations were similarly stronger for GPx with both PM0.25 and PM2.5‑10 as compared with PM0.25‑2.5 (Figure 1). Inverse associations of SOD and GPx with OC were attributable to the primary OC fraction, not the secondary fraction (Figure 2). We also found that associations were generally stronger for SOD during the cooler second phase of study compared with the warmer first phase (Figure 3). These findings suggest enzyme inactivation within erythrocytes by pollutant components or ultrafine particles. We also found within-subject inverse associations of IL-6 with GPx, and sP-selectin with SOD. This provides evidence that antioxidant enzyme inactivation may in part be responsible for pollutant-related increase in biomarkers of inflammation and thrombosis such as IL-6 and sP-selectin.
These results suggest that emission sources of primary PM2.5 OC, quasi-ultrafine particles and related particle number concentrations lead to decreased circulating erythrocyte antioxidant enzymes in elderly people with coronary artery disease. Such effects may be partly behind reported morbidity and mortality associations in the time series studies. To better understand this association, we have conducted a series of laboratory experiments in the last year as summarized below, and plan on continuing this work in the following year.
Figure 1. Associations of Cu,Zn-SOD and GPx-1 with outdoor PM size fractions.
|
|
|
Figure 2. Associations of Cu,Zn-SOD and GPx-1 with outdoor organic carbon (OC): differences by primary OC (OCpri) and secondary OC (SOC) fractions.
|
|
|
Figure 3. Associations of Cu,Zn-SOD with outdoor air pollutants: effect modification by study phase
Important new findings reported in Project 3 are results using GAPDH in an in vitro redox assay to assess the electrophilic properties of PM. This may be relevant to our suggestive finding that air pollutants may inactivate antioxidant enzymes. Because electrophiles lead to irreversible inactivation of the protein GAPDH, then it is conceivable that this is the mechanism behind our findings for Cu,Zn-SOD and GPx-1 proteins. Additional work in Year 5 is aimed to further assess this possibility using GPx-1 as the target protein. Preliminary work is discussed below.
Carbonylated proteins:
We used the OxiSelect™ Protein Carbonyl ELISA kit (Cell Biolabs, Inc., San Diego, CA) for the rapid detection and quantification of protein carbonyls in plasma samples. We measured the protein carbonyl concentrations (nmol/mg) for year two samples in 31 subjects (312 samples assayed in duplicate). In this assay, the protein concentrations of each plasma sample were determined (Pierce BCA Protein Assay kit) and diluted to 10 µg/mL in PBS. The diluted plasma samples then were adsorbed onto a 96-well plate and reacted with dinitrophenylhydrazine (DNP) to mark the carbonyl residues. The derivatized protein carbonyls were probed with an anti-DNP antibody, followed by an HRP conjugated secondary antibody. The protein carbonyl concentration in unknown plasma samples was quantified by comparing it with known concentrations of reduced and oxidized BSA standards.
Analysis of plasma protein carbonyl concentration as a marker of oxidative stress in relation to air pollutant measurements was conducted for the year two samples using mixed models as described for the other biomarkers. There were no associations between this biomarker and any of the air pollutants. As a result, we decided that this was not an informative biomarker and the assay of year 1 samples was not done. We also found no association between protein carbonyls and GPx-1 or Cu,Zn-SOD. Possible reasons for a lack of association may be the complexity of the endogenous antioxidant defense mechanisms and temporality of oxidative stress impacts. As such, it is conceivable that oxidative stress occurs transiently but changes to other biological systems such as inflammation are sustained.
Ethane and n-pentane in exhaled breath:
Briefly, we tested whether exhaled ethane or n-pentane in exhaled breath were biomarkers of exposure as opposed to biomarkers of effect, namely oxidative stress (Gorham, et al., 2009). In this study, 16 elderly subjects from the Riverside retirement community were followed over 12 weeks and their exhaled breath was analyzed using canister sampling and GC/GC-MS analysis that has been developed by the UCI Department of Chemistry, Rowland-Blake research group (Kamboures, et al., 2005). End expiratory air was collected to avoid contamination with room air and we subtracted room ethane and n-pentane from exhaled breath concentrations (residual levels were slightly positive). Exhaled hydrocarbon concentrations are normalized to exhaled CO2, which had a mean concentration of 4.3 ± 0.7 percent (1σ). Exhaled NO was used as a biomarker of airway inflammation. It was measured offline using procedures recommended by the American Thoracic Society and European Respiratory Society. Plasma carbonylated proteins were used as a marker of oxidative stress and measured as described above. Statistical analyses were performed using mixed models to investigate the within-subject relation of the inflammatory and oxidative stress biomarkers with the breath hydrocarbons. In addition, we tested the relationship between the breath hydrocarbons and indoor and outdoor gaseous air pollutants (NO, NO2, and CO).
We found that hydrocarbon concentrations in breath were correlated with room air hydrocarbon samples (Figure 4, R2 = 0.93 and 0.74, for ethane and n-pentane, respectively).
Figure 4. Exhaled breath concentrations versus room air concentrations for A: ethane l and B: n-pentane.
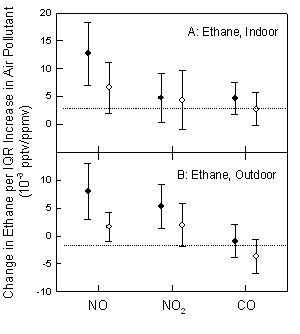
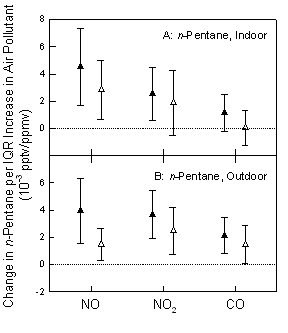
Mixed model analysis of exhaled concentrations of ethane and n-pentane from elderly subjects did not show significant associations with exhaled NO, which is a marker of airway inflammation. Additionally, neither of the exhaled hydrocarbons had positive associations with carbonylated proteins measured in the blood, which are indicative of oxidative stress. Finally, positive but weak associations between measurements of exhaled hydrocarbons (ethane and n-pentane) and air pollutants (NOx, and CO) were observed in mixed model analyses (Figure 5).
Figure 5. Relationship between change in ethane and in n-pentane at indoor (panel A) and outdoor (Panel B) environments in relation to air pollutants. Shown are the last one-hour concentration average (solid symbols) and the last 8-hour concentration average (open symbols) for NO, NO2 and CO. Estimates and 95% CI are for interquartile increases in NO, NO2 and CO. Exhaled hydrocarbon concentrations (pptv) are normalized to exhaled CO2 (ppmv).
|
|
|
Exposure to gaseous air pollutants had a weak but significant impact on exhaled ethane and n-pentane possibly secondary to residual ethane and n-pentane in the airways not adjusted for by subtracting room ethane and n-pentane from exhaled breath concentrations. It is likely that these hydrocarbon gases are positively correlated with criteria pollutant gases in both outdoor and indoor environments of the subjects. Therefore, it is possible that the association reflects exposure to air pollutant mixtures. We conclude that exhaled ethane and n-pentane are not sufficiently accurate markers of oxidative stress or airway inflammation, but instead are markers of previous exposure to air pollutants.
In vitro assay of antioxidant enzyme inactivation:
To follow up on the findings in the panel cohort, we are developing an assay for investigating the inhibitory properties of electrophilic compounds and other components (present in air and air particulate suspensions) on antioxidant enzymes such as GPx. So far, this is showing good promise as a key tool for in vitro studies with erythrocytes, some of which remain to be completed in the coming months. We have developed a rapid assay to screen for a number of electrophilic compounds (including acrolein, crotonaldehyde, and benzoquinone) using immobilized erythrocyte glutathione peroxidase (GPx) as a probe. The immobilization of GPx to the wells of a microtiter plate enables us to remove the reactive compounds simply by washing before adding the substrate mixture. These compounds include electrophiles well known to interfere with the components of the GPx assay by inhibiting glutathione reductase (i.e., the excess of electrophilic GPx inhibitors needs to be removed from the reaction mixture). This can be achieved by dialysis that is, however, a time consuming procedure. Our new assay uses a rapid 96-well format that allows us to test many samples (in replicates) simultaneously. Using this method, we were able to show that electrophilic compounds inhibit GPx in the low mmolar range (Figure 6).
We also used this assay to preliminarily test ultrafine particle suspensions (PM < 0.15 µm) collected in the field from Project 4 at the Riverside community using the VACES concentrator as described for Project 1 (Figure 7). The figure shows some inhibition of GPx by biosampler-collected UFP and SIN-1, which generates peroxynitrite by decomposing and releasing nitric oxide and superoxide simultaneously in aqueous solution. The strong inhibition by p-benzoquinone in the low mmolar range is shown again in this experiment.
Figure 6. Inhibition of GPx by Acrolein, Crotonaldehyde, and p-Benzoquinone:
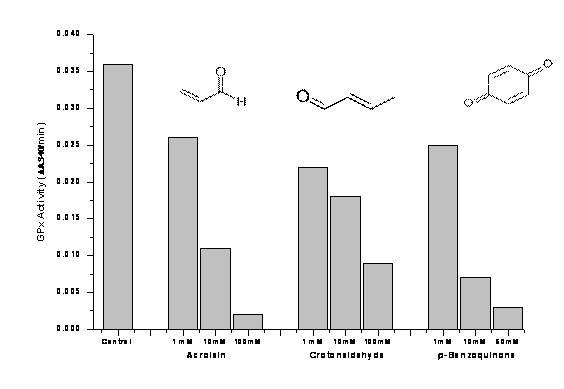
Assay Conditions: In a 96-well microtiter plate format, immobilized bovine erythrocyte glutathione peroxidase (GPx) was incubated with different electrophiles for 1 h at room temperature (pH 7.2). The excess of inhibitors was removed by washing each well with assay buffer after incubation. Enzyme activity was determined by adding a mixture of GPx co-substrate and cumene hydroperoxide and measuring the decrease in absorbance at 340 nm (oxidation of NADPH to NADP+). GPx results are the average of duplicate samples (variations between duplicates at 1 mM: < 25%, at 10 mM: < 16%, at 100 mM: < 28%).
We also tested a PM2.5 sample collected on a quartz filter and extracted into aqueous solution. Dr. Sioutas and colleagues collected two 24-hr quartz filter samples using a Harvard Impactor run at 10 L/min. Collection occurred at the Particle Instrumentation Unit (PIU) trailer of the Southern California Particle Center that is located in an urban-industrial area of USC about 150 m downwind of a major freeway (Interstate-CA-110), adjacent to a six-level parking structure, and around 3 km south of downtown Los Angeles, CA. The extract was produced by a new method developed in our lab. Briefly, the extraction procedure combines the efficient disruption of quartz filter membranes by using a high speed, reciprocating instrument (FastPrep, MP Biomedicals, Inc.) with conventional sonication. The extracted particulate suspensions were spun at 18,000 g, 15 min, 4°C before analysis. Results show GPx inhibition by the PM2.5 quartz filter extraction.
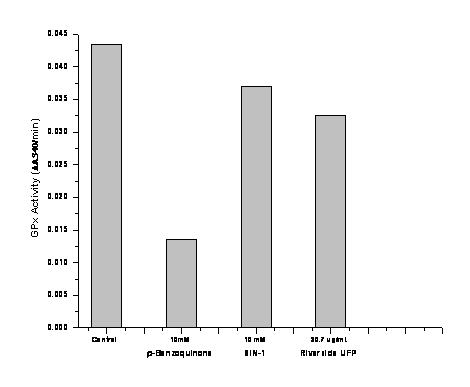
Figure 7. Inhibition of GPx by p-Benzoquinone, SIN-1, and concentrated ultrafine PM.
Assay Conditions: In a 96-well microtiter plate format, immobilized bovine erythrocyte Glutathione peroxidase (GPx) was incubated with p-benzoquinone (positive control), SIN-1 (peroxynitrite precursor) and a Biosampler suspension of UFP for 1 h at room temperature (pH 7.5). The excess of inhibitors was removed by washing each well with assay buffer after incubation. Enzyme activity was determined by adding a mixture of GPx co-substrate and cumene hydroperoxide and measuring the decrease in absorbance at 340 nm (oxidation of NADPH to NADP+). GPx results are the average of duplicate samples (variation between duplicate measurements < 15%).
Figure 8. Inhibition of GPx by fine PM collected at a site with high traffic density.
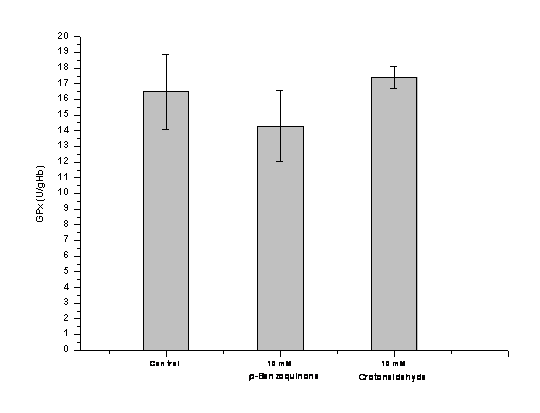
Furthermore, we successfully set up a system for direct exposure of isolated human erythrocytes to different electrophilic compounds in vitro (Figure 9), including crotonaldehyde and p-benzoquinone. We tested for GPx inhibition as in the above assay. Unlike results for the purified enzyme, mmolar concentrations of crotonaldehyde and p-benzoquinone did not affect GPx activities in erythrocytes. This may be due to inactivation at the erythrocyte membrane or binding to intracellular glutathione and erythrocyte proteins including hemoglobin. We also will test for different air particulate suspensions. This will be accomplished during the next several months.
Figure 9. GPx activity* in human erythrocytes exposed to electrophiles. *triplicate measurements, normalized to hemoglobin (Hb) concentration in erythrocytes (Units/g Hb).
Assessment of gene-environment interactions:
We previously reported genotyping work for glutathione S-transferase M1 (GSTM1) gene located on chromosome 1p13.3. The homologous recombination between two almost identical 4.2 Kb regions flanking the GSTM1 gene results in a 16Kb deletion containing the entire gene. It is referred to as a null allele of the GSTM1 gene. Similarly, the null allele was reported in the glutathione S-transferase T1 (GSTT1) gene. For both of these key enzymes involved in oxidative stress, the presence of homozygous null alleles leads to a lack of function in the enzyme. Out of 60 subjects genotyped, we found 29 subjects (48%) were homozygous GSTM1 null, and 7 subjects (12%) were homozygous GSTT1 null.
We found little to no evidence for effect modification by either GSTM1 or GSTT1 null genotypes. Results are shown for a key biomarker of inflammation (IL-6) and the two erythrocyte antioxidant enzymes (Cu,Zn-SOD and GPx-1) in relation to two air pollutants (PM0.25 and OCpri) at 3-day averaging times given that they were strongly associated with biomarkers (Tables 1-2).
Table 1. Associations of circulating biomarkers with 3-day average outdoor home PM0.25: Interaction with GSTM1 and GSTT1 null genotypes pollution.a
|
Gene variant |
IL-6 (pg/mL) |
SOD (U/g Hb) |
GPx-1 (U/g Hb) |
|
GSTM1 |
|||
|
Non-null |
0.61 (0.10, 1.11)* |
-280 (-550, -11)* |
-0.77 (-2.16, 0.61) |
|
Null |
0.64 (0.13, 1.16)* |
-234 (-508, 40) |
-1.77 (-3.21, -0.33)* |
|
p-value for interaction |
0.91 |
0.80 |
0.31 |
|
GSTT1 |
|||
|
Non-null |
0.66 (0.27, 1.05)* |
-260 (-470, -50)* |
-1.36 (-2.45, -0.26)* |
|
Null |
0.38 (-0.62, 1.39) |
-261 (-801, 279) |
-0.41 (-3.11, 2.29) |
|
p-value for interaction |
0.61 |
0.99 |
0.52 |
* p < 0.05 for the specific strata
a Regression coefficients (95% confidence intervals) are for the expected change in the biomarker associated with an interquartile increase in PM0.25 (7.0 µg/m3), adjusted for 3-day average temperature. Stratified results are from product term models of the air pollutant by gene variant. Results for SOD and GPx-1 are normalized to hemoglobin (Hb) concentration in erythrocytes (Units / g Hb).
Table 2. Associations of circulating biomarkers with 3-day average outdoor home primary OC: Interaction with GSTM1 and GSTT1 null genotypes pollution.a
|
Gene variant |
IL-6 (pg/mL) |
SOD (U/g Hb) |
GPx-1 (U/g Hb) |
|
GSTM1 |
|||
|
Non-null |
0.74 (-0.01, 1.48) |
-542 (-973, -111)* |
-1.73 (-3.62, 0.16) |
|
Null |
0.78 (-0.03, 1.59) |
-425 (-907, 57) |
-1.37 (-3.57, 0.83) |
|
p-value for interaction |
0.93 |
0.72 |
0.81 |
|
GSTT1 |
|||
|
Non-null |
0.80 (0.20, 1.39)* |
-495 (-846, -144)* |
-1.65 (-3.22, -0.08)* |
|
Null |
0.54 (-0.94, 2.03) |
-470 (-1309, 369) |
-1.30 (-5.05, 2.44) |
|
p-value for interaction |
0.76 |
0.96 |
0.86 |
* p < 0.05 for strata
a Regression coefficients (95% confidence intervals) are for the expected change in the biomarker associated with an interquartile increase in estimated primary OC (4.0 µg/m3) , adjusted for 3-day average temperature. Stratified results are from product term models of the air pollutant by gene variant. Results for SOD and GPx-1 are normalized to hemoglobin (Hb) concentration in erythrocytes (Units / g Hb).
Relationship of biomarkers to ROS production by PM using in vitro bioassays of concentrated particles collected at indoor and outdoor retirement community sites:
The bioassay methods and results are discussed in Project 3. Linear mixed effects models were used to analyze relationships of biomarkers in 60 subjects to in vitro bioassay activity for PM, adjusted for between-subject group and between-phase exposure effects, and temperature at the same averaging time as the air pollutant, and excluding person-weeks with acute infectious illnesses. We analyzed biomarkers that were informative in the main analysis described above, representing both systemic inflammation and erythrocyte antioxidant activity.
There was almost no evidence that biomarkers of effect were associated with either DTT or DHBA activity measured using either concentrated fine PM2.5 or ultrafine PM0.15 (Table 3). We found only one significant inverse association between Cu,Zn-SOD and DHBA activity in year 1 fine PM. This finding is no more than expected by chance.
The lack of associations is probably due to the limited sampling periods for concentrated particles from around 9:00-14:00 hours on the Thursdays and Fridays before the Friday blood draws. There also were no clear associations between biomarkers and the air pollutant exposures measured in the 8 hours preceding the blood draw (similar time frame to Friday VACES samples). On the other hand, we found numerous significant associations found for longer-term multi-day average air pollutant exposures. Statistical power also was limited by the restriction of each analysis of fine (year 1 panel) and ultrafine PM (year 2 panel) to half of the study population. In addition, there may have been unknown problems in sample quality or in some assays since there were many samples with 0 activities for DTT. The reasons for this are unknown.
The null results and suspected reasons are helpful in planning future studies. We first need to understand the determinants of variability in the in vitro assays that may be due to sample handling or experimental conditions. For epidemiologic research, sampling periods should be over longer durations of time prior to outcome measurements rather than the several hours employed in the present study. The sampling should be continuous over multiple days (say 5 days) prior to the health measurement. These design characteristics were not a part of the present study primarily due to limitations in current technology for the collection and assay of concentrated PM.
Table 3. Associations of circulating biomarkers with in vitro DTT and DHBA activity of concentrated fine and ultrafine PM.a
|
In vitro assay |
IL-6 (pg/mL) |
sP-selectin (ng/mL) |
TNF-RII (pg/mL) |
SOD (U/g Hb) |
GPx-1 (U/g Hb) |
|
Fine PM (Yr 1)b |
|||||
|
DTT indoor |
0.05 (-0.48, 0.58) |
1.35 (-1.48, 4.18) |
90 (-138, 319) |
-221 (-547, 104) |
-0.73 (-1.91, 0.44) |
|
DTT outdoor |
-0.28 (-0.71, 0.15) |
0.90 (-1.42, 3.22) |
95 (-98, 289) |
-271 (-548, 7) |
-0.53 (-1.53, 0.47) |
|
DHBA indoor |
-0.23(-1.08, 0.62) |
2.85 (-1.75, 7.45) |
178 (-231, 589) |
-691 (-1286, -96)* |
-1.16 (-3.32, 1.00) |
|
DHBA outdoor |
-0.34 (-0.98, 0.29) |
0.28 (-3.16, 3.73) |
145 (-164, 455) |
-315 (-743, 113) |
-0.77 (-2.36, 0.83) |
|
Ultrafine PM (Yr 2)b |
|||||
|
DTT indoor |
-0.32 (-0.91, 0.27) |
-8.18 (-23.1, 6.76) |
-177 (-564, 211) |
376 (-351, 1103) |
1.36 (-3.70, 6.43) |
|
DTT outdoor |
-0.32 (-0.98, 0.33) |
-3.27 (-19.2, 12.7) |
-206 (-609, 197) |
571 (-210, 1352) |
-1.53 (-6.63, 3.56) |
|
DHBA indoor |
-0.17 (-0.49, 0.15) |
-5.62 (-13.7, 2.43) |
1 (-206, 208) |
260 (-130, 650) |
1.95 (-0.84, 4.75) |
|
DHBA outdoor c |
-0.34 (-1.01. 0.33) |
-2.22 (-18.4, 14.0) |
-212 (-628, 204) |
745 (-56, 1546) |
-1.10 (-6.36, 4.16) |
* p < 0.05
a Regression coefficients (95% confidence intervals) are for the expected change in the biomarker associated with a one nmoles/min/µg change in DTT and DHBA activity.
b There were 29 subjects in year 1 and 31 subjects in year 2.
c Models do not include an extreme DHBA outlier = 0.625 nmoles/m3/min.
Future Activities:
Although experimental data point to redox active particle components from fossil fuel combustion, most past epidemiologic studies have relied solely on PM2.5 mass data because they are freely available from local government regulatory agencies. Aside from our own research in Project 4 (Delfino, et al., 2008; Delfino, et al., in press), there are no data on the relative cardiovascular health effects of two basic classes of organic PM, primary and secondary organic aerosols, which are expected to differ in their redox activity. We found that associations of circulating biomarkers of inflammation with PM2.5 SOA were weaker than PM2.5 primary organic carbon (OC) and nonsignificant, and this contributed to weaker associations for total OC. This novel finding suggests that associations were attributable to primary combustion aerosols, which are largely from automobile and truck traffic in the Los Angeles area.
To substantiate these findings, in the coming year we will test associations using alternate characterization of PM sources and composition for two PM size fractions (< 0.25 and 0.25-2.5 µm). We will evaluate differences in association between PM fractions estimated with chemical mass balance models using component tracers for traffic exhaust (hopanes and steranes) and photochemically generated PM (water soluble organic carbon). We also will evaluate associations with transition metals known to induce oxidative stress through Fenton reactions. Metals were measured in three size fractions (< 0.25, 0.25-2.5, and 2.5-10 µm). These data have just been assembled over the last year by coinvestigators at the subcontract site at the University of Southern California, Department of Civil and Environmental Engineering (Constantinos Sioutas and consultant Dr. James Schauer at the University of Wisconsin-Madison, Environmental Chemistry and Technology Program). Two papers below have been recently accepted for publication using some of these data (below). The epidemiologic analysis using the composition data is planned for year 5.
Arhami M, Minguillón MC, Polidori A, Schauer JJ, Delfino RJ, Sioutas C. Organic compound characterization and source apportionment of indoor and outdoor quasi-ultrafine PM in retirement homes of the Los Angeles basin. Indoor Air, in press.
Polidori A, Cheung KL, Arhami M, Delfino RJ, Schauer JJ, Sioutas C. Relationships between size-fractionated indoor and outdoor trace elements at four retirement communities in southern California. Atmospheric Chemistry and Physics, in press.
Given results in large part from the SCPC, there is sufficient reason to believe that unregulated ultrafine PM is capable of inducing the greatest amount of inflammation and oxidative stress per unit of PM mass compared with larger particle size fractions currently regulated by the US EPA. In project 4 we found that associations with biomarkers were generally strongest for PM < 0.25 µm than for larger particles, supporting hypotheses regarding ultrafine PM (Delfino, et al., 2008; Delfino, et al., submitted). Therefore, we also anticipate that associations for the source tracers and for metals will be strongest for PM < 0.25 µm.
Other outcomes in addition to the biomarkers (e.g., blood pressure) will be tested as well for gene-environment interaction. The expectation is that results will be useful to estimate sample sizes in future panel studies aiming to evaluate gene-environment interactions.
Work on blood pressure responses largely funded by NIEHS is completed and shows positive associations between ambulatory blood pressure in 64 subjects (N=6,539 measurements) and air pollutant exposure at the retirement communities (Delfino, et al., submitted). To further enhance this work, we will incorporate data related to antioxidant/oxidative stress responses from Project 4 to better understand potential differences in responses between individuals. The work of coinvestigator N. Vaziri and others suggest that oxidative stress and hypertension are part of a self-perpetuating cycle (Vaziri and Rodríguez-Iturbe, 2006). In supplemental work over the next year, we will evaluate whether individuals with higher levels of GPx-1 and Cu,Zn-SOD activities in erythrocytes are less likely to show associations between air pollutants and blood pressure.
Finally, we will submit a manuscript reporting Project 4 results evaluating biomarker associations with indoor PM of indoor vs. outdoor origin using a variety of CMB methods. We have already found that associations were generally stronger for indoor PM of outdoor origin (EC, PN, PM2.5) as compared with uncharacterized PM that includes PM from indoor sources such as cooking. This was reported previously for the first of 2 years of data (Delfino, et al., 2008) and we have confirmed it with the whole 60-subject panel (unpublished data).
References:
Kamboures MA, Blake DR, Cooper DM, Newcomb RL, Barker M, Larson JK, Meinardi S, Nussbaum E, Rowland FS. Breath sulfides and pulmonary function in cystic fibrosis. PNAS 2005;102:15762-15767.
Polidori A, Arhami M, Delfino RJ, Allen R, Sioutas C. Indoor-outdoor relationships, trends and carbonaceous content of fine particulate matter in retirement communities of the Los Angeles basin. J Air Waste Manage Assoc 2007;57:366-379.
Vaziri ND, Rodríguez-Iturbe B. Mechanisms of disease: oxidative stress and inflammation in the pathogenesis of hypertension. Nat Clin Pract Nephrol. 2006;2:582-593.
Journal Articles on this Report : 7 Displayed | Download in RIS Format
| Other subproject views: | All 35 publications | 15 publications in selected types | All 15 journal articles |
|---|---|---|---|
| Other center views: | All 241 publications | 157 publications in selected types | All 157 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Arhami M, Polidori A, Delfino RJ, Tjoa T, Sioutas C. Associations between personal, indoor, and residential outdoor pollutant concentrations:implications for exposure assessment to size-fractionated particulate matter. Journal of the Air & Waste Management Association 2009;59(4):392-404. |
R832413 (2009) R832413 (Final) R832413C001 (2009) R832413C001 (Final) R832413C004 (2009) R832413C004 (2010) R832413C004 (Final) |
Exit Exit |
|
|
Arhami M, Minguillon MC, Polidori A, Schauer JJ, Delfino RJ, Sioutas C. Organic compound characterization and source apportionment of indoor and outdoor quasi-ultrafine particulate matter in retirement homes of the Los Angeles Basin. Indoor Air 2010;20(1):17-30. |
R832413 (2009) R832413 (Final) R832413C001 (2009) R832413C001 (Final) R832413C004 (2009) R832413C004 (2010) R832413C004 (Final) |
Exit Exit Exit |
|
|
Delfino RJ, Staimer N, Tjoa T, Polidori A, Arhami M, Gillen DL, Kleinman MT, Vaziri ND, Longhurst J, Zaldivar F, Sioutas C. Circulating biomarkers of inflammation, antioxidant activity, and platelet activation are associated with primary combustion aerosols in subjects with coronary artery disease. Environmental Health Perspectives 2008;116(7):898-906. |
R832413 (2007) R832413 (2008) R832413 (2009) R832413 (Final) R832413C001 (2007) R832413C001 (2008) R832413C001 (Final) R832413C004 (2007) R832413C004 (2008) R832413C004 (2009) R832413C004 (2010) R832413C004 (Final) |
|
|
|
Delfino RJ, Staimer N, Tjoa T, Gillen DL, Polidori A, Arhami M, Kleinman MT, Vaziri ND, Longhurst J, Sioutas C. Air pollution exposures and circulating biomarkers of effect in a susceptible population: clues to potential causal component mixtures and mechanisms. Environmental Health Perspectives 2009;117(8):1232-1238. |
R832413 (2009) R832413 (Final) R832413C001 (2009) R832413C001 (Final) R832413C004 (2009) R832413C004 (2010) R832413C004 (Final) |
|
|
|
Gorham KA, Sulbaek Andersen MP, Meinardi S, Delfino RJ, Staimer N, Tjoa T, Rowland FS, Blake DR. Ethane and n-pentane in exhaled breath are biomarkers of exposure not effect. Biomarkers 2009;14(1):17-25. |
R832413 (Final) R832413C004 (2009) R832413C004 (2010) R832413C004 (Final) |
Exit Exit |
|
|
Polidori A, Cheung KL, Arhami M, Delfino RJ, Schauer JJ, Sioutas C. Relationships between size-fractionated indoor and outdoor trace elements at four retirement communities in southern California. Atmospheric Chemistry and Physics 2009;9(14):4521-4536. |
R832413 (2009) R832413 (Final) R832413C001 (2009) R832413C001 (Final) R832413C004 (2009) R832413C004 (2010) R832413C004 (Final) R833743 (Final) |
Exit Exit |
|
|
Shinyashiki M, Rodriguez CE, Di Stefano EW, Sioutas C, Delfino RJ, Kumagai Y, Froines JR, Cho AK. On the interaction between glyceraldehyde-3-phosphate dehydrogenase and airborne particles:evidence for electrophilic species. Atmospheric Environment 2008;42(3):517-529. |
R832413 (2008) R832413 (2009) R832413 (Final) R832413C001 (2008) R832413C001 (Final) R832413C003 (2007) R832413C003 (2008) R832413C003 (2010) R832413C003 (Final) R832413C004 (2009) R832413C004 (2010) |
Exit Exit Exit |
Supplemental Keywords:
Health effects, human health, sensitive populations, dose-response, enzymes, particulates, epidemiology, environmental chemistry, modeling, Health, RFA, Air, Scientific Discipline, Health Risk Assessment, Risk Assessments, particulate matter, Biochemistry, Ecology and Ecosystems, human exposure, airborne particulate matter, cardiovascular vulnerability, oxidative stress, particulates, vascular dysfunction, atmospheric particulate matter, air pollution, cardiovascular disease, human health risk, human health effects, elderly adults, airway diseaseProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R832413 Center for Comprehensive, optimaL, and Effective Abatement of Nutrients Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R832413C001 Contribution of Primary and Secondary PM Sources to Exposure & Evaluation of Their Relative Toxicity
R832413C002 Project 2: The Role of Oxidative Stress in PM-induced Adverse Health Effects
R832413C003 The Chemical Properties of PM and their Toxicological Implications
R832413C004 Oxidative Stress Responses to PM Exposure in Elderly Individuals With Coronary Heart Disease
R832413C005 Ultrafine Particles on and Near Freeways
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2011
- 2010 Progress Report
- 2008 Progress Report
- 2007 Progress Report
- 2006 Progress Report
- Original Abstract
15 journal articles for this subproject
Main Center: R832413
241 publications for this center
157 journal articles for this center