Grantee Research Project Results
Final Report: Design of Novel Petroleum Free Metalworking Fluids
EPA Grant Number: R831457Title: Design of Novel Petroleum Free Metalworking Fluids
Investigators: Hayes, Kim F. , Skerlos, Steven J.
Institution: University of Michigan
EPA Project Officer: Aja, Hayley
Project Period: January 1, 2004 through December 31, 2006
Project Amount: $325,000
RFA: Technology for a Sustainable Environment (2003) RFA Text | Recipients Lists
Research Category: Pollution Prevention/Sustainable Development , Sustainable and Healthy Communities
Objective:
Metals machining is an important industrial sector in the United States that relies heavily on metalworking fluids (MWFs) as coolants, lubricants, and corrosion inhibitors. MWFs have traditionally been based on a petroleum feedstock emulsified in water, raising concerns about environmental degradation and toxicity throughout their life cycle. The objectives of this research was 1) to design 100% petroleum-free metalworking fluids with equal or greater performance when compared with traditional metalworking fluids, and 2) to explore the feasibility of substituting water with supercritical carbon dioxide. Both bio-based bulk lubricants and extreme pressure lubricants were considered so that the resulting fluids would have less toxicity, environmental burden, and offer renewable alternatives to meet the increasingly stringent MWF disposal limits required by the federal governments and the international community.
Summary/Accomplishments (Outputs/Outcomes):
Surfactant Selection for Vegetable Oil Microemulsions
The severity of machining operations can vary widely depending on workpiece material and operation leading to the selection of one of four types of MWFs as appropriate: straight oils, soluble oils, semi-synthetics, and synthetics. Since semi-synthetics have the greatest share of the market (40%), this research was focused on the design of vegetable-based semi-synthetic MWFs. Semi-synthetic MWFs are oil-in-water microemulsions, where surfactants play the key role in dispersing and stabilizing the oil in water. Formulation of petroleum-based semi-synthetic MWFs has long been carried out using empirical rules developed through trial and error experience, as well as using the HLB (hydrophile-lipophile balance) method. To our knowledge the fundamental structure-stability relationships driving surfactant selection for semi-synthetic MWFs, especially bio-based MWFs, had yet to be identified in the academic literature when we began this study. To facilitate the development of bio-based MWFs, this research started with the investigation of the relationship between surfactant chemistry and microemulsion stability for the primary candidate vegetable oils being considered for MWFs.
In this research, three representative vegetable oils were investigated: canola oil, soybean oil, and trimethylolpropane trioleate. Commercially available surfactants from the major classes used by MWF manufacturers were also selected for investigation. Anionic surfactants were selected from six different classes: fatty acid soaps, alcohol sulfates, alcohol ether sulfates, alkane sulfonates, alkyl aryl sulfonates, and sulfo-carboxylic esters. Nonionic surfactants were selected from four classes: ethoxylated alcohols, ethoxylated glyceryl esters, polysorbitan esters, and alkyl polyglucosides. Individual surfactants were investigated for their emulsion characteristics as well as surfactant combinations. Since surfactant combinations are generally more effective and efficient emulsifiers, they were given the majority of attention. Such combinations contain a “primary” surfactant as well as a “complementary” surfactant or co-surfactant which works synergistically to increase the amount of oil that can be emulsified.
In the experimental phase of this research, all possible combinations of primary and co- surfactants were investigated over a wide range of concentrations. To represent the different concentration combinations for a given primary and co-surfactant, a formulation triangle was considered to be the most effective way to represent the wide range of system compositions, with points within it representing formulations with different oil and surfactant molar fraction. One formulation was produced at each concentration within a given composition triangle (Figure 1). For each of these points, three metrics of emulsion stability were monitored, viz., visual inspection, particle size distribution, and light transmittance.
Over 1000 MWF formulations were produced to cover all combinations of primary and co-surfactant, with each combination being investigated at 10 concentration combinations of oil, primary surfactant, and complementary surfactant. Noting that the triangle represents the formulation of the MWF concentration, and that the concentrate is typically diluted to approximately 5% by mass in water, the molar ratio of oil to surfactant in MWF microemulsions was typically found to be about 1:1. Therefore, the results of the experiments can be interpreted in the context of swollen micelle systems. In such systems, a stable dispersion of oil in water can only be achieved when the specified oil concentration in MWF is below the oil solubilization limit of the surfactant solution, which is determined by the product of the solubilization capacity of the micelle (amount of oil solubilized per micelle) and the surfactant solubility (number of micelles that can be dispersed per volume without coalescence or aggregation). Interpreting the results of experiments from the perspective of swollen micelle systems, the following guidelines were established for selecting surfactants to achieve stable vegetable oil in water MWFs:
- Surfactant packages consisting of one nonionic surfactant and one water soluble co-surfactant (either anionic or nonionic) are preferable to single surfactant systems in order to reduce the amount of surfactant needed.
- A nonionic surfactant with tail length at least 16 and an intermediate size of head group, such as ethoxylated glyceryl ester with C=18 and EO=20, is preferable.
- The co-surfactant should have a tail length similar to that of the nonionic surfactant (e.g., the tail length difference should be less than 6). This complementary surfactant can be anionic or nonionic depending on considerations such as foaming, hard water stability and waste treatability.
Experimental results show that the above guidelines apply to all the three representative vegetable oil investigated. To use these guidelines to select surfactants based on bio-based constituents rather than petroleum, it can be noted that among all the surfactants used in this research, only the anionic surfactants of the sulfonate class are currently manufactured exclusively from petroleum feedstock. All other surfactants can be manufactured without using petroleum although most are still at least partially derived from petroleum. To replace sulfonates, one can use an anionic surfactant from the classes of fatty acid soap, alcohol sulfate, alcohol ether sulfate, or sulfo-carboxylic ester. Alternatively, one can use a suitable nonionic surfactant available from any of the chemical classes considered in this paper. For other additives that are generally present in semi-synthetic MWF formulations such extreme pressure (EP) additives, corrosion inhibitors, and chelating agents, research is ongoing to develop bio-based alternatives. The combinations of vegetable oil and bio-based surfactant packages investigated in this research serve as a starting point for the development of 100% petroleum-free formulations. It is expected that the guidelines developed here will facilitate the transition from petroleum-based MWFs to renewable bio-based MWFs in machine tool industry.
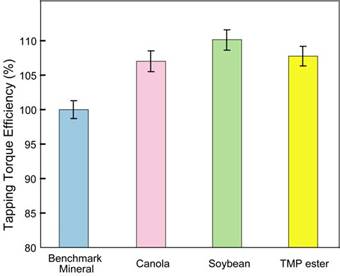
With the characteristics of MWF microemulsion systems revealed, it was necessary to determine whether the newly established bio-based MWFs had equal or better manufacturing performance when compared to existing mineral oil based MWFs since the transition to bio-based MWFs will not occur if manufacturing performance is compromised. In Figure 2, the vegetable oil based MWFs developed in this research are compared with the benchmark mineral oil based MWF using the tapping torque efficiency metric, where higher efficiency is indicative of improved manufacturing performance. It is observed that higher manufacturing performance is achieved for vegetable oil based fluids relative to the mineral oil MWFs with soybean oil based fluid having the highest performance.
Besides manufacturing performance, foaming tendency of the newly designed vegetable oil based MWFs were evaluated and compared with the benchmark fluid. Foams are undesirable since they can reduce the cooling and lubrication properties of MWFs and shorten the life of pumps due to cavitation. Experimental results indicate that emulsions formed using a high concentration of surfactants, such as in the cases of single surfactant systems or polysorbitan ester as the primary surfactant, tended to create long lasting foams as shown in Figure 3 in comparison to the benchmark fluid. From the foaming perspective, formulations produced using ethoxylated glyceryl ester (C=18/EO=20), which have the lowest surfactant to oil ratio (1:3.5), are preferred over ones produced using ethoxylated glyceryl ester (C=18/EO=40), polysorbitan ester (C=18/EO=20) and ethoxylated alcohol (C=16/EO=20), which have the highest surfactant to oil ratio (3.5:1).
Figure 3. Foaming height measured at t=0, 2 minutes, and 2 hours for MWF formulations with different oil:surfactant ratios based on ASTM D3519.
Formulation of MWF for improved microfiltration recycling
Microfiltration-based recycling of metalworking fluids (MWF) can increase the usable life of the fluids, decrease the costs of procurement and disposal, and reduce occupational health risks and environmental impact. Despite these advantages, microfiltration of MWFs is not practiced widely in industry due to a poor understanding of the relationship between MWF formulation and recycling rates. Using the knowledge gained in the surfactant formulation work we extended a mathematical model of the microfiltration process to establish the connection between MWF formulation and microfiltration performance. The goal was to recommend optimal formulation parameters that can be used to significantly improve microfiltration fluxes.
Previous research identified the fundamental mechanisms of flux decline for semi-synthetic MWF microemulsions during microfiltration recycling. Three physicochemical interactions have been proposed between semi-synthetic MWFs and membranes that drive flux decline:
- Internal Pore Restriction. Membrane fouling begins due to the coating of the membrane with free surfactants in MWFs as shown in Figure 4 Stage 1. This reduces the available pore area for permeation, exerts a drag force on permeation, and serves to capture additional hydrophobic fouling material from the MWF.
- External Pore Blocking. As additional MWF interacts with the adsorbed material at the membrane surface and inside the pores, coalescence occurs as shown in Figure 4 Stage 2. This generates aggregates that are too large to pass through the membrane pores, blocking the flow path of the MWF and effectively sealing individual pores off.
- Surface Film Formation. As pores are blocked, additional fouling material accumulates on the membrane surface as shown in Figure 4 Stage 3. This leads to a porous film at the membrane surface that imposes a significant additional resistance to MWF permeation.
The model used to describe the process outlined above is based mechanistically on adsorption, desorption, queuing theory, and coalescence kinetics. It was extended to provide an estimate of the pore blocking rate constant as a function of MWF formulation parameters. In particular, the oil to surfactant ratio and the ratio of anioinic to nonionic surfactants was explored. It is observed that surfactant packages able to produce high emulsion stability with low adsorption to membranes improve flux performance. The model was used to derive a mathematical expression that revealed the optimal concentrations of anionic and nonionic surfactant to maximize the flux of a given MWF microemulsion (Figure 5).
- Formulate MWF for small emulsion droplet size so that a large number of droplets must coalesce before they can block pores.
- Select surfactants that result in strong repulsion between droplets. Although reducing the concentration of oil would also likely increase repulsion, and reduce the droplet size of the microemulsion, this approach would likely impact manufacturing performance and is therefore not recommended.
- Select surfactants that weakly adsorb to membranes.
Evaluation of EP Additive Performance and Activation Conditions Using Thread Forming Test
Extreme Pressure (EP) additives are commonly included in metalworking fluids formulated for severe machining operations such as drilling, grinding and forming. EP additives work by reacting with the surface of the workpiece through physical sorption, chemi-sorption, or chemical reaction. The reaction produces a thin (several nanometers), low shear strength, solid-film boundary layer between the work piece and the tool. Whether or not an EP additive reacts to form a solid film boundary layer depends not only on the chemical compositions of the workpiece and the additives, but also on the contact conditions (mainly temperature) established by the machining conditions. The complicated nature of the EP activation process makes it desirable to have a laboratory performance evaluation procedure for selection and design of environmentally friendly, bio-based EP additives.
As pointed out in previous research, performance of MWFs in field machining operations can be reasonably predicted only by using a real machining operation such as reaming, milling, drilling, or tapping. Previously, it had been shown that thread cutting tests can be used to effectively evaluate MWF performance for MWFs that do not contain EP additives. Although thread cutting is not an effective means to evaluate MWF performance when different EP additives are included (Figure 6), thread forming (Figure 7) is a process in which EP additives are utilized for reducing process friction. In this research, we developed a method based on the thread forming test to evaluate MWF performance when EP additives are present, and to estimate the activation temperature for the EP additive in thread forming.
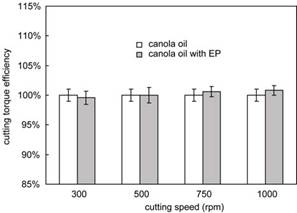
Figure 6. Comparison of cutting torque efficiency for MWFs with and without EP additive at different speeds.
Figure 7. Comparison of a cutting tap (left) and a forming tap (right).
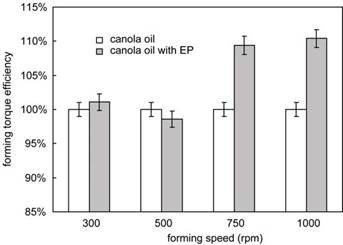
Figure 8 shows the forming torque efficiency observed at different speeds for MWF with and without an EP additive. It can be seen that the MWF including EP additive leads to a forming torque efficiency about 10% higher than the one with no EP when forming speed is higher than 500 rpm. With respect to actual torque values, a 10% higher forming torque efficiency can be thought of as a process torque decrease of about 15-20% depending on the conditions. At or below 500 rpm, the difference between forming torque efficiency for fluids with and without the EP additive is statistically insignificant. This suggests that the thread forming test can be used to evaluate EP effectiveness for a specific workpiece material if an appropriate forming speed is selected.
Figure 9 illustrates the coefficient of friction estimated based on torque data obtained from thread forming tests with and without the EP additive at 300, 500, 750, and 1000 RPM. It can be seen that at 300 and 500 RPM, there is no observed coefficient of friction difference between a MWF with and without the EP additive. However, as the forming speed increases further, a clear difference in coefficient of friction is observed. The coefficient of friction difference increases with process speed. This deviation begins between 500 and 750 RPM and indicates that there exists a critical forming speed above which the EP additives start reacting with the workpiece material to form a boundary layer to reduce friction significantly. The difference in observed with and without dimethyl disulfide suggests that this EP additive starts to react with workpiece when the forming speed increases above 500 RPM. The interpolation shown in Figure 9 suggests that the approximate “critical speed” where the temperature becomes high enough to induce the dimethyl disulfide reaction on the steel workpiece is at 580 RPM. If we can calculate the process temperature at 580 RPM, then we would expect that this activation temperature would be relatively constant for other thread forming operations if the only changes to the process were factors such as thread size or type of workpiece steel alloy. We also would expect that the activation temperature is relevant toward predicting EP activation conditions for other machining operations where EP additives are known to reduce process friction. With the temperature model it is estimated that at the critical forming speed that the contact temperature is 270 °C. This is consistent with the results reported for the reaction kinetics of dimethyl disulfide on iron foil. The activation temperature observed here (270 °C) is also within the range of activation temperature for sulfur-based EP additives.
Figure 9. Top: coefficient of friction estimated from tapping torque for MWFs with and without EP additive at different speed. Bottom: estimation of EP additive activation temperature.
To verify the relevance of the activation temperature for predicting the effectiveness of dimethyl disulfide under different thread forming process conditions, we investigated the replacement of 1018 steel with a harder 4140 steel. Naturally, the forming of harder steel should lead to a slower speed where the EP activation temperature occurs. With the tapping torque data, the contact temperature model yielded a critical speed of about 400 RPM where the activation temperature of 270 °C is achieved. To determine whether an activation speed of 400 RPM would be observed when the dimethyl disulfide additive was included in the MWF formulation, we performed thread forming tests with 4140 steel at 300, 450, and 600 RPM. We expected that at 300 RPM there would be no observable tapping torque efficiency difference between forming with and without the dimethyl disulfide EP additive. We also expected to see a significant difference at 450 RPM and an even greater difference at 600 RPM. As shown in Figure 10, the forming torque and forming torque efficiency data support these predictions.
Results above suggest that the laboratory method based on the thread forming torque test can be used to evaluate the friction reduction performance of EP additives as well as the speeds at which they are likely to activate. Through the analysis of a modest amount of data, the method yields a useful prediction of activation temperature for EP additives that has relevance to other thread forming process conditions. It is possible that the activation temperature for thread forming might serve to predict EP effectiveness under different process conditions for alternative manufacturing processes (e.g., drilling) if appropriate process models can be developed.
In the originally proposed work plan of this project, the new thread forming method would have been used to develop a family of environmentally preferable EP additives. In the course of developing the method, however, a novel, water-free MWF delivered in supercritical carbon dioxide was discovered. This new MWF had dramatically higher performance than conventional fluids and to date, no processes have been identified in which EP additives are required for the CO2 -based fluids. The expectation was that CO2 -based MWF would eliminate or at least dramatically reduce the need for EP additives in metalworking operations and so the remainder of the project focused on testing the feasibility of a CO2 -based MWF.
Feasibility of Supercritical Carbon Dioxide based MWFs
The use of water in MWFs brings with it a number of undesirable consequences such as rust, microbial growth, hardwater ion accumulation, and others that have traditionally been dealt with by including chemical additives (up to 15) in the MWF formulation. Moreover, water has only marginal ability to cool and extend tool life when compared with other cooling systems which could be devised. In short, we concluded that the environmental impacts, health impacts and technical limitations associated with aqueous MWFs could be eliminated if 1) lubrication were delivered directly to the cutting zone in minimal quantities, 2) a solvent other than water were used, and 3) lubrication were provided using inherently benign chemical components.
To achieve these goals, sprays of supercritical carbon dioxide with dissolved vegetable oils were explored as an alternative to conventional MWFs. Supercritical carbon dioxide (scCO2) is being used increasingly in industry as an alternative to traditional organic, halogenated, or aqueous solvents. The supercritical temperature and pressure of CO2 (Tc = 31.1 °C and Pc = 1070 psia) are easily achieved in industrial environments. Under these conditions CO2 is a good solvent for many materials with some vegetable-based oils being highly soluble. These characteristics make scCO2 an ideal delivery medium for MWFs because the uniform coating of oil ensures that lubrication arrives at the cutting surface and the dry ice blast that forms during the expansion provides equal or better cooling than water. Furthermore, the pressure release of CO2 also provides a chip evacuation function previously achieved using water.
Figure 11. Thread cutting experiments using MWF microemulsion (left) scCO2 (right).
Figure 11 shows photographs of thread cutting, using water- and a basic scCO2 -based MWF developed by the research team of this project. The performance of the MWFs developed during this research was initially measured via the tapping torque test. Figure 12 shows a plot of thread cutting torque efficiency. Values above 100% imply a better performance than a commercially available MWF that was used as a normalizing reference.
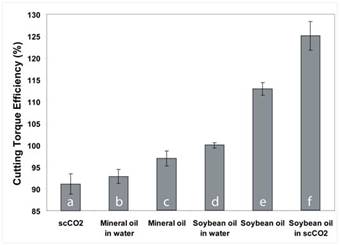
Figure 12. Cutting torque efficiency for straight oil and aqueous MWFs compared with a demonstration scCO2 -soybean oil MWF.
Figure 12 demonstrates the performance advantage of combining scCO2 and soybean oil in MWF applications. It is observed that the soybean oil/scCO2 system (f) performs on average approximately 10% better than straight soybean oil (e), 20% better than aqueous soybean oil microemulsions (d), and 30% better than straight scCO2 (c). The scCO2 results were the best ever observed by this research team in the thousands of tests conducted with aqueous and straight oil MWFs over 5 years of research. The data demonstrate that the combination of soybean oil and scCO2 performs better than either can alone, and that soybean oil and scCO2 have complimentary roles when formulated together as a MWF. The performance of scCO2 alone can be improved by adding soybean oil for lubricity, and the performance of straight soybean oil can be improved by using scCO2 for enhanced delivery of the dissolved oil to the cutting zone. Dissolving soybean oil in scCO2 also allows at least 5 times less soybean oil to be applied during each cut while achieving improved performance relative to the use of soybean oil alone. Taken together, the data indicate the feasibility of developing a new class of MWFs based upon oils dissolved or dispersed in scCO2 . Such fluids hold potential to greatly reduce environmental and occupational health concerns associated with existing straight-oil and water-based MWF formulations, as they are based on waste or renewable components and are incapable of supporting microbiological growth.
The performance advantage of combining scCO2 and soybean oil as MWFs is also demonstrated by tool wear reduction during the turning of compressed graphite iron (CGI) using polycrystalline diamond (PCD) tools. It is known that tool wear is excessive under dry cutting conditions. For the benchmark process, dry machining led to tool failures after 15-30 minutes. Improvements in tool wear rates were observed using a commercial oil-in-water mist metalworking fluid system. This system has extended tool life times beyond 3 hours. However, significant wear is still observed using the oil-in-water mist system. To further reduce tool wear in CGI turning, we explored the use of scCO2 and soybean oil based metalworking fluids (MWFs).
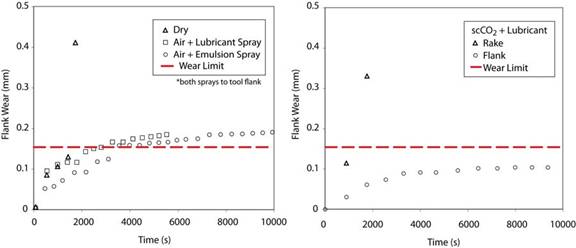
The data obtained in this investigation was unequivocal in supporting the claim that scCO2 has superior performance relative to the oil-in-water mist system, as shown in Figure 13 for a turning operation. After 3 hours of turning, the observed amount of PCD tool wear using scCO2 MWF was approximately one half that observed when turning using the oil-in-water mist system. After 3 hours of CGI turning the wear scar was less than 0.1 mm using scCO2 MWF. Signs of thermal damage were minimal compared with CGI turning using oil-in-water mist MWF. The results did suggest that the direction of the spray application had important ramifications. In the case of scCO2 + lubricant, when applied from the rake face, the spray was unable to penetrate the cutting surface and effectively dissipate heat. When applied directly to the flank face, the spray reduced heat generation locally and controlled tool wear.
Based on these encouraging tapping and tool wear results, we investigated the solubility of several lubricants in CO2 with the goal of formulating complete MWFs in CO2 . The initial work performed in this project on scCO2 -based MWFs focused on binary mixtures of soybean oil and carbon dioxide. Soybean oil was selected initially because CO2 has been employed as a replacement solvent in soybean oil extraction and the solubility behavior of the mixture has been well studied. But several other lubricant types are commercially significant in MWF formulations. To cover these other MWF formulations, in addition to soybean oil other lubricants were selected: polyalpha olefin (PAO), polyalkylene glycol (PAG), trimethylolpropane trioleate ester (TMP), and mineral oil. The solubilities of these MWF lubricants in CO2 were explored to develop a library of data for common MWF lubricants. The analysis performed indicates that even though significant differences in lubricant + CO2 solubility are found, the machining performance results are not linearly correlated with solubility. Instead, the performance of CO2 -based MWF is found to depend more on the phase composition of the spray than on differences in relative solubility. Furthermore, the experiments conducted with PAG (Figure 14) show a dramatic decrease in performance when the lubricants are dissolved in CO2 but a 10% better performance when CO2 is dissolved in the lubricant compared to lubricant alone. This increase in performance is likely due to better penetration of the CO2 in PAG into the contact zone between the chip and the tool to reduce cutting forces and wear. In general we conclude that large performance improvements are possible using MWF based on scCO2 but that phase behavior must be controlled to match the requirements of the machining operation.
The results in Figure 14 were obtained using a lubricant rich phase from the bottom of a high-pressure reactor. This mixture of CO2 dissolved in lubricant is better suited for slower machining processes like tapping that require more lubrication. The tool wear results in Figure 13 obtained using a spray of CO2 rich phase from the top of the high-pressure reactor showed that this mixture provided excellent cooling and little lubrication, important properties for a turning operation in which tool wear is controlled through effective heat removal rather than lubrication. These results also support the notion that it may be possible to tune the level of lubricant or CO2 delivered to the cutting zone in a CO2 -based fluid without changing fluids for different machining applications in which cooling is more important than lubrication or vice versa.
Figure 14. Thread forming results when cutting using straight PAG and A. PAG in vapor phase CO2 and B. CO2 dissolved in liquid phase PAG.
Journal Articles on this Report : 5 Displayed | Download in RIS Format
| Other project views: | All 22 publications | 5 publications in selected types | All 5 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Clarens AF, Hayes KF, Skerlos SJ. Feasibility of metalworking fluids delivered in supercritical carbon dioxide. Journal of Manufacturing Processes 2006;8(1):47-53. |
R831457 (Final) |
Exit |
|
|
Clarens AF, Zimmerman JB, Keoleian GA, Hayes KF, Skerlos SJ. Comparison of life cycle emissions and energy consumption for environmentally adapted metalworking fluid systems. Environmental Science & Technology 2008;42(22):8534-8540. |
R831457 (Final) |
|
|
|
Skerlos SJ, Hayes KF, Clarens AF, Zhao F. Current advances in sustainable metalworking fluids research. International Journal of Sustainable Manufacturing 2008;1(1/2):180-202. |
R831457 (Final) |
Exit |
|
|
Zhao F, Clarens A, Murphree A, Hayes K, Skerlos SJ. Structural aspects of surfactant selection for the design of vegetable oil semi-synthetic metalworking fluids. Environmental Science & Technology 2006;40(24):7930-7937. |
R831457 (Final) |
|
|
|
Zhao F, Clarens A, Skerlos SJ. Optimization of metalworking fluid microemulsion surfactant concentrations for microfiltration recycling. Environmental Science & Technology 2007;41(3):1016-1023. |
R831457 (Final) |
|
Supplemental Keywords:
pollution prevention, green chemistry, life-cycle analysis, life cycle assessment, waste reduction, waste minimization, environmentally conscious manufacturing, metal fabricated products industry, sustainable industry/business, chemicals management, economics and business, environmental chemistry, sustainable environment, technology for sustainable environment, cleaner production/pollution prevention, pollution prevention, alternative materials, alternative metal finishing, clean technology, cleaner production, environmentally benign waste lubricant, green chemistry, machining, metal finishing, petroleum free metalworking fluids, vegetable based lubricants, supercritical carbon dioxide, Sustainable Industry/Business, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Technology for Sustainable Environment, pollution prevention, Sustainable Environment, Environmental Chemistry, Economics and Business, Chemicals Management, cleaner production/pollution prevention, petroleum free metalworking fluids, clean technology, environmentally benign waste lubricant, machining, life cycle analysis, vegetable based lubricants, alternative metal finishing, alternative materials, life cycle assessment, cleaner production, vegetable based surfactants, green chemistryRelevant Websites:
http://www.engin.umich.edu/labs/EAST/ Exit
http://www.engin.umich.edu/labs/EAST/chiplube.html
http://www.engin.umich.edu/alumni/engineer/03FW/research/vegetable
http://www.umich.edu/news/?Releases/2005/Jul05/r071805a Exit
http://www.imakenews.com/lng/e_article000432685.cfm?x=b11,0,w Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.