Grantee Research Project Results
2016 Progress Report: Toxicogenetics of tetrachloroethylene metabolism and toxicity: Using Collaborative Cross mouse population approach to address remaining gaps in human health assessments
EPA Grant Number: R835612Title: Toxicogenetics of tetrachloroethylene metabolism and toxicity: Using Collaborative Cross mouse population approach to address remaining gaps in human health assessments
Investigators: Rusyn, Ivan , Wright, Fred A.
Institution: Texas A & M University , North Carolina State University at Raleigh
EPA Project Officer: Aja, Hayley
Project Period: September 1, 2014 through March 30, 2017
Project Period Covered by this Report: September 1, 2015 through August 31,2016
Project Amount: $800,000
RFA: Susceptibility and Variability in Human Response to Chemical Exposure (2013) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Tetrachloroethylene (Perchloroethylene, PERC) is a high-production chemical of great concern to both risk assessors and public health officials worldwide. It has long been assumed that PERC metabolism and toxicity closely mimic those of trichloroethylene (TCE), a structurally similar chlorinated solvent. However, recent human health assessments of these chemicals determined that major differences in toxicokinetics and toxicodynamics exist between TCE and PERC. Furthermore, it was concluded that critical gaps remain in understanding of the human health hazard of PERC, including toxicokinetics, toxicodynamics and population variability. Thus, the long-term objective of this research proposal is to uncover the mechanistic linkages between the genome (e.g., variation in DNA sequence among individuals); metabolism (e.g., formation of organ-specific toxic intermediates); and adverse molecular events (e.g., transcriptional changes associated with toxicity) in response to PERC.
The central hypotheses of this proposal are that: (1) genetic variability-associated differences in PERC metabolism affect organ-specific toxicity of PERC and (2) a population-based experimental design utilizing Collaborative Cross (CC) can be used to exploit the variability in toxicity responses to better characterize uncertainties in human health assessments. The proposal includes three specific objectives:
Specific Objective 1: To characterize variability in the toxicokinetics of PERC by using the Collaborative Cross mouse model of the human population.
Specific Objective 2: To characterize variability in the toxicodynamics of PERC by evaluating inter-strain differences in dose-dependent effects on the liver and kidney in a sub-acute study.
Specific Objective 3: To evaluate the effects of inter-strain variability in PERC metabolism on liver and kidney toxicity in a sub-chronic study.
Progress Summary:
Most of the work in the past year has focused on the activities under proposed Specific Objectives 1 and 3. The assessment of the toxicodynamics of PERC in the acute study (24 hrs exposure) in 45 CC strains constitutes evaluation of the genetic component of inter-individual variability. In addition, we also conducted a study that explored another potential contributor to inter-individual variability in toxicokinetics and toxicodynamics of PERC, non-alcoholic fatty liver disease (NAFLD), which affects nearly 25% of the U.S. population. We hypothesized that sensitivity to PERC toxicity would be enhanced in individuals with underlying NAFLD. To expand the coverage of the toxicity pathways elicited by PERC, we also developed new sensitive analytical methods for detection of glutathione conjugation metabolites of PERC in various tissues. Concomitantly, in consultation with EPA program staff, toxicologists at the National Toxicology Program, and geneticists at NIEHS and the University of North Carolina and Texas A&M University, we decided to conduct a large-scale 90-day toxicity study with a related chemical, trichloroethylene. The latter study effectively expanded the scope of Specific Aim 3 to provide a much more informative data set to accomplish our original long-term objective of uncovering the mechanistic linkages between the genome (e.g., variants in DNA sequence among individuals); metabolism (e.g., formation of organ-specific toxic intermediates); and adverse molecular events (e.g., transcriptional changes associated with toxicity) in response to chemical exposures.
In a study of the impact of NAFLD on toxicokinetics of tetrachloroethylene in mice, we examined the effect of NAFLD on toxicokinetics of tetrachloroethylene (PERC), a ubiquitous environmental contaminant that requires metabolic activation to induce adverse health effects. Mice (C57Bl/6J, male) were fed a base diet (BD), high fat diet (HFD), or methionine/folate/choline-deficient diet (MCD) to model a healthy liver, steatosis, or nonalcoholic steatohepatitis (NASH), respectively. After 8 weeks, mice were orally administered a single dose of PERC (300 mg/kg) or vehicle (aqueous Alkamuls-EL620) and sacrificed at various time points (1-36 hours). Levels of PERC and its metabolites were measured in blood/serum, liver, and fat. Effects of diets on liver gene expression and tissue: air partition coefficients were evaluated. We found that hepatic levels of PERC were 6- and 7.6-fold higher in HFD- and MCD-fed mice compared to BD-fed mice; this was associated with an increased PERC liver: blood partition coefficient. Liver and serum Cmax for trichloroacetate (TCA) was lower in MCD-fed mice; however, hepatic clearance of TCA was profoundly reduced by HFD or MCD feeding, leading to TCA accumulation. Hepatic mRNA/protein expression and ex vivo activity assays revealed decreased xenobiotic metabolism in HFD- and MCD-, compared to BD-fed, groups. These observations are significant because they demonstrate that experimental NAFLD was associated with modulation of xenobiotic disposition and metabolism, and increased hepatic exposure to PERC and TCA. Underlying NAFLD may be an important susceptibility factor for PERC-associated hepatotoxicity.
Toxicity of PERC is mediated through oxidative and glutathione conjugation metabolites. The conjugation of PERC by glutathione-s-transferase to generate S-(1,2,2-trichlorovinyl) glutathione (TCVG), which is subsequently metabolized to form S-(1,2,2-trichlorovinyl)-L-cysteine (TCVC) is of special importance to human health. Specifically, TCVC can be metabolized to N-acetyl-S-(1,2,2-trichlorovinyl)-L-cysteine (NAcTCVC) which is excreted through urine, or to electrophilic metabolites that are nephrotoxic and mutagenic. Little is known about toxicokinetics of TCVG, TCVC, and NAcTCVC as analytical methods for simultaneous determination of these metabolites in tissues have not yet been reported. Hence, we aimed also to develop an ultra-high performance liquid chromatography electrospray ionization tandem mass spectrometry-based method for analysis of TCVG, TCVC, and NAcTCVC in liver, kidney, serum, and urine. We developed a rapid, sensitive, robust, and selective method for detection of all three analytes in every tissue examined, with limits of detection ranging from 1.8-68.2 femtomoles on column, depending on the analyte and tissue matrix. We also demonstrated that this method can be applied to quantify levels of TCVG, TCVC, and NAcTCVC in tissues from mice treated with PERC with limits of quantitation of 1-2.5 pmol/g in liver, 1-10 pmol/g in kidney, 1-2.5 pmol/mL in serum, and 2.5-5 pmol/mL in urine. This research is significant because the availability of this method is invaluable for further characterization of the glutathione conjugative pathway of PERC in vivo and improved understanding of PERC toxicity.
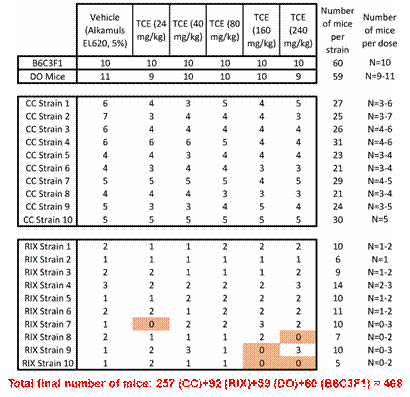
Finally, we conducted a 13-week toxicity and metabolism study of trichloroethylene in a mouse population model. We selected trichloroethylene based on the following considerations: (1) widespread human exposure to TCE and related chemicals; (2) it is classified as a known human carcinogen; (3) there are several issues critical for human health assessments of TCE that remain and include better characterization of inter-individual variability in metabolism and toxicity, and better understanding of the inter-species differences in target organ toxicity; (4) discordance between target tissues for cancer in humans (kidney – sufficient; liver and non-Hodgkin lymphoma – limited) and mice (liver, malignant lymphomas [females] and alveolar/bronchiolar adenomas) exist and require mechanistic explanation; (5) human toxicokinetics data are available; inter-strain variability in acute and sub-acute treatment-associated toxicokinetics in mice is characterized; and (6) mechanisms of toxicity are well-described but the variability in these mechanisms is poorly characterized. We note that our data showed that kidney toxicity (Kim-1 expression in renal tubular cells) has been observed in several “non-traditional” mouse inbred strains upon exposure to TCE (600 mg/kg/d; 5 days), and kidney toxicity (NGAL or lipocalin-2 in urine) has been observed in subpopulations of intercross mice after low-dose chronic exposure to TCE (1.25 ug/kg/d; 16 weeks). Thus, we proposed that a “rational” selection of strains for a 13-week study is possible because toxicity and TCE metabolism data are available for ~50 CC strains and ~20 inbred strains. Overall, the outputs of the proposed experiment were:
- Selection of several (2-3) strains for a 2-year study (i.e., select most representative “outlier” strains);
- Selection of the doses for a 2-year study;
- Characterize inter-strain variability in TCE toxicity in a 13-week study;
- Characterize inter-strain variability in TCE metabolism in a 13-week study.
We have successfully completed the in-life portion of the experiment and collected samples from the following strains and dosing groups. Sample analyses are underway.
Future Activities:
- First, we will continue exploring the impact of NAFLD on toxicodynamics of tetrachloroethylene in mouse liver and kidney. We are conducting gene expression and other toxicity phenotyping analyses from the NASH study.
- While toxicity testing is ongoing, we will continue work on the pipeline for genome-wide analysis of genetic susceptibility loci that may explain the variability in responses to PERC among CC strains. This will then be applied to the PERC toxicokinetic data to understand genetic determinants of PERC metabolism, and to the future data from the 13-week toxicity data to understand genetic determinants of treatment-associated target organ toxicity.
- We will analyze tissues from the 13-week study in three different diverse mouse populations in a way similar to that detailed in the results for the acute study. We will evaluate metabolites, organ-specific effects and quantify inter- and intra-strain variability in the collected phenotypes.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 14 publications | 9 publications in selected types | All 9 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Cichocki JA, Guyton KZ, Guha N, Chiu WA, Rusyn I, Lash LH. Target organ metabolism, toxicity, and mechanisms of trichloroethylene and perchloroethylene: key similarities, differences, and data gaps. Journal of Pharmacology and Experimental Therapeutics 2016;359(1):110-123. |
R835612 (2016) R835612 (Final) |
Exit Exit Exit |
|
|
Cichocki JA, Furuya S, Konganti K, Luo Y-S, McDonald TJ, Iwata Y, Chiu WA, Threadgill DW, Pogribny IP, Rusyn I. Impact of nonalcoholic fatty liver disease on toxicokinetics of tetrachloroethylene. Journal of Pharmacology and Experimental Therapeutics 2017;361(1):17-28. |
R835612 (2016) R835612 (Final) |
Exit Exit Exit |
Supplemental Keywords:
Genome-wide association, toxicity pathway, kidney, liver, mouse, population, tetrachloroethylene, trichloroethylene, metabolism, toxicogenetics;Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.