Grantee Research Project Results
2014 Progress Report: Neurodevelopment and Improving Children's Health following EtS exposure (NICHES)
EPA Grant Number: R835437Center: The Center for Study of Neurodevelopment and Improving Children's Health
Center Director: Murphy, Susan K.
Title: Neurodevelopment and Improving Children's Health following EtS exposure (NICHES)
Investigators: Murphy, Susan K.
Institution: Duke University
EPA Project Officer: Hahn, Intaek
Project Period: June 1, 2013 through May 31, 2018 (Extended to May 31, 2019)
Project Period Covered by this Report: June 1, 2014 through May 31,2015
Project Amount: $3,907,780
RFA: Children's Environmental Health and Disease Prevention Research Centers (with NIEHS) (2012) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
Project 1 is examining the relationship between smoke exposure during early life and neurobehavioral outcomes in children followed from prior to birth through up to age 7 years, with a particular focus on attention deficit/hyperactivity disorder (ADHD). This project also is examining the relationship between smoke exposure, ADHD and DNA methylation.
Our objective is to evaluate the associations of both ETS exposure on cognitive and neurobehavioral outcomes across early development and to examine the role of exposure-induced DNA methylation changes on these outcomes.
Aim 1: To characterize the extent and developmental timing of ETS exposure effects on cognitive and neurobehavioral outcomes in young children.
This aim will be addressed once we have recruited sufficient participants for analysis.
Aim 2: To determine the relation between DNA methylation and cognitive and neurobehavioral outcomes in young children.
This aim also relies on a larger number of individuals to be recruited into the study. MEG3 methylation data is being generated in Project 3 for a subset of the potential participants.
Project 2 is determining how exposure during early development to tobacco smoke extract and nicotine influences growth and neurobehavioral outcomes in rats and neural differentiation and neurotransmitter phenotypes in vitro. This project also is working to define the most sensitive developmental window(s) of vulnerability to tobacco smoke and nicotine exposure and will determine if dietary interventions can ameliorate the effects of these exposures.
In Project 2, we are determining with rat models and cell lines the behavioral consequences of developmental exposure to tobacco smoke constituents and the neural mechanisms.
Aim 1: Determine the behavioral phenotypes resulting from developmental nicotine and ETS exposure.
Aim 2: Determine the deficits in specific neural circuits that cause the behavioral anomalies.
Aim 3: Determine cellular and molecular mechanisms for nicotine/tobacco extract-induced developmental neurotoxicity.
Project 3 is investigating how in utero tobacco smoke and nicotine exposure in rats influences DNA methylation in the brain and blood, and how the methylation profiles in the brain relate to gene expression. These findings are being applied to the study of cord blood from our human cohort to determine if methylation patterns can be used to stratify risk of ADHD prior to onset of symptoms. ADHD-associated genes also will be examined in the in vitro models of neurodifferentiation and neurotransmission to determine associations with these phenotypes.
Aim 1: Identify ETS-related methylation targets. The cohorts of rats to be used for this aim have been generated (breeding, exposure, behavioral measures, tissue collection) in Project 2 and the tissues have been transferred to the Murphy laboratory for processing.
Our specific objectives are as follows:
- Pool equal amounts of frontal cortex and peripheral blood genomic DNA from a total of 12 adult rats in 4 groups: 1) rats exposed to placebo in utero to serve as controls, 2) rats exposed in utero to nicotine levels equivalent to that of ETS, 3) rats exposed in utero to nicotine levels equivalent to that of active maternal smoking, and 4) rats exposed in utero to tobacco smoke extract with nicotine levels equivalent to that of active maternal smoking.
- Ship pooled specimens to the University of Southern California Epigenome Center for bisulfite modification and high throughput sequencing.
- At Duke, align the paired-end reads to the most current in silico bisulfite modified Rattus norvegicus reference genome (March 2012; RGSC 5.0/rn5).
- Calculate methylation levels at individual base pair locations.
- Test for association between methylation levels and outcome variables.
- Technical validation for top hits by bisulfite pyrosequencing.
Aim 2: Identify ETS-altered methylation-expression relationships in frontal cortex. This is a future objective.
The same cohorts of rats to be used for this aim as in Aim 1 have been generated in Project 2 and the tissues have been transferred to the Murphy laboratory for processing.
Our specific objectives are as follows:
- Purify RNA from snap frozen frontal cortex; pool for transcriptome analysis.
- Assess RNA for quality and integrity.
- Submit RNA pools to the Duke IGSP Sequencing Facility for fragmentation of poly A tailed RNA, cDNA synthesis, end repair, addition of 3’ A ends, adapter ligation, PCR amplification, library validation and cluster generation.
- Align reads to the rat reference genome and to an exon-exon database.
- Combine aligned sequence reads to estimate transcript abundances.
- Validate top hits using real time RT-PCR.
- Identify those transcripts that are most strongly associated with our outcomes.
- Compare the group of differentially expressed genes to the methylation levels detected from the analysis in Aim 1.
Aim 3: Determine if DNA methylation varies with ETS dose in humans. We have designed pyrosequencing assays for human ADRA2A, DRD2, MAOA, NGF, OPRD1 and BDNF; optimization and validation are complete. We also have designed Sequenom EpiTYPER assays to screen candidate genes, including TH (3 regions), Slc6a2 (3 regions), Slc18a1 (4 regions), Slc18a2 (5 regions) in rat cerebellum (from Project 2, Slotkin) for areas of differential methylation in response to prenatal nicotine exposure (6 mg/kg/day); once identified, pyrosequencing will be used to analyze these regions in cerebellum collected longitudinally every other day from prenatal day 4 through postnatal day 21 (synaptogenesis). This will help determine if these genes undergo dynamic changes in methylation during brain development, and may help pinpoint the best timing for subsequent analyses in this project.
Our specific objectives are as follows:
- Generate methylation data in humans for 12 loci including those identified as top candidates (ADRA2A, DRD2, MAOA, SLC6A2, MEG3 [2 regions], MMP9 and OPRD1) as well as genes identified in Project 1 and/or from our preliminary data as top candidates (IGF2/H19 [2 regions], MEG3 [2 regions], NGF and BDNF), and 20 additional loci that are identified from Aims 1 and 2.
o Umbilical cord blood from 800 NEST children, selecting those with known ETS exposure and matching to controls with no reported ETS exposure.
o Peripheral blood specimens collected postnatally from the children being followed in Project 1.
- Analyze rat frontal cortex and peripheral blood from Project 2, to validate the findings from Aim 1.
- Analyze methylation in cultured neuronal cells from Project 2, to determine if genes identified from Aim 1 and candidate genes exhibit methylation changes that affect differentiation as a result of nicotine and ETS exposure.
- Ship umbilical cord blood plasma to the UCSF Clinical Pharmacology laboratory for determinations of cotinine levels.
- Genotype genes involved in detoxification (e.g., GSTP1 I105V, CYP1A1, EPHX1 Y113H SNP variants and GSTM1, GSTT1 and GSTT2 null variants) in both the mothers and the infants.
- Obtain blood lead levels for the children being followed in Project 1 using peripheral blood specimens collected at 3-5 years of age.
- Obtain gestational blood lead levels for the mothers of the children being followed in Project 1.
- Determine if relationships exist between the level of methylation measured and cord blood cotinine levels; results will be analyzed by gender.
- Determine relationships between genotypes of detoxification genes, DNA methylation, and exposure to environmental tobacco smoke.
- Analyze the methylation data in the children from Project 1 with respect to prenatal maternal lead levels and lead levels in the child at 3-5 years of age.
The Community Outreach and Translation Core has developed an educational primer about the effects of tobacco smoke on children with particular emphasis on ADHD. The primer was used as a tool to elicit interest in community participation in an Instagram contest that requires that the entry reflect what has been learned from the educational primer. The COTC now is working to determine the best social media method to educate the public about the effects of tobacco smoke, epigenetics and risk of ADHD.
Progress Summary:
Project 1:
Our focus over this past year has been to increase enrollment and refine our retention activities.
Within the NEST cohort (parent cohort) there are 759 eligible participants for NICHES Project 1. As of May 31 2015, we have invited 573 of these cohort children to participate in the study. Of these 573, we have enrolled 127 children (60% African American, 33% Caucasian, 4.7% More than one race, 1.6% Asian, 0.7% American Indian/Alaska Native) with a mean age of 4.3 years (June 1, 2014 to May 31, 2015). A total of 53 have refused participation and 374 are pending enrollment.
As of May 31, 2015, we have completed neurodevelopmental assessments of the 125 children and their mothers. For both mothers and their children, this includes assessments of executive functioning using the NIH toolbox and an IQ test. Of these children, 20 have provided blood and saliva samples, 2 children provided only blood sample and 103 children provided only saliva sample. We were unable to collect any specimen sample from two children. Collected samples are being stored and will be used to assay cotinine and for studies of DNA methylation. Stored prenatal maternal blood will be assayed for cotinine and umbilical cord samples will be assayed for DNA methylation. Assays for cotinine from prenatal maternal blood currently are being conducted and it is anticipated that they will be complete by the end of 2015.
Our current battery of neurodevelopmental tests for the participants includes the NIH Toolbox with eight tasks, the Differential Abilities Scale (DAS) with four-six subtests, Wechsler Abbreviated Scale of Intelligence –II (WASI) with four subtests, parent self-report measures that include: the Conners Adult ADHD Rating Scales (CAARS), Strengths and Weaknesses of ADHD Symptoms and Normal Behavior Rating Scale (SWAN), Strengths and Difficulties Questionnaire (SDQ), Behavior Rating Inventory of Executive Function (BRIEF), Behavior Assessment System for Children (BASC) and Parent Stress Index (PSI). During this report period, we have completed the full battery of tests on 125 mother and child dyads. Two young children did not complete the assessments and will be rescheduled to come in before their year 2 visit.
With internal funding, Dr. Fuemmeler has led the development of a unique platform for increasing recruitment, retention and maintaining engagement of study participants. The platform uses a combination of communication channels [web, email, SMS text, and interactive voice recording (IVR)] to facilitate interactive communication with cohort participants over the course of the study. The platform is programmed to semi-automatically perform many of the recruitment/retention procedures traditionally done by research assistants. For instance, the platform reads dates in our database and sends automated pre-programed messages that we developed for delivery on specific dates and times (e.g., in advance of a study visit, child’s birthday, etc.) or it sends generic messages to all participants on specific dates (e.g., quarterly requests to mailing address updates, links to a website). Messages are stored, so if the participant misses a call, they can call back at their convenience to receive the message. In addition, the interactive system can easily be programmed to collect data on specific behaviors, such as sleep duration and quality by prompting participants to respond to simple queries (e.g., “using your keypad, enter the time your baby went to bed”; “how many times did your baby wake up?”). For our ongoing studies, all procedures using this platform have been vetted through our information security office and have been approved by the IRB. We believe this method will enhance our ability to keep participants engaged throughout the study as well as in possible follow-on studies.
In addition to digital methods we are using to enhance retention, we also have been sending newsletters twice yearly, thank-you cards, and holiday greeting cards.
We also have developed RedCap databases and SAS merge codes for assessing data quality, which are run after every 15-20 participants have been enrolled (about once every other month). This helps to minimize missing data and ensure that our data are of high quality.
Project 2
This project is addressing three aims (defined above).
Neurobehavioral Toxicology in Rats: The first major neurobehavioral toxicology study has been completed in Dr. Levin’s laboratory. It determined the neurobehavioral toxicology of developmental tobacco smoke extract exposure compared with nicotine alone. This study is being prepared for peer review publication. The current study was conducted to determine in a rat model the cause-and-effect relationship between low-level exposure to nicotine or nicotine in tobacco smoke extract (TSE) and persisting neurobehavioral effects in the offspring. Young adult female Sprague-Dawley rats were implanted with osmotic minipumps (Alzet 2ML4) containing TSE or an equivalent dose of nicotine alone (0.2 mg/kg/day), which modeled second hand smoke (environmental tobacco smoke) exposure. Early developmental exposure to TSE caused significant locomotor hyperactivity in the Figure-8 apparatus during adolescence. Hyperactivity was not seen in adulthood. This mirrors the age-specific expression of hyperactivity seen in ADHD. Both tobacco smoke extract and nicotine exposure during development caused significant impairment in novel object recognition, a low motivation cognitive test. In the high motivation signal detection task, tobacco smoke extract or nicotine alone did not cause impairments in choice accuracy. This difference between performance on low vs. high motivation tasks also is seen in people with ADHD. This study shows that even low dose exposure to tobacco smoke extract during early development can have neurobehavioral effects that persist into adulthood. Overall, the test results for the TSE effects more closely correlated with the effects of the 2 mg/kg/day than the 0.2 mg/kg/day nicotine dose, which was the dose level of nicotine delivered by the TSE. This indicates that the other components of TSE greatly magnified nicotine effects on behavioral function.
The second major study is investigating the windows of exposure for low-dose tobacco smoke extract exposure effects on neurobehavioral function. TSE exposure for 10-day periods pre-mating, during the first half of gestation and during the second half of gestation are being compared with vehicle treated controls. The iPrecio pumps are being used for this study because they can be programmed to deliver TSE during the specified time periods. A very similar behavioral test battery is being used. The principal adjustment has been to test the radial-arm maze performance and attention task performance under food restricted and fed conditions to investigate the role of differential motivation on the TSE effects. This study is in progress and should be completed during the first quarter of 2016.
Training Activities: Our faculty development fellow Brandon Hall, Ph.D. has attended and presented NICHES research at a variety of important national and international conferences including those of the Society of Toxicology, The International Neurotoxicology Association, The Neurobehavioral Teratology Society and the Society for Neurosciences. He regularly participates in the Duke Integrated Toxicology and Environmental Health Seminar Series organized by Dr. Levin and made a presentation in the series this semester. He also participated in the Duke Epigenetics and Epigenomics Program Seminar Series and has completed the Duke scientific writing workshop.
In Vivo Neurochemistry: Drs. Slotkin and Seidler have completed and published work delineating the synaptic dysfunction elicited by developmental exposure to TSE. We evaluated the developmental neurotoxicity of TSE administered to pregnant rats starting preconception and continued through the second postnatal week. We simulated nicotine concentrations encountered with second-hand smoke, an order of magnitude below those seen in active smokers, and compared TSE to an equivalent dose of nicotine alone, and to a 10-fold higher nicotine dose. We conducted longitudinal evaluations in multiple brain regions, starting in adolescence (postnatal day 30) and continued to full adulthood (day 150). TSE exposure impaired presynaptic cholinergic activity, exacerbated by a decrement in nicotinic cholinergic receptor concentrations. Although both nicotine doses also produced presynaptic cholinergic deficits, these were partially compensated by hyperinnervation and receptor upregulation, effects that were absent with TSE. TSE also produced deficits in 5HT receptors in females that were not seen with nicotine. Regression analysis showed a profound sex difference in the degree to which nicotine could account for overall TSE effects: whereas the two nicotine doses accounted for 36-46% of TSE effects in males, it accounted for only 7-13% in females. Our results show that the adverse effects of TSE on neurodevelopment exceed those that can be attributed to just the nicotine present in the mixture, and further, that the sensitivity extends down to levels commensurate with second-hand smoke exposure. Because nicotine itself evoked deficits at low exposures, “harm reduction” nicotine products do not eliminate the potential for neurodevelopmental damage.
In Vitro: The published study of Drs. Slotkin and Seidler (Neurotoxicol. Teratol. 43:19-24, 2014) showed that TSE promotes neurodifferentiation, resulting in a more rapid transition from cell replication to differentiation. Consequently, it produces deficits in the number of cells while promoting cell growth, neurite formation and emergence of neurotransmitter phenotypes; this effect was not seen with comparable concentrations of nicotine, although similar results could be elicited by raising the concentration of nicotine to 10x that found in TSE. TSE also diverted neurodifferentiation toward the dopaminergic phenotype at the expense of the cholinergic phenotype, effects that would serve to disrupt the wiring of neural circuits. The newly-completed in vivo studies (see above) entirely confirmed that these direct effects of TSE on neurodifferentiation contribute to its adverse effect on brain development. In the current year, we completed and published studies attempting to ameliorate these adverse effects, using strategies based on the likely contributory mechanisms for TSE toxicity. In undifferentiated PC12 cells, TSE impaired DNA synthesis and cell numbers to a much greater extent than nicotine alone; TSE also impaired cell viability to a small extent. In differentiating cells, TSE enhanced cell growth at the expense of cell numbers and promoted emergence of the dopaminergic phenotype. Nicotinic receptor blockade with mecamylamine was ineffective in preventing the adverse effects of TSE and actually enhanced the effect of TSE on the dopamine phenotype. A mixture of antioxidants (Vitamin C, Vitamin E, N-acetyl-L-cysteine) provided partial protection against cell loss but also promoted loss of the cholinergic phenotype in response to TSE. Notably, the antioxidants themselves altered neurodifferentiation, reducing cell numbers and promoting the cholinergic phenotype at the expense of the dopaminergic phenotype, an effect that was most prominent for N-acetyl-L-cysteine. Treatment with methyl donors (Vitamin B12, folic acid, choline) had no protectant effect and actually enhanced the cell loss evoked by TSE; they did have a minor, synergistic interaction with antioxidants protecting against TSE effects on growth. Thus, components of tobacco smoke perturb neurodifferentiation through mechanisms that cannot be attributed to the individual effects of nicotine, oxidative stress or interference with one-carbon metabolism. Consequently, attempted amelioration strategies may be partially effective at best, or, as seen here, can actually aggravate injury interfering with normal developmental signals and/or by sensitizing cells to TSE effects on neurodifferentiation.
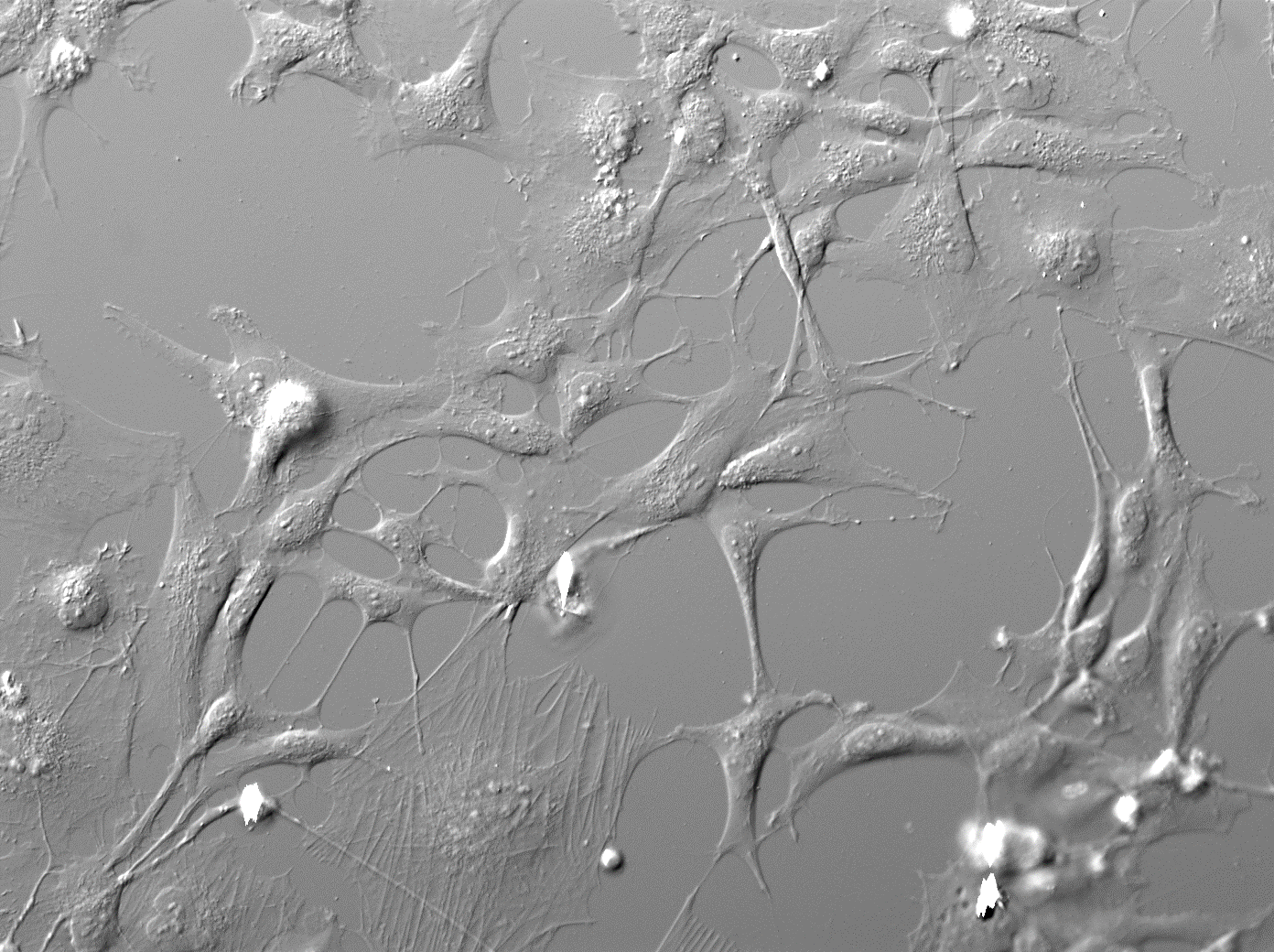
Dr. Satterwhite is developing an in vitro human neural stem cell (hNSC) model to identify a genomic/epigenomic signature for nicotine/TSE exposure and to test concordance with human tissues including brain and whole blood. We are testing the ability of simple vitamins and micronutrients to reverse nicotine/TSE-induced epigenetic change observed in vitro and in vivo. The Year 3 focus is aimed at identifying the underlying mechanisms of exposure-associated DNA methylation changes in genes of neuronal specification/dopaminergic specification pathways (SOX2, WNT1, WNT5, FOXA2, LMX1, PTX3, EN1, EN2, EPHB1, EFNB2, NURR1, PITX3, SOX6, OTX2, DAT, DRD1-5 and TH). These early targets are associated with midbrain development, axonal guidance and dopaminergic specification in the substantia nigral and mesolimbic regions. Epigenetic and genomic changes in these genes are associated with ADHD, tobacco use, autism, impulse control, addiction, Parkinson’s disease and obsessive compulsive disorder. The effect of nicotine or TSE exposure on cell cycle progression has been fully characterized; experiments to define effects on differentiation and synaptic phenotype are in progress. Nicotine (1-10 µM) induces cell cycle arrest and differentiation, whereas 1-10 µM nicotine in TSE increases apoptosis, cell death and alters cell polarity and fate. Remarkably, 1 to10 mM vitamin C reverses many of the nicotine or TSE-associated proliferation phenotypes. DIC images of hNSCs exposed to 3 µM TSE (left) or 3 mM TSE + 10 mM vitamin C (right) at 24 hours show increase in cell number, cell area, and cell perimeter in contact with other cells; EdU incorporation and live cell Hoechst imagining shows increase in cell proliferation and viability with vitamin C.

To define DNA methylation signatures for nicotine/TSE in hNSCs in vitro, hNSCs were exposed to 1 µM nicotine or 1 µM nicotine in TSE in growth medium or differentiation medium (growth factors withdrawn), and DNA methylation quantified by bisulfite pyrosequencing (Qiagen) for the ADHD-associated loci SCL6A2 (norepinephrine/dopamine transporter) and SLC6A3 (dopamine transporter) (first row below). DNA methylation was reduced at SLC6A2 in nicotine-exposed hNSCs relative to TSE or controls. In contrast, DNA methylation was reduced in SLC6A3 in both nicotine or TSE exposed hNSCs. During differentiation, DNA methylation in SLC6A2 was increased in both nicotine and TSE exposed cells relative to controls, but was unchanged at SLC6A3. DNA methylation at CHRNA4 (alpha subunit of the nicotinic acetylcholine receptor) increased ~4% in differentiating hNSCs but was unchanged by both treatments.
Nicotine or TSE induced an increase in methylation of the DMR of the maternally imprinted non coding RNA tumor suppressor H19 in proliferative phase hNSCs (second row); a decrease in methylation of the DMR of the tobacco use-associated paternally imprinted IGF2 (insulin-like growth factor 2) in differentiating cells; and a decrease in nicotine exposed differentiating hNSCs in the DMR of the imprinted and developmentally regulated PLAG1 (pleomorphic adenoma gene-like 1). These early results support that exposure to nicotine or TSE induces measurable changes in DNA methylation at genes highly relevant to both ADHD and tobacco use. We also found that if hNSCs are exposed to nicotine or TSE in the proliferative phase prior to changing to differentiation medium, the DNA methylation patterns resemble those of the earlier proliferative phase, consistent with the idea that critical windows of susceptibility to nicotine/TSE are present during human neurodevelopment.
Project 3
One modification to our plan for Aim 1 is that we intend to include both frontal cortex and hippocampus as well as peripheral blood for whole genome bisulfite sequencing. Favorable cost reductions have made this possible, and we believe hippocampus also is a highly relevant part of the brain to examine in the context of ADHD.
Work on Aim 2 is ongoing. We currently are in the process of preparing the RNA from the same rat tissues as those used to prepare DNA for Aim 1, using a simultaneous DNA/RNA preparation kit that was decided upon after extensive testing and optimization. Nucleic acid yields have been good, and the quality of the purified nucleic acids is very good. One modification to our plan is that we intend to include both frontal cortex and hippocampus for whole transcriptome profiling. Favorable cost reductions in the technology have made this possible, and hippocampus also is a highly relevant part of the brain to examine in the context of ADHD.
The Sequenom EpiTYPER system was implemented to screen for differential methylation in four genes in rat cerebellar tissue (slc6a2, slc18a1, slc18a2 and th) involved in monoamine biosynthesis and transport. slc18a1 and slc18a2 encode for vesicular monoamine transporters 1 and 2, respectively. These transporters are involved in capturing cytosolic monoamines and packaging into vesicles for release during exocytosis or action potentials. th encodes a tyrosine hydroxylase gene. th is the rate-limiting enzyme that catalyzes the reaction converting tyrosine to l-dopa, the precursor to dopamine. Three loci (slc6a2-region2, slc18a1-region4 and slc18a2-region2) were identified for more quantitative analysis on the Pyrosequencing platform. Pyrosequencing assays for slc6a2, slc18a1 and slc18a2 have been developed. Optimization and calibration of the assay currently is in progress.
Significance: We have shown changes in DNA methylation in umbilical cord blood resulting from tobacco smoke exposure at two ADHD-relevant genes in unadjusted analysis. Adjusted analyses are underway. This supports that epigenetic alterations are associated with in utero exposure to tobacco smoke. We have determined the optimal methodology for simultaneous purification of DNA and RNA and are beginning to prepare the brain and blood from the in utero exposed/unexposed rats for whole genome bisulfite sequencing and transcriptome analyses.
Community Outreach and Translation Core
A Duke student team has been working on all aspects of the project under the guidance of the PI and the two co-investigators.
Implementation of the field-test: Our team carried out a field intervention in Durham Health clinics over a 2 month period. Participants (patients and family members in the waiting rooms) reviewed the CEASE (Community Education About Smoke Exposure) brochures that were created during the previous funding period and provided feedback about their opinions and knowledge of the science content. Data were collected from 236 patients and their families, and 32 Health Care practitioners. We obtained the following results:
- Subjects with higher education scored significantly higher on knowledge-based questions compared to subjects with lower education regardless of brochure type (ANOVA, p = 0.009)
- Our brochure was more effective in conveying new information than the control brochure by a statistically significant difference (ANCOVA, p = 0.005). None of the other covariates, such as pregnancy status and race/ethnicity were statistically significant.
- Language was a significant factor in knowledge regardless of brochure type (ANCOVA, p = 0.016). Subjects using the English and French versions scored higher than subjects using the Spanish version.
- Only 1 of the 32 health care providers indicated that patients specifically asked for more information than was in the brochure.
- 78% of the health care providers indicated they are likely or very likely to share the brochure with their patients.
Implementation of the “contest”: Students completed development of the “instagram” contest, in which the public could submit photos, graphics, or original artwork depicting what they’ve learned from the CEASE brochure and the website developed the previous year. The contest was open for 2 months, and a judging panel was created from faculty (including Murphy, PI) and other Duke staff. There were seven winning entries from the public.
Design a large-scale intervention: Our student team has developed a survey to be given to the public to assess the best social media approach for delivering educational information about pregnant women and children exposure to environmental tobacco smoke. Using some of the winning Instagram images from the contest, the intervention will compare facebook, twitter, instagram, and website approaches for dissemination of educational information. The intervention has a unique design that will determine how far participants will “drill down” to acquire additional information about exposure to tobacco smoke, ADHD and epigenetics.
Future Activities:
Project 1
We have been steadily recruiting and expect to be on target to achieve the aims. We anticipate completing the first wave (of two waves) in the late summer of 2016. We then will begin to perform preliminary analyses for publications. In addition, we have secured funding through NICHD (1R01HD084487-01; MPIs: Fuemmeler & Kollins) to further study executive functioning development and risk for obesity in this same cohort. The activities of the two projects do not overlap, but will synergize to allow for more effective recruitment and retention activities.
Project 2
Levin Lab
- Publish the results of the first study comparing the neurobehavioral consequences of tobacco smoke extract vs. nicotine alone.
- Finish neurobehavioral assessment for all cohorts of attention, learning, spatial working and reference memory, non-spatial memory, anxiety, fear and anxiety for the male and female offspring from dams exposed to tobacco smoke extract modeling environmental tobacco smoke exposure during critical time windows prior to conception, during the first half of gestation and during the second half of gestation.
- Begin assessments of pharmacotherapies to attenuate the neurobehavioral dysfunction caused by tobacco smoke exposure during development.
Slotkin-Seidler Lab
- Perform brain regional dissections in an age range from birth through weaning, adolescence and adulthood of male and female rats exposed to TSE at a dose modeling environmental tobacco smoke exposure, during critical time windows prior to conception, during the first half of gestation and during the second half of gestation.
- Perform brain region specific neurochemical analyses characterizing monoaminergic and cholinergic neurotransmitter systems of in vivo tissues from TSE administration in these critical windows.
- Elucidate mechanisms underlying the TSE effects on rat neural stem cells, characterizing the diversion of phenotypic fates of stem cells toward or away from neurons or glia.
- Prepare TSE exposed samples from in vivo and in vitro studies for epigenetics.
Satterwhite Lab
- Publish both the characterization of hNSC in vitro cell proliferation and differentiation phenotypes in response to nicotine or TSE exposure (manuscript in preparation) and the DNA methylation and gene expression changes associated with nicotine or TSE exposure in biological pathways associated with ADHD, tobacco use, dopaminergic specification pathways and vitamin C metabolism (in progress).
- Begin experiments to identify mechanism of vitamin C remediation of nicotine or TSE exposure phenotypes using siRNA and CRSPR-mediated inhibition and define remediation by key anti-oxidant and methyl donors.
- Begin concordance studies to draw connections between DNA methylation changes observed in vitro and human clinical samples including whole blood and fetal tissues.
Project 3
- For Aim 1, the rat cohorts have been bred, exposed, and evaluated for neurobehavior in Project 2. The rats have been sacrificed and tissues collected. In this coming year, we will send genomic DNA from these tissues for whole genome bisulfite sequencing using pooled samples and conduct data analysis to identify targets of methylation changes due to prenatal exposure to nicotine or tobacco smoke extract.
- For Aim 2, the same rat tissues from Aim 1 will be used for pooled RNA transcriptome profiling to identify gene expression changes that occur in frontal cortex as a result of prenatal exposure to nicotine or tobacco smoke extract. Expression changes will be validated in the individual rats. These data also will be integrated with the whole genome bisulfite sequencing data from Aim 1 to identify top candidate genes (i.e., those showing a methylation change from prenatal exposure to nicotine or tobacco smoke extract and concordance in methylation change between brain and blood and that also show a correlation between methylation and expression in control versus exposed animals) for analysis in our human cohort in Aim 3.
- For Aim 3, we will continue to examine candidate targets of methylation in exposed humans and rats. This will shift to examination of candidates identified from results obtained by Aims 1 and 2 once the data analysis is completed.
- We will continue to bank and track biological specimens as they are collected from Projects 1 and 2.
- We will outsource maternal plasma specimens to quantify cotinine at the time of enrollment into the NEST study during early pregnancy and use these data to compare to self reports of smoke exposure, to the outcome measures for NICHES participants in Project 1, and to the methylation data generated for the NICHES participants.
Community Outreach and Translation Core
In the next year, we will carry out the large-scale intervention (at least 500 participants including 50 pregnant women participants) via a Qualtrics database online. The intervention will proceed until 500 participants have completed the survey. Prior to the intervention, a small beta-test with a diverse group of local participants will be conducted to identify any problems/mistakes with the wording and organization of the survey itself. Finally, data analysis after completion of the intervention will be initiated. A variety of variables will be considered in determining the role of the different social media approaches for educational dissemination.
Journal Articles: 34 Displayed | Download in RIS Format
| Other center views: | All 118 publications | 40 publications in selected types | All 34 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Breton CV, Marsit CJ, Faustman E, Nadeau K, Goodrich JM, Dolinoy DC, Herbstman J, Holland N, LaSalle JM, Schmidt R, Yousefi P, Perera F, Joubert BR, Wiemels J, Taylor M, Yang IV, Chen R, Hew KM, Hussey Freeland DM, Miller R, Murphy SK. Small-magnitude effect sizes in epigenetic end points are important in children’s environmental health studies: the Children’s Environmental Health and Disease Prevention Research Center’s Epigenetics Working Group. Environmental Health Perspectives 2017;125(4):511-526. |
R835437 (2017) |
|
|
|
Small Magnitude Effect Sizes in Epigenetic Endpoints are Important in Children's Environmental Health Studies:The Children's Environmental Health and Disease Prevention Research Center's Epigenetics Working Group. Environmental Health Perspectives 2017; 125(4):511-526. |
R835437 (2016) |
|
|
|
Cauley M, Hall BJ, Abreu-Villaca Y, Junaid S, White H, Kiany A, Slotkin TA, Levin ED. Critical developmental periods for effects of low-level tobacco smoke exposure on behavioral performance. Neurotoxicology 2018;68:81-87. |
R835437 (2017) |
Exit Exit Exit |
|
|
Fleisch AF, Kloog I, Luttmann-Gibson H, Gold DR, Oken E, and Schwartz JD. Air Pollution Exposure and Gestational Diabetes Mellitus Among Pregnant Women in Massachusetts:a Cohort Study. Environmental Health 2016; 15:40-48. |
R835437 (2016) R834798 (Final) R834798C005 (Final) |
Exit |
|
|
Fuemmeler BF, Wang L, Iversen ES, Maguire R, Murphy SK, Hoyo C. Association between prepregnancy body mass index and gestational weight gain with size, tempo, and velocity of infant growth: analysis of the Newborn Epigenetic Study cohort. Childhood Obesity 2016;12(3):210-218. |
R835437 (2015) |
Exit Exit Exit |
|
|
Fuemmeler BF, Lee CT, Soubry A, Iversen ES, Huang Z, Murtha AP, Schildkraut JP, Jirtle RL, Murphy SK, Hoyo C. DNA methylation of regulatory regions of imprinted genes at birth and its relation to infant temperament. Genetics and Epigenetics 2016;8:59-67. |
R835437 (2017) |
Exit Exit |
|
|
Fuemmeler B, Glasgow T, Schechhter J, Maguire R, Sheng Y, Bidopia T, Barsell D, Ksinan A, Zhang J, Lin Y, Hoyo C, Murphy S, Qin J, Wang X, Kollins S. Prenatal and Childhood Smoke Exposure Associations with Cognition, Language, and Attention-Deficit/Hyperactivity Disorder. JOURNAL OF PEDIATRICS 2023;256:77 |
R835437 (Final) |
Exit |
|
|
Fuemmeler B, Dahman B, Glasgow T, Barsell D, Oliver J, Zhang J, Hoyo C, Murphy S, McClernon F, Wheeler D. Tobacco Exposures are Associated With Healthcare Utilization and Healthcare Costs in Pregnant Persons and Their Newborn Babies. NICOTINE & TOBACCO RESEARCH 2024; |
R835437 (Final) |
Exit |
|
|
Gao L, Liu X, Millstein J, Siegmund KD, Dubeau L, Maguire RL, Zhang JJ, Fuemmeler BF, Kollins SH, Hoyo C, Murphy SK, Breton CV. Self-reported prenatal tobacco smoke exposure, AXL gene-body methylation, and childhood asthma phenotypes. Clinical Epigenetics 2018;10(1):98 (11 pp.). |
R835437 (2017) |
Exit Exit Exit |
|
|
Hall BJ, Cauley M, Burke D, Kiany A, Slotkin TA, Levin ED. Cognitive and behavioral impairments evoked by low-level exposure to tobacco smoke components: comparison with nicotine alone. Toxicological Sciences 2016;151(2):236-244. |
R835437 (2015) |
Exit |
|
|
Hall BJ, Abreu-Villaca Y, Cauley M, Junaid S, White H, Kiany A, Levin ED. The ventral hippocampal muscarinic cholinergic system plays a key role in sexual dimorphisms of spatial working memory in rats. Neuropharmacology 2017;117:106-113. |
R835437 (2017) |
Exit Exit |
|
|
Hoffman K, Butt C, Webster T, Preston E, Hammel S, Makey C, Lorenzo A, Cooper E, Carignan C, Meeker S, Price T, Hoyo C, Mendelsohn E, Congleton J, Daniels J, Stapleton H. Temporal Trends in Exposure to Organophosphate Flame Retardants in the United States. ENVIRONMENTAL SCIENCE & TECHNOLOGY LETTERS 2017;4(3):112-118. |
R835437 (Final) |
Exit Exit |
|
|
King KE, Kane JB, Scarbrough P, Hoyo C, Murphy SK. Neighborhood and family environment of expectant mothers may influence prenatal programming of adult cancer risk: discussion and an illustrative DNA methylation example. Biodemography and Social Biology 2016;62(1):87-104. |
R835437 (2015) |
Exit |
|
|
Lee W-C, Shen L, Catalano PJ, Mickley LJ, and Koutrakis P. Effects of Future Temperature Change on PM2.5 Infiltration in the Greater Boston Area. Atmospheric Environment 2017;150:98-105. |
R835437 (2016) R834798 (Final) R835755 (2016) |
Exit Exit Exit |
|
|
Levin ED. Learning about cognition risk with the radial-arm maze in the developmental neurotoxicology battery. Neurotoxicology and Teratology 2015;52(Pt A):88-92. |
R835437 (2014) |
Exit Exit Exit |
|
|
Murphy SK, Erginer E, Huang Z, Visco Z, Hoyo C. Genotype-epigenotype interaction at the IGF2 DMR. Genes 2015;6(3):777-789. |
R835437 (2014) |
Exit Exit Exit |
|
|
Nye MD, Fry RC, Hoyo C, Murphy SK. Investigating epigenetic effects of prenatal exposure to toxic metals in newborns: challenges and benefits. Medical Epigenetics 2014;2(1):53-59. |
R835437 (2013) R835437 (2014) |
Exit Exit Exit |
|
|
Nye MD, Hoyo C, Murphy SK. In vitro lead exposure changes DNA methylation and expression of IGF2 and PEG1/MEST. Toxicology In Vitro 2015;29(3):544-550. |
R835437 (2014) |
Exit Exit Exit |
|
|
Nye MD, King KE, Darrah TH, Maguire R, Jima DD, Huang Z, Mendez MA, Fry RC, Jirtle RL, Murphy SK, Hoyo C. Maternal blood lead concentrations, DNA methylation of MEG3 DMR regulating the DLK1/MEG3 imprinted domain and early growth in a multiethnic cohort. Environmental Epigenetics 2016;2(1):1-8. |
R835437 (2015) |
Exit Exit |
|
|
Schechter JC, Kollins SH. Prenatal smoke exposure and ADHD: advancing the field. Pediatrics 2017;139(2):e20163481 (2 pp.). |
R835437 (2017) |
Exit Exit Exit |
|
|
Schechter JC, Fuemmeler, BF, Hoyo C, Murphy SK, Zhang JJ, Kollins SH. Impact of smoking ban on passive smoke exposure in pregnant non-smokers: using cotinine as a biomarker of exposure. International Journal of Environmental Research and Public Health 2018;15(1):E83 (16 pp.). |
R835437 (2017) |
Exit Exit Exit |
|
|
Slotkin TA, Card J, Seidler FJ. Adverse benzo[a]pyrene effects on neurodifferentiation are altered by other neurotoxicant coexposures: interactions with dexamethasone, chlorpyrifos, or nicotine in PC12 cells. Environmental Health Perspectives 2013;121(7):825-831. |
R835437 (2013) R835437 (2014) |
|
|
|
Slotkin TA, Card J, Seidler FJ. Nicotine administration in adolescence reprograms the subsequent response to nicotine treatment and withdrawal in adulthood: sex-selective effects on cerebrocortical serotonergic function. Brain Research Bulletin 2014;102:1-8. |
R835437 (2014) |
Exit Exit Exit |
|
|
Slotkin TA, Card J, Stadler A, Levin ED, Seidler FJ. Effects of tobacco smoke on PC12 cell neurodifferentiation are distinct from those of nicotine or benzo[a]pyrene. Neurotoxicology and Teratology 2014;43:19-24. |
R835437 (2013) R835437 (2014) |
Exit Exit Exit |
|
|
Slotkin TA, Skavicus S, Card J, Levin ED, Seidler FJ. Amelioration strategies fail to prevent tobacco smoke effects on neurodifferentiation: nicotinic receptor blockade, antioxidants, methyl donors. Toxicology 2015;333:63-75. |
R835437 (2014) |
Exit Exit Exit |
|
|
Slotkin TA, Skavicus S, Card J, Stadler A, Levin ED, Seidler FJ. Developmental neurotoxicity of tobacco smoke directed toward cholinergic and serotonergic systems: more than just nicotine. Toxicological Sciences 2015;147(1):178-189. |
R835437 (2014) |
Exit |
|
|
Slotkin TA, Stadler A, Skavicus S, Seidler FJ. Adolescents and adults differ in the immediate and long-term impact of nicotine administration and withdrawal on cardiac norepinephrine. Brain Research Bulletin 2016;122:71-75. |
R835437 (2015) |
Exit Exit Exit |
|
|
Slotkin TA, Skavicus S, Card J, Levin ED, Seidler FJ. Diverse neurotoxicants target the differentiation of embryonic neural stem cells into neuronal and glial phenotypes. Toxicology 2016;372:42-51. |
R835437 (2016) R835437 (2017) |
Exit Exit Exit |
|
|
Slotkin TA, Stadler A, Skavicus S, Card J, Ruff J, Levin ED, Seidler FJ. Is there a critical period for the developmental neurotoxicity of low-level tobacco smoke exposure? Toxicological Sciences 2017;155(1):75-84. |
R835437 (2017) |
Exit Exit Exit |
|
|
Slotkin, T.A., Stadler, A., Skavicus, S., Card, J., Ruff, J., Levin, E.D., Seidler, F.J. 2016. Is there a critical period for the developmental neurotoxicity of low-level tobacco smoke exposure? Toxicological Sciences. DOW:10.1093. |
R835437 (2016) |
not available |
|
|
Smeester L, Yosim AE, Nye MD, Hoyo C, Murphy SK, Fry RC. Imprinted genes and the environment: links to the toxic metals arsenic, cadmium, lead and mercury. Genes 2014;5(2):477-496. |
R835437 (2014) |
Exit Exit Exit |
|
|
Soubry A, Hoyo C, Jirtle RL, Murphy SK. A paternal environmental legacy: evidence for epigenetic inheritance through the male germ line. BioEssays 2014;36(4):359-371. |
R835437 (2013) R835437 (2014) |
Exit Exit Exit |
|
|
Tindula G, Murphy SK, Grenier C, Huang Z, Huen K, Escudero-Fung M, Bradman A, Eskenazi B, Hoyo C, Holland N. DNA methylation of imprinted genes in Mexican-American newborn children with prenatal phthalate exposure. Epigenomics 2018;10(7):1011-1026. |
R835437 (2017) |
Exit Exit |
|
|
Vidal AC, Benjamin Neelon SE, Liu Y, Tuli AM, Fuemmeler BF, Hoyo C, Murtha AP, Huang Z, Schildkraut J, Overcash F, Kurtzberg J, Jirtle RL, Iversen ES, Murphy SK. Maternal stress, preterm birth, and DNA methylation at imprint regulatory sequences in humans. Genetics and Epigenetics 2014;6:37-44. |
R835437 (2014) |
Exit Exit Exit |
Supplemental Keywords:
Secondhand smoke, nicotine, tobacco smoke extract, attention deficit hyperactivity disorder, neurobehavior, cognitive function, neurotransmission, human neural stem cells, pyrosequencing, DNA methylation, epigenetics, InstagramRelevant Websites:
The Center for Study of Neurodevelopment and Improving Children’s Health following Environmental tobacco Smoke exposure (NICHES) Exit
RISE at Duke Exit
Help Babies Avoid Smoke! Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2017 Progress Report
- 2016 Progress Report
- 2015 Progress Report
- 2013 Progress Report
- Original Abstract
34 journal articles for this center