Grantee Research Project Results
2021 Progress Report: Instrumenting phenotypic immunological responses to toxicants that threaten human reproduction
EPA Grant Number: R839501Title: Instrumenting phenotypic immunological responses to toxicants that threaten human reproduction
Investigators: Osteen, Kevin G , Cliffel, David , McLean, John , Bruner-Tran, Kaylon L.
Institution: Vanderbilt University Medical Center , Vanderbilt University
EPA Project Officer: Callan, Richard
Project Period: August 1, 2019 through July 31, 2022 (Extended to July 31, 2024)
Project Period Covered by this Report: August 1, 2020 through July 31,2021
Project Amount: $848,923
RFA: Advancing Actionable Alternatives to Vertebrate Animal Testing for Chemical Safety Assessment (2018) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Our goals are to 1) examine whether a pre-existing inflammatory condition impacts the endometrium’s acute responses to an environmental toxicant known to impact fertility and 2) develop in vitro models that are appropriate alternatives to animal studies for accurate reproductive endpoint assessment.
Progress Summary:
Specific Aim 1: To utilize the EndoChip model to examine how a pre-existing inflammatory disease affects cellular response(s) to an environmental toxicant.
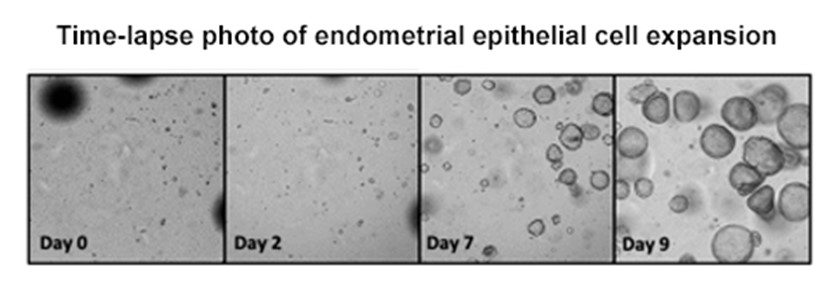
As presented in our previous progress report, we have developed organoid technology using isolated “epithelial fragments” that now allow us to routinely propagate primary epithelial cells in a matrix, maintaining the 3-D environment necessary for the function of endometrial glands during each menstrual cycle (Figure 1). Additionally, we have initiated co-cultures of epithelial organoids with endometrial stromal/decidual cells in a similar 3D matrix. This co-culture model provides an in vivo-like environment to monitor toxicant influences on the changing steroid-mediated cell-cell communication that regulates growth and differentiation during each menstrual cycle. Based on the power of our organoid/matrix technology, over the course of the current reporting period, we specifically focused on the influence that endometrial epithelial cells play in transmitting disease and/or TCDD-mediated inflammatory responses to adjacent stromal cells (as noted below, new information about somatic mutations in endometriosis-related epithelial cells), suggesting these cells are driving invasive disease. Using control epithelial cells, we found that epithelial-to-stroma communication in vitro can significantly impact the ability of progesterone to mediate stromal decidualization as monitored by expression of prolactin. Significantly, stromal cell resistance to progesterone in endometriosis patients alters their response to inflammatory cytokines of either epithelial or immune cell origin. Using organoid technology, we demonstrated that increasing the ratio of epithelial cells to stromal cells in vitro acts to inhibit stromal cell expression of prolactin in response to progesterone. Although addition of 2.5% conditioned media (CM) from epithelial cells led to the highest level of prolactin production by stroma/decidual cells, increasing the amount of epithelial CM inhibited prolactin production by stromal cells. Taken together, these results demonstrate the critical need for a balanced stromal-epithelial ratio in vitro for appropriate communication via paracrine signaling for monitoring progesterone responses within stromal-epithelial co-cultures. Going forward, as Covid-19 restrictions end, we will utilize epithelial cells acquired from endometriosis patients (including epithelial cells with disease-related somatic mutations of KRAS and ARID1) versus control donor epithelial cells. This approach will allow us to construct the appropriate EndoChip epithelial/stromal cell ratios while testing the relative impact of control versus disease-related epithelial phenotypes using prolactin (via ELISA) and matrix metalloproteinase-3 (via MMP-3 sensor) as reporter proteins for toxicant studies.
Figure 1: Endometrial epithelial cell expansion in synthetic matrix over time. (Gnecco et al unpublished)
Specific Aim 2: To Examine Disease-Toxicant Interactions in EndoChips by utilizing cell-type specific biosensors combined with metabolomic analysis via IM-MS.
As originally proposed, our primary analytical goals for the EndoChip were to determine the relative value of this organ-on-a-chip device to identify toxicant risks and potentially replace the need for in vivo animal models. Nevertheless, as noted in our grant proposal, our ongoing and published studies in TCDD exposed mice and mice bearing human tissues and immune cells from endometriosis patients (Experimental Endometriosis Model) remain important for guiding the specific design of our human cell-based in vitro EndoChip and organoid studies. More specifically, using our chimeric human/murine model of Experimental Endometriosis, we found that adoptive transfer of healthy immune cells impeded experimental disease progression while transfer of immune cells from endometriosis patients promoted disease establishment and progression. These findings demonstrate that the function of innate immune cells becomes modified by the presence of endometriosis such that disease progression is enhanced. In turn, we identified that perfusion of TCDD-exposed human immune cells (from control donors) within the EndoChip also acted to “induce” the endometriosis phenotype of reduced progesterone responsiveness related to decidualization. Thus, going forward, we are examining TCDD/Disease alterations in immune cells based on the value of our biosensor technology. We are specifically focusing on innate immune cell “training” responses (a new concept in immunology) that have not yet been examined relative to either endometriosis or toxicant exposure. Immune cell over-training can occur in response to common life-stage infections and subsequently drive inflammatory diseases. In our studies, we found that immune cells acquired from women with endometriosis, compared to control cells, exhibited increased expression of IL-1 and IL-6 in response to either TCDD or the infection mimic lipopolysaccharide (LPS). In turn exposure of endometrial stromal cells to these “activated” immune cell cytokines reduced progesterone sensitivity and elevated MMP-3 secretion while decreasing prolactin secretion. Thus, both epithelial cells and immune cells can act to disrupt endometrial progesterone responses when exposed to TCDD, a response that is amplified by both the presence of disease and an infection mimic. Additionally, over the current year of funding, we have made substantial progress in the development of hardware need for our studies. Specifically:
Electrochemical Antibody Sensors for MMP-3:
The design of the sensor specific for matrix metalloproteinase-3 (MMP-3) aims to construct an electrochemical, magnetic bead-based competitive assay in a microfluidic device. In the microfluidic system, magnetic beads linked to the MMP-3 antibody are flowed through and held on an electrode via a magnet. Then, a solution containing MMP-3 protein conjugated to Alkaline Phosphatase (AP) is flowed through the system and the AP-conjugated protein binds to the antibodies on the beads. Finally, p-aminophenyl phosphate (PAPP) is flown through the system. PAPP is converted into p-aminophenol (PAP) by the alkaline phosphatase that is conjugated to the MMP-3 protein. The PAP is then oxidized by the electrode into p-quinone (PQI), which generates a measurable signal. In our current testing, a calibration curve with PAP was developed to ensure a linear current output in the area of interest. Preliminary negative control experiments indicate that the bead/MMP3-AP conjugate is not rinsing out of the microfluidic as expected, and we continue to work on the washing protocol to improve the background signals for these controls.
Electrochemical Wired Electrode Sensors:
Electrochemical sensors that utilize enzymes are a sensitive, inexpensive means of detecting biologically relevant analytes. These sensors are categorized based on their construction and method of signal transport. Type I sensors consist of a crosslinked enzyme on an electrode surface, and are potentially subject to interference from byproducts and other biological analytes. However, type II sensors help alleviate this problem with the addition of a redox polymer layer that assists in signal transduction, thus minimizing interferences. An osmium-loaded poly(vinylimidazole) polymer (Os-PVI) is commonly used with successful results, and when combined with an enzyme yields a type II sensor. Our initial attempts at the synthesis of this polymer resulted in an unexpected osmium precursor, which had fluorescent and redox properties that did not match with the desired Os-PVI polymer. Careful exclusion of oxygen during the Os complex precursor synthesis was necessary to avoid this unexpected oxygen containing Os-precursor, which had been seen previously in mass spectrometry studies. All precursors and osmium polymers were characterized with 1H NMR, fluorescence, mass spectrometry, and cyclic voltammetry in order to provide a better understanding of these compounds and assist in the building of new sensors.
We are optimistic that these sensors will be ready to use once we are again able to obtain human endometrial samples.
Specific Aim 3: To perform Case Studies to assess the EndoChip as an alternative to vertebrate animal testing to identify reproductive toxicants.
As noted above, the Covid-19 pandemic continues to hamper our ability to obtain human endometrial biopsies from our clinics. Therefore, we have focused on isolating and expanding primary endometrial stromal and epithelial cells from a limited number of endometriosis patients as well as our control tissue donors. Epithelial cells have been expanded in vitro within organoids and are now available for comparison studies with an endometriosis epithelial cell line (12z). We have previously frozen stocks of endometrial stromal cells which are easier to expand, and freeze compared to epithelial cells. Additionally, as a component of a separately funded NIH endometriosis grant, we and our colleagues have identified specific cancer-driver somatic mutations within epithelial cells of women with deep infiltrating endometriosis (DIE). The presence of these mutations in vivo has been shown to be associated with both a loss of progesterone responsiveness (in adjacent stromal cells) as well as increased invasiveness. Interestingly, the most common mutations identified to date are KRAS and ARID1. Previous murine studies from our group revealed that developmental TCDD exposure of mice was associated with hypomethylation of Kras and Arid1 within the placenta at sexual maturity. Interestingly, these placentae also exhibited reduced expression of the progesterone receptor (Ding et al, 2018). Since numerous epidemiology studies suggest a role of environmental exposures in the development of endometriosis; taken together with our epithelial data discussed above, it seems plausible that both naturally occurring and TCDD-mediated alterations in Kras/Arid1A gene expression in epithelial cells may contribute to development of reduced progesterone sensitivity in stromal/decidual cells associated with the endometriosis phenotype. Equally important, since the epigenetic loss of stromal cell sensitivity to progesterone is a primary cellular phenotype of endometriosis, we examined the effect of TCDD on stromal cells expression of the DNA methyltransferase (DNMT) family, enzymes that have a central role in epigenetic gene regulation of progesterone receptors in these cells. Expression of the canonical DNMT enzymes - DNMT1, DNMT3A and DNMT3B have all been found to be disrupted by TCDD exposure. We found that that TCDD enhanced expression of DNMT1 in exposed stromal cells but had only a modest effect of DNMT3a and DNMT3b. Interestingly, co-treatment of stromal cells with both IL-1 and TCDD led to a dramatic decrease in expression of all three genes, which directly corelated with a significant loss of progesterone receptor expression. To further clarify the results of our epithelial and stromal cross-communication, we established organoid cultures containing both endometrial stromal and epithelial cells. Supporting the role of TCDD acting via IL-1, addition of this inflammatory cytokine resulted in a loss of both progesterone receptor and a reduced production of prolactin expression. Together, these findings will be incorporated in the EndoChip and organoid approaches in the remaining grant period.
Future Activities:
Over the next year, dependent upon increased availability of patient samples, we anticipate using primary cells within our organoid models. Even without additional samples, we will continue to advance our studies using previously obtained cells and/or cell lines. We will continue to examine these cultures in the presence and absence of an inflammatory challenge. The next step will be to introduce immune cells into the cultures. We will measure established biomarkers of P4-mediated stromal-epithelial communication as well MMP3. Together these studies will allow us to further validate the EndoChip as a screening tool for identifying disease-toxicant interactions.
We also anticipate that the biosensors being developed in the Cliffel lab will be fully online and in use. We also anticipate performing IM-MS on conditioned media from a variety of EndoChips (primary vs cell lines) and with and without toxicant exposure.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 19 publications | 3 publications in selected types | All 3 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Stephens VR, Rumph JT, Ameli S, Bruner-Tran KL, Osteen KG. The potential relationship between environmental endocrine disruptor exposure and the development of endometriosis and adenomyosis. Frontiers in Physiology 2022;12:807685. |
R839501 (2021) |
Exit |
|
|
Rumph JT, Stephens VR, Martin JL, Brown LK, Thomas PL, Cooley A, Osteen KG, Bruner-Tran KL. Uncovering Evidence:Associations between Environmental Contaminants and Disparities in Women’s Health. International Journal of Environmental Research and Public Health 2022;19(3):1257. |
R839501 (2021) |
Exit |
|
|
Rumph JT, Stephens VR, Archibong AE, Osteen KG, Bruner-Tran KL. Environmental endocrine disruptors and endometriosis. Animal Models for Endometriosis:Evolution, Utility and Clinical Relevance 2020:57-78. |
R839501 (2021) |
|
Supplemental Keywords:
Biology, environment, contaminant, toxicsRelevant Websites:
Department of Obstetrics and Gynecology Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.