Grantee Research Project Results
2013 Progress Report: Imaging Assessment of G-protein-coupled Estrogen Receptor Activation
EPA Grant Number: R835169Title: Imaging Assessment of G-protein-coupled Estrogen Receptor Activation
Investigators: Volz, David C.
Institution: University of South Carolina at Columbia
EPA Project Officer: Aja, Hayley
Project Period: June 1, 2012 through May 31, 2015
Project Period Covered by this Report: April 23, 2013 through April 22,2014
Project Amount: $1,063,460
RFA: Developing High-Throughput Assays for Predictive Modeling of Reproductive and Developmental Toxicity Modulated Through the Endocrine System or Pertinent Pathways in Humans and Species Relevant to Ecological Risk Assessment (2011) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
The overall objective of this study is to develop high-content screening (HCS) assays to rapidly identify chemicals that exhibit aberrant G-protein-coupled estrogen receptor (GPER)-mediated developmental toxicity. Using zebrafish as a model for vertebrate embryogenesis, published findings from our laboratory show that (1) GPER is expressed as early as 1 hpf and localized to the brain and heart during embryogenesis; (2) continuous exposure to a selective GPER agonist (G-1) – but not a structurally similar selective GPER antagonist (G-15) – results in gross abnormalities and disruption of axial muscles; and (3) G-1-induced effects are blocked by co-exposure to G-15, suggesting that aberrant GPER activation alone is responsible for G-1-induced developmental toxicity. Therefore, our working hypothesis is that xenobiotic-induced activation of GPER results in targeted effects on the cardiovascular and/or nervous system, leading to indirect adverse effects on muscle development within zebrafish larvae. We will test our central hypothesis and accomplish the overall objective of this application by pursuing the following two research objectives:
- Develop HCS assays to assess the potential impacts of reference GPER agonists on cardiovascular or nervous system development during zebrafish embryogenesis.
- Screen EPA’s teratogenic ToxCast Phase-I chemical library to identify and classify chemicals that mediate developmental toxicity via a GPER-dependent adverse outcome pathway (AOP).
Progress Summary:
During Year 1 of this project, we accomplished Research Objective 1 by developing and optimizing two HCS assays that rapidly identify chemicals impacting cardiovascular or nervous system function in the absence of effects on survival and growth. Abstracts summarizing each these assays are below.
High-Content Screening Assay for Identification of Chemicals Impacting Cardiovascular Function
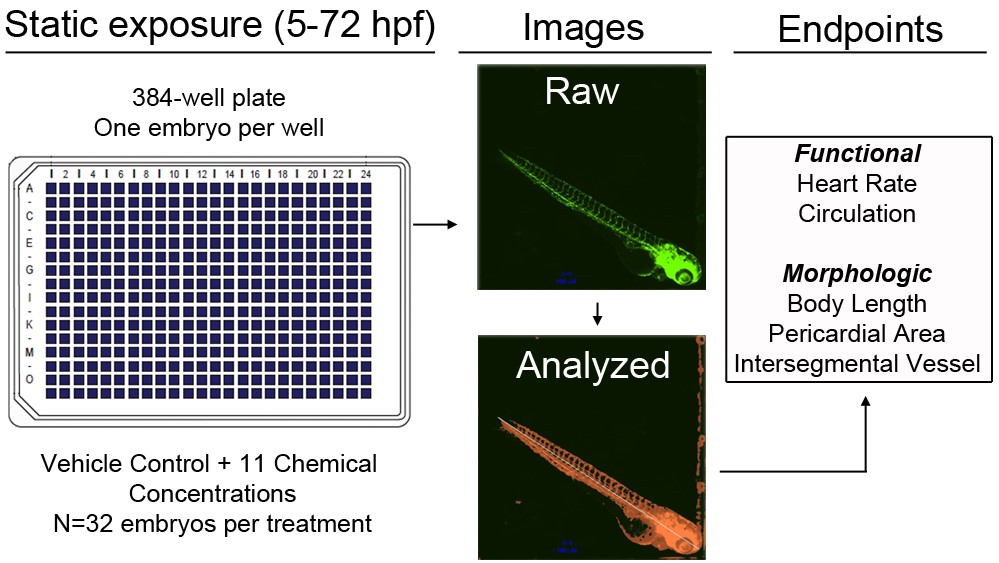
Targeted assays are needed to better evaluate effects of chemicals on organogenesis and begin classification of chemicals by toxicologically relevant modes-of-action. Using transgenic zebrafish (fli1:egfp) that stably express eGFP within vascular endothelial cells, we have developed and optimized a 384-well-based HCS assay that enables us to screen and identify chemicals affecting cardiovascular function at sub-lethal, non-teratogenic concentrations (Figure 1). Following static exposure of one embryo per well from 5-72 hours post-fertilization (hpf), automated image acquisition procedures and custom image analysis protocols are used to quantify body length, circulation, heart rate, pericardial area (a biomarker for cardiac looping defects), and intersegmental vessel area within freshly hatched live embryos. After optimizing 72-hpf anesthetization procedures, we evaluated each endpoint across four independent control plates containing 384 initial embryos per plate. Survival and imaging success rates across these plates ranged from 93-99% and 42-74%, respectively. Criteria then were defined for assay success and analysis of treatments, and 10 chemicals were screened for targeted effects on cardiovascular function. Compared to existing zebrafish-based assays, this method provides a comprehensive discovery platform with 1) increased sample sizes; 2) broad concentration-response format; and 3) the ability to identify chemicals that target cardiovascular function at non-teratogenic concentrations.
Figure 1. Overview of HSC assay for identification of chemicals impacting cardiovascular function.
High-Content Screening Assay for Identification of Chemicals Impacting Spontaneous Activity
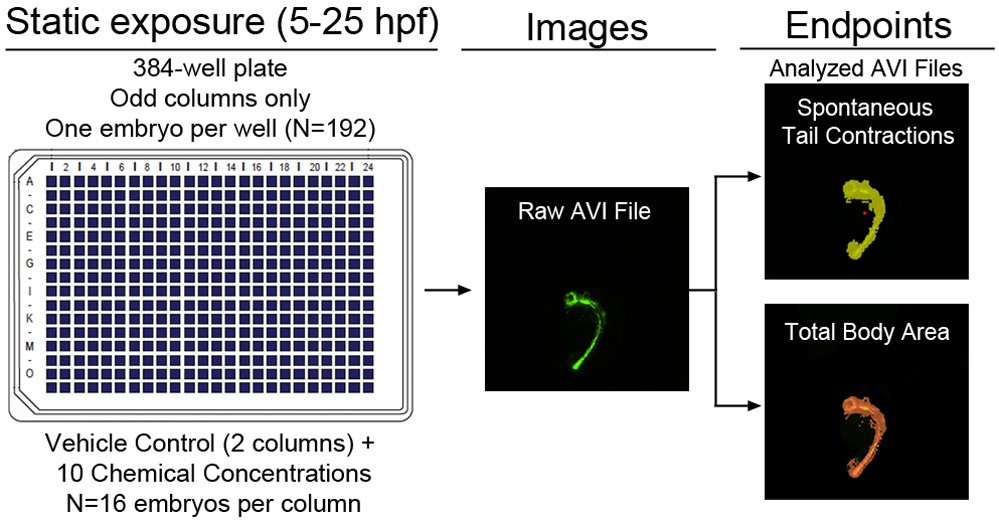
Although cell-based assays exist, rapid and cost-efficient HCS assays within intact organisms are needed to support prioritization for developmental neurotoxicity (DNT) testing in rodents. During zebrafish embryogenesis, spontaneous tail contractions occur from late-segmentation (~19 hours post-fertilization, hpf) through early-pharyngula (~29 hpf) and represent the first sign of locomotion. Using transgenic zebrafish (fli1:egfp) that stably express eGFP beginning at ~14 hpf, we have developed and optimized a 384-well-based HCS assay that quantifies spontaneous activity within single zebrafish embryos after exposure to test chemicals in concentration-response format (Figure 2). Following static exposure of one embryo per well from 5-25 hpf, automated image acquisition procedures and custom analysis protocols were used to quantify total body area and spontaneous activity in live embryos. Survival and imaging success rates across control plates ranged from 87.5-100% and 93.3-100%, respectively. Using our optimized procedures, we screened 16 chemicals within the EPA’s ToxCast Phase-I library, and found that exposure to abamectin and emamectin benzoate – both potent avermectins – abolished spontaneous activity in the absence of gross malformations. Overall, compared to existing locomotion-based zebrafish assays conducted later in development, this method provides a simpler discovery platform for identifying potential developmental neurotoxicants.
Figure 2. Overview of HSC assay for identification of chemicals impacting spontaneous activity.
During Year 2 of this project, we accomplished Research Objective 2 by (1) screening the most acutely toxic chemicals within EPA’s ToxCast Phase-I library in concentration-response format using our HSC assay for cardiovascular toxicity and (2) identifying chemicals that may mediate cardiovascular toxicity via a GPER-dependent AOP. Abstracts summarizing each of these projects are below.
High-Content Screening in Zebrafish Embryos Identifies Butafenacil as a Potent Inducer of Anemia
After ranking developmental toxicity data generated from the EPA’s zebrafish teratogenesis assay, we screened 26 of the most acutely toxic chemicals within EPA’s ToxCast Phase-I library in concentration-response format (0.05-50 μM) using our HCS assay for cardiovascular toxicity. Based on this screen, we identified butafenacil as a potent inducer of anemia, as exposure from 0.39 to 3.125 μM butafenacil completely abolished arterial circulation in the absence of effects on all other endpoints evaluated (Figure 3). Butafenacil is a herbicide that inhibits protoporphyrinogen oxidase (PPO) – an enzyme necessary for heme production in vertebrates. Using o-dianisidine staining, we then revealed that severe butafenacil-induced anemia in zebrafish was due to a complete loss of hemoglobin following exposure during early development. Therefore, six additional PPO inhibitors within the ToxCast Phase-I library were screened to determine whether anemia represents a common adverse outcome for these herbicides. Embryonic exposure to only one of these PPO inhibitors – flumioxazin – resulted in a similar phenotype as butafenacil, albeit not as severe as butafenacil. Overall, this study highlights the potential utility of this assay for (1) screening chemicals for cardiovascular toxicity and (2) prioritizing chemicals for future hypothesis-driven and mechanism-focused investigations within zebrafish and mammalian models.
Figure 3. Butafenacil abolished circulation in the absence of effects on all other endpoints evaluated.
Identification of Chemicals Mediating GPER-dependent Cardiovascular Toxicity
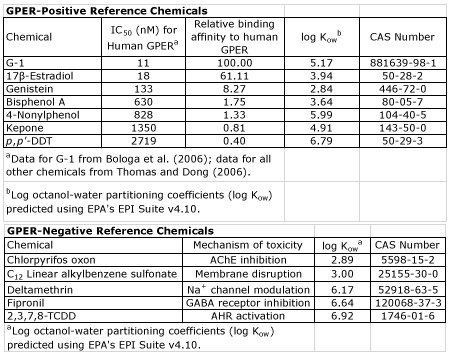
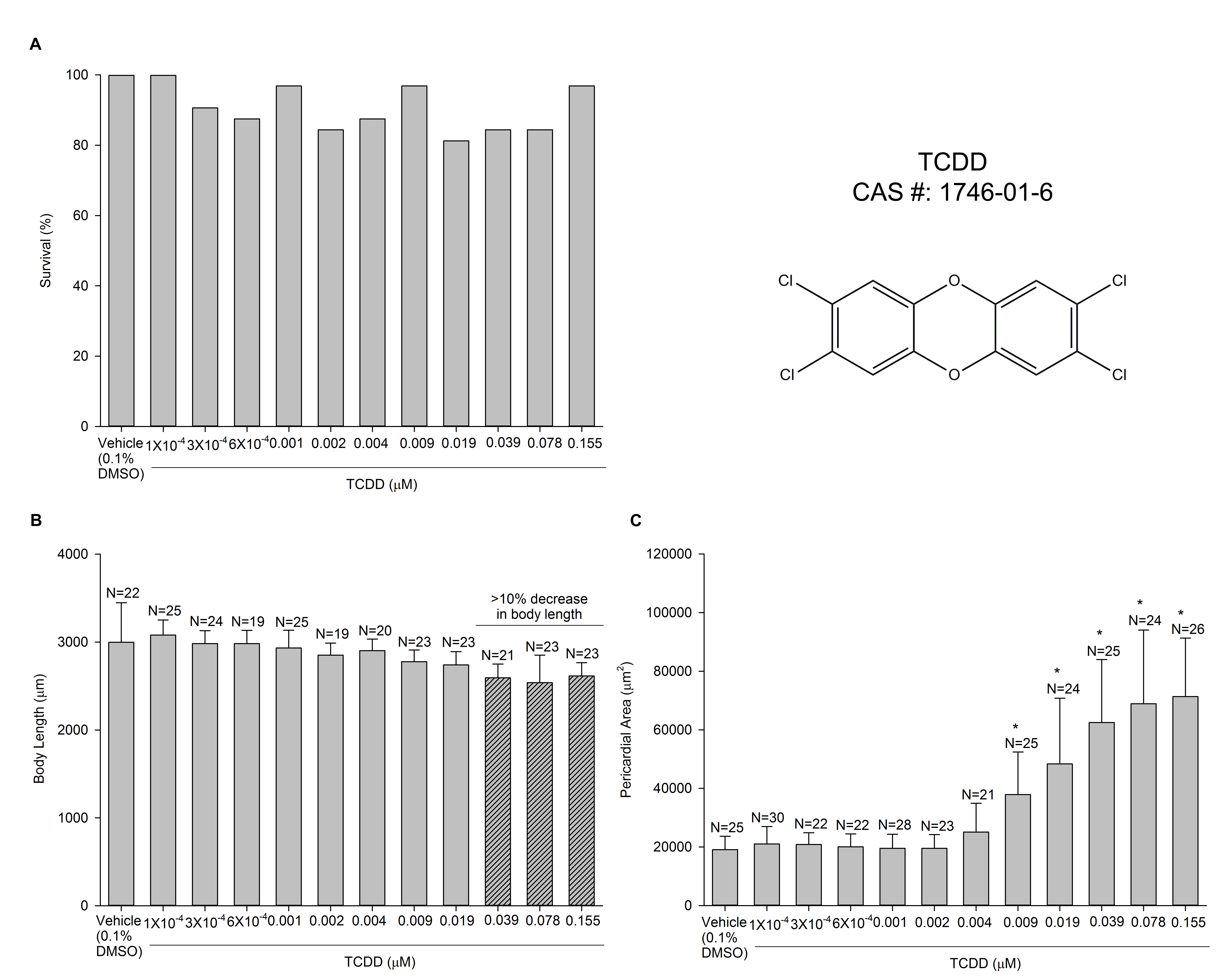
We relied on a 12-chemical training set that consisted of seven reference GPER agonists with increasing potency to human GPER (GPER-positive chemicals), as well as five reference chemicals that represent varying chemical classes and, to our knowledge, do not bind GPER nor initiate GPER-mediated AOPs (GPER-negative chemicals) (Table 1). All reference chemicals exhibited sufficient hydrophobicity (log Kow > 2) to ensure adequate chemical movement across the chorion during embryogenesis. The selective GPER antagonist (G-15) also was used to test whether cardiovascular toxicity was mitigated (or exacerbated) in the presence of G-15; the predicted log Kow and IC50 (for human GPER) for G-15 are 5.49 and 20 nM, respectively. With the exception of G-1 (a selective GPER agonist) and TCDD (a potent AHR agonist and well known cardiotoxicant) (Figure 4), none of the GPER-positive and GPER-negative chemicals that we screened impacted cardiovascular development and function in the absence of effects on survival and body length. Despite these negative findings, we proceeded to test whether TCDD-induced cardiotoxicity was partially mediated via activation of GPER, as evidence in the literature suggests that TCDD-induced toxicity may, in part, be mediated by activation of non-genomic signaling. Unfortunately, by the time we were ready to conduct this follow-up study, our existing G-15 stocks were expired and G-15 is no longer sold in the United States by our original vendor. Therefore, we purchased new G-15 from an alternative vendor and, unlike G-15 obtained from our original vendor, found that G-15 from this alternative vendor was insoluble in 1X embryo media (EM) – a reverse osmosis (RO) water-based media we routinely use for rearing zebrafish embryos. However, since G-15 was soluble in RO water alone (rather than 1X EM), we reared zebrafish embryos in RO water to address this unexpected issue and, as a positive control, exposed embryos to G-1 in the presence or absence of G-15. While rearing control embryos in RO water alone resulted in no adverse effects on embryonic development (compared to rearing in 1X EM), G-15 was unable to block G-1-induced toxicity, suggesting that G-15 from this alternative vendor lacked GPER antagonist activity. As a result, we discontinued this project, as we no longer have access to a reliable source of G-15.
Table 1. GPER-positive and GPER-negative reference chemicals selected for screening.
Figure 4. TCDD affects cardiac development in the absence of effects on survival and body length.
Future Activities:
For the final year (Year 3) of this project, we will focus on hypothesis-driven, follow-up studies that uncover mechanisms of toxicity for novel ‘hits’ – specifically, abamectin and butafenacil – identified from our screens during Years 1 and 2. To better understand how abamectin induces developmental neurotoxicity, we will (1) identify developmental stages susceptible to abamectin exposure within the first 24 h of zebrafish embryogenesis; (2) determine whether abamectin-induced effects on spontaneous activity are reversible; and (3) determine whether abamectin-induced hypoactivity is mediated by activation of the γ-aminobutyric acid (GABA) receptor. To better understand how butafenacil induces anemia during cardiovascular development, we will (1) identify developmental stages susceptible to butafenacil exposure within the first 72 h of zebrafish embryogenesis; (2) determine whether butafenacil-induced anemia is reversible; and (3) determine whether butafenacil-induced anemia is mediated by inhibition of protoporphyrinogen oxidase (PPO).
During Year 3, we will continue to publish our research within high-impact journals and present our findings at high-profile national meetings. We anticipate that two more journal articles will be published in Year 3 (one focused on abamectin and one focused on butafenacil), resulting in a total of six journal articles published during the 3-year project period. In addition, we will be presenting at the 35th Annual Conference of the Society of Environmental Toxicology and Chemistry–North America in November 2014 (two platforms and one poster), 7th Aquatic Animal Models of Human Disease Conference in December 2014 (one platform and two posters), and 54th Annual Meeting of the Society of Toxicology in March 2015 (one platform and two posters), resulting in a total of 21 presentations at national meetings during the 3-year project period.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 28 publications | 8 publications in selected types | All 8 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Leet JK, Lindberg CD, Bassett LA, Isales GM, Yozzo KL, Raftery TD, Volz DC. High-content screening in zebrafish embryos identifies butafenacil as a potent inducer of anemia. PLoS ONE 2014;9(8):e104190, 10 pp. |
R835169 (2013) R835169 (Final) |
Exit Exit Exit |
|
|
Raftery TD, Isales GM, Yozzo KL, Volz DC. High-content screening assay for identification of chemicals impacting spontaneous activity in zebrafish embryos. Environmental Science & Technology 2014;48(1):804-810. |
R835169 (2012) R835169 (2013) R835169 (Final) |
Exit Exit Exit |
Supplemental Keywords:
zebrafish, high-content screening assay, cardiovascular toxicity, developmental neurotoxicity, G-protein-coupled estrogen receptor, EPA's ToxCastRelevant Websites:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.