Grantee Research Project Results
2014 Progress Report: A Multi-scale Dose-response Model of AHR Toxicity Pathway Activation in the Human Liver
EPA Grant Number: R835000Title: A Multi-scale Dose-response Model of AHR Toxicity Pathway Activation in the Human Liver
Investigators: Bhattacharya, Sudin , Andersen, Melvin E. , Clewell, Harvey
Current Investigators: Bhattacharya, Sudin
Institution: The Hamner Institutes
EPA Project Officer: Chung, Serena
Project Period: June 1, 2011 through May 30, 2015 (Extended to April 30, 2016)
Project Period Covered by this Report: June 1, 2014 through May 31,2015
Project Amount: $750,000
RFA: Computational Toxicology: Biologically-Based Multi-Scale Modeling (2010) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Progress Summary:
Project 1: Development of a multi-scale liver model of AhR pathway activation:
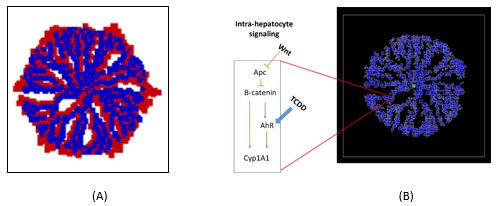
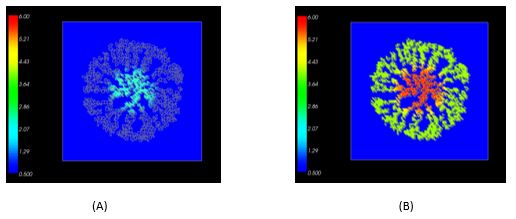
In the past year, we have developed a “virtual tissue” model of a liver lobule. This model was implemented with a modified cluster aggregation algorithm in Python where particles representing endothelial cells underwent random diffusion from the edge of the lobule towards the central vein. The diffusing network of endothelial cells created the sinusoidal vasculature permeating the lobule, which was then used as a framework to add alternating layers of hepatocytes to the lobule (Figure 1A). This virtual liver model was then incorporated into the multi-scale, multicellular modeling environment CompuCell3D (http://www.compucell3d.org/). An intracellular signaling model of Wnt/Beta-catenin and TCDD-induced cytochrome P-450 (CYP) induction was developed in JARNAC (http://sbw.kgi.edu/software/jarnac.htm) and embedded in each hepatocyte in the CompuCell3D lobule model (Figure 1B). This multi-scale model was then simulated for various doses of TCDD, exhibiting progression of CYP induction from centrilobular areas at low doses (Figure 2A) to pan-lobular induction at high doses (Figure 2B).
Figure 1. (A) “Virtual” liver lobule created with a cluster aggregation algorithm. (B) Multi-scale liver lobule model implemented in CompuCell3D.
Figure 2. Progression of CYP induction from centrilobular at low doses of TCDD (A) to pan-lobular at high doses of TCDD (B).
Subsequent steps in this project include extending the model to simulate cell proliferation at higher doses of TCDD and linking the CompuCell3D model to a PBPK model to estimate hepatic TCDD dose.
Project 2: Assembling and analyzing the AhR regulatory network in hepatocytes from existing genomic data sets:
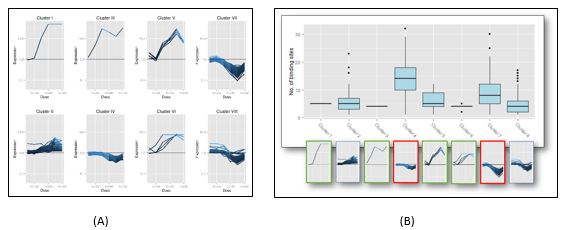
We have assembled an in vivo AhR regulatory network in mouse liver from gene expression and AhR ChIP-chip data published by the Zacharewski lab at Michigan State University. Corresponding ChIP data for genome-wide AhR binding is not currently available for human and rat, either in vivo or in vitro. A grant proposal has been submitted to the Dow Chemical Company requesting funding for ChIP-seq studies to be carried out in human and rat primary hepatocytes. We have also analyzed TCDD-induced gene expression data from primary hepatocytes (human, mouse and rat) published by the Zacharewski lab using k-means clustering (Figure 3A), and examined the promoter regions of genes in different clusters for differential patterns of dioxin response element (DRE) distribution. Preliminary analyses of human primary hepatocyte data indicates that in general downregulated genes have more DREs in their promoter regions than upregulated genes (Figure 3B).
Figure 3. (A) Dose-response of genes activated by TCDD in primary human hepatocytes clustered by k-means. (B) Analysis of number of DREs in the promoter regions of genes in each cluster shows that in general downregulated genes have more DREs than upregulated genes.
In subsequent steps, we will further analyze the differentially expressed genes for direct (those that have DREs in their promoter regions) and indirect (those that do not have DREs) AhR targets, and look for differential patterns of localization of the DREs among the direct targets in different clusters. We will also investigate the enriched Gene Ontology (GO) categories in different gene clusters with the aid of GO visualization tools developed at The Hamner.
Journal Articles:
No journal articles submitted with this report: View all 11 publications for this projectSupplemental Keywords:
Computational model, hepatotoxic injury, dose-response, PPARα, 2,3,7,8-tetrachlorodibenzo-p-dioxin, TCDD, aryl hydrocarbon receptor, AhRProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.