Grantee Research Project Results
2013 Progress Report: Assessment of Allergic Responses to Food Proteins Using a Novel and Sensitive Adjuvant-Free Ingestion-Based Mouse Model
EPA Grant Number: R834825Title: Assessment of Allergic Responses to Food Proteins Using a Novel and Sensitive Adjuvant-Free Ingestion-Based Mouse Model
Investigators: Oettgen, Hans C
Institution: Children’s Hospital, Boston
EPA Project Officer: Aja, Hayley
Project Period: September 15, 2010 through September 14, 2012 (Extended to September 14, 2013)
Project Period Covered by this Report: September 15, 2012 through September 14,2013
Project Amount: $424,803
RFA: Approaches to Assessing Potential Food Allergy from Genetically Engineered Plants (2009) RFA Text | Recipients Lists
Research Category: Human Health
Objective:
The overall goal of this project is to test the utility of a novel adjuvant-free mouse model of food allergy as a biological probe for food protein allergenicity. We proposed to test whether a novel strain of mice, F709, that exhibits an intense allergic phenotype can serve as sensor and predictor of allergenicity of foods. This was determined by feeding foods known to be of high and low allergenicity to the mice and assessing whether any aspects of the response of F709 mice to these foods correlate with their known allergenicity.
Progress Summary:
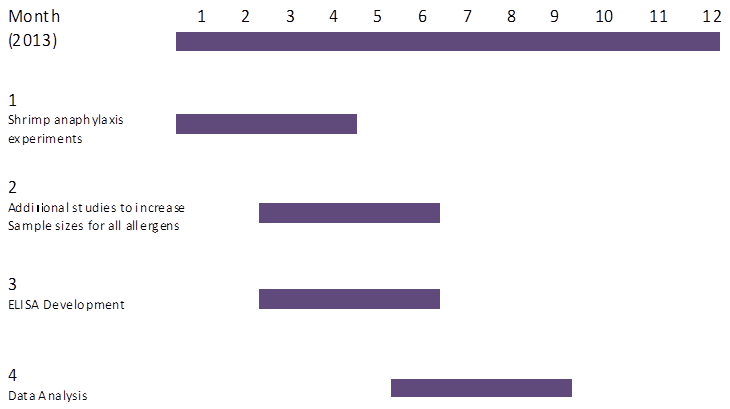
(1) Work Status: Since our last Progress Report, we have worked to determine the most effective and consistent protocol for evaluating the allergenicity of food proteins in F709 mice, and we have gathered data for peanut, egg white, rice, and chicken meat proteins using the decided-upon sensitization and challenge protocol. Chicken meat was substituted for corn as a source of food protein normally not allergenic in humans, after it was determined that the standard mouse chow contained significant amounts of corn. Cholera toxin was used as an enteral adjuvant, since it was determined that this helped to provide clearer and more consistent results. Consistent with our hypothesis that F709 mice could serve as food allergy sensors, peanut- and egg white-sensitized mice exhibited symptoms of anaphylaxis upon food challenge, while chicken meat- and rice-sensitized mice did not, although these differences are marginal and need to be verified by repetition with more mice. While serum mast cell protease (mMCP-1) levels largely matched the anaphylactic response, chicken meat-sensitized mice unexpectedly released significantly more mMCP-1 into the serum upon challenge than did their mock-sensitized counterparts. We plan to explore and verify this result, increase sample sizes for the other foods, and complete experiments using shrimp as a protein source in the coming months.
(2) Results to date: We have previously shown that F709 mice, which harbor an activating mutation of the IL-4R chain, have an intrinsically allergic phenotype with very high IgE levels and strong specific IgE responses to both inhaled and ingested antigens (Mathias et al., 2011 Tachdjian et al., 2010). In addition to generating robust IgE responses and Th2 cellular reactions to ingested proteins, the animals exhibit intense anaphylaxis upon ingestion challenge, a unique phenotype in mouse models of food allergy and one that recapitulates the type of reaction that is most concerning with food allergens. We hypothesized that such animals could be used to discriminate between allergenic and non-allergenic foods in a natural setting of purely enteral immune sensitization and challenge.
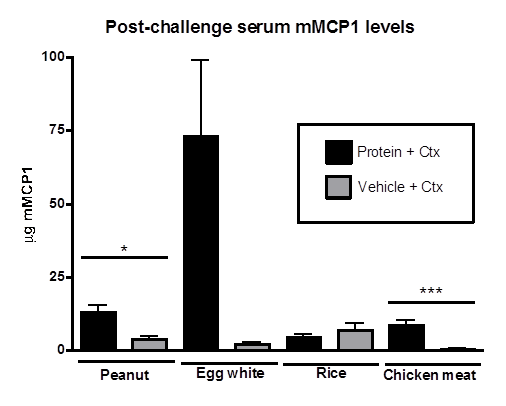
Since our last report, we have tested variables in the sensitization protocol in order to determine the most effective amount of protein for sensitization doses, amount and type of adjuvant for each sensitization dose, and the optimal number of weekly sensitization doses. The most consistent protocol was determined to be the following: F709 mice were sensitized once per week for 5 weeks by oral gavage with 2.5 milligrams food protein and 10 micrograms cholera toxin as adjuvant. Seven days after the last sensitization dose, all mice were challenged with 150 milligrams of the relevant food protein. Temperatures were monitored for 60 minutes using subdermal microchips, and levels of mMCP-1 and protein-specific IgE were measured by ELISA in serum collected 60 minutes after challenge (Figure 1). A decrease in core body temperature and a release of mMCP-1 into the serum are evidence of mast cell degranulation. Mast cell degranulation results in release of mediators of allergic reactions, including anaphylaxis, and individuals undergoing food anaphylaxis exhibit increased levels of the mast cell mediator, tryptase, consistent with degranulation. The first commonly human-allergenic protein, peanut, elicited an allergic response in F709 mice, as evidenced by significantly higher levels of serum mMCP-1 and peanut-specific IgE in peanut-sensitized mice compared to mock-sensitized controls. The core body temperature decrease was modest, however. Sensitization and challenge with egg-white elicited similar mMCP-1 and specific IgE responses, but with an exaggerated temperature drop. This experiment must be repeated in order to increase the sample size and verify these results. As expected, the relatively non-allergenic proteins, rice and chicken meat, resulted in no significant drop in temperature in experimental mice, and very little rice-specific IgE (results for chicken-specific IgE are pending). Unexpectedly, mice sensitized and challenged with chicken protein released significantly more mMCP-1 into the sera compared to their mock-sensitized counterparts, although these levels are in general lower than those usually observed in mice that have just experienced an anaphylactic reaction. While we have yet to examine shrimp as a protein source under this specific protocol, we have acquired promising results in an experiment very similar to those outlined above, but without using adjuvant. Y709F mice received 5 milligrams of shrimp protein without adjuvant, or vehicle alone, by oral gavage once per week for 9 weeks. One week after the last sensitization dose, all mice were challenged with 150 mg shrimp protein. Upon challenge, mice sensitized with shrimp protein experienced a modest drop in core body temperature compared to mock-sensitized mice (though this difference was not significantly different), and released significantly more mMCP-1 into the sera, as measured by ELISA (data not shown).
In interim experiments, we have also further delineated the biology of mast cell expansion in this model, showing that IL-4 has direct growth promoting effects on cultured mast cells as well as on intestinal mast cells in the setting of food allergy in vivo. These findings were accepted for publication in Mucosal Immunology (with cited EPA support). These results represent an important advance in our understanding of mechanisms of food allergy, establishing that enhanced IL-4 signaling (which is observed in allergic diseases) directly drives expansion of mast cells, the effectors of food anaphylaxis.

Figure 1. Anaphylaxis and mast cell activation in food protein-sensitized F709 mice. A. Once weekly for 5 weeks mice received 2.5 mg food protein + 10 mg cholera toxin in 200 ml 0.1M Bicarbonate buffer pH 8.3, or 10 mg cholera toxin alone in vehicle, by oral gavage. 7 days after last sensitizing dose, all mice were challenged orally with 150 mg protein, and temperatures were monitored using implanted transponders for 60 minutes. B. Blood samples were collected from each mouse 60 minutes after food protein challenge. Serum mMCP-1 and specific IgE (not shown) were measured by ELISA.
Future Activities:
Future activity (see timeline below): Moving forward, we plan to carry out the above protocol using shrimp as the source of food protein. We also plan to repeat sensitization and challenge experiments for several of the food proteins in order to increase sample size and clarify and verify results. While we have developed successful ELISA protocols for peanut-, egg white-, rice-, and shrimp-specific IgE, we have yet to design a comparable protocol to measure chicken meat-specific IgE. This is one of our goals in the coming months. After determining the ability of shrimp to elicit an allergic response in these mice using the described protocol, we will consider testing an additional allergenic food, such as tree nuts, and/or an additional non-allergenic food, in order to verify these results. This work has potential practical application in the identification of potential allergic risks of new proteins prior to introduction into human diets and could support EPA's mission of developing strategies for monitoring risks of genetically modified foods. As our budget is almost fully expended, we do not anticipate requesting a second NCE.
Timeline
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 2 publications | 2 publications in selected types | All 2 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Burton OT, Darling AR, Zhou JS, Noval-Rivas M, Jones TG, Gurish MF, Chatila TA, Oettgen HC. Direct effects of IL-4 on mast cells drive their intestinal expansion and increase susceptibility to anaphylaxis in a murine model of food allergy. Mucosal Immunology 2013;6(4):740-750. |
R834825 (2012) R834825 (2013) R834825 (Final) |
Exit Exit |
Supplemental Keywords:
Food allergy, allergen, IgE, anaphylaxis, IL-4;Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.