Grantee Research Project Results
2010 Progress Report: Integrated Design, Modeling, and Monitoring of Geologic Sequestration of Anthropogenic Carbon Dioxide to Safeguard Sources of Drinking Water
EPA Grant Number: R834386Title: Integrated Design, Modeling, and Monitoring of Geologic Sequestration of Anthropogenic Carbon Dioxide to Safeguard Sources of Drinking Water
Investigators: McPherson, Brian J. , Solomon, Douglas Kip , Deo, Milind D. , Goel, Ramesh
Current Investigators: McPherson, Brian J. , Deo, Milind D. , Solomon, Douglas Kip , Goel, Ramesh
Institution: University of Utah
EPA Project Officer: Aja, Hayley
Project Period: December 1, 2009 through November 30, 2012 (Extended to November 30, 2013)
Project Period Covered by this Report: March 16, 2010 through March 15,2011
Project Amount: $899,567
RFA: Integrated Design, Modeling, and Monitoring of Geologic Sequestration of Anthropogenic Carbon Dioxide to Safeguard Sources of Drinking Water (2009) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
Progress Summary:
Figure 1. (a) Potential leakage pathway through an abandoned well [Norbotten, et al., 2005]; (b) Changes of geochemical signature above CO2 injection formation in Weyburn CO2-enhanced oil recovery site [Emberley, et al., 2004]
| Name | Location | Height | Interval | Duration |
| Crystal Geyser | Green River Utah, USA | 1520 meters | 1118 hours | 1545 minutes |
| Woodside Geyser (Roadside Geyser) | Woodside Utah, USA | 610 meters | 28 minutes | 1.01.5 hours |
| Champagne Geyser (Chaffin Ranch Geyser) | Green River Utah, USA | 78 meters | 2 hours | 5 minutes |
| Ten Mile Geyser | Green River Utah, USA | 2.53.5 meters | 6 hours 42 minutes | 51 seconds |
| Tumbleweed Geyser | Green River Utah, USA | 0.31.5 meters | 28.5 minutes | 4694 minutes |
| Unnamed Geyser | Salton Sea California, USA | 0.10.5 meters | 1060 seconds | Seconds |
| Jones Fountain of Life | Clearlake California, USA | < 1.0 meters | 60 minutes | 22 minutes |
| Cold Water Geyser | Yellowstone Wyoming, USA | 0.5 meters | Unknown | 10 minutes |
| Source Intermittente de Vesse | Bellerive, France | 16 meters | 230270 minutes | 4550 minutes |
| Andernach Geyser | Andernach, Germany | 4060 meters | 1.54 hours | 78 minutes |
| Boiling Fount Local name: Brubbel | Wallenborn, Germany | 23 meters | 30 minutes | a few minutes |
| Mokena Geyser | North Island, New Zealand | 0.55 meters | minuteshours | secondsminutes |
| Povremeni Geyser | Sijarinska, Serbia | 20 meters | 9 minutes | 2 minutes |
| Herlany Geyser | Herlany, Slovakia | 2030 meters | 3234 hours | 30 minutes |
| Persi Geyser | Persi, Slovakia | smaller than Herlany Geyser | hours (shorter than Herlany Geyser) | minutes (probably < 30) |
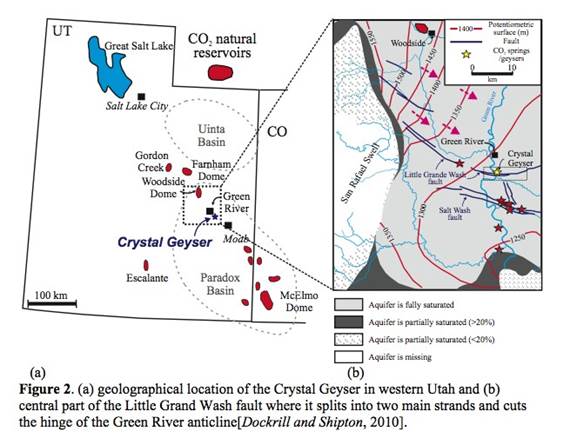
Figure 3. Schematic diagram of well configuration and piezometer installation
| Field Trip | Starting time | Ending time | Eruption type | Duration | Comment |
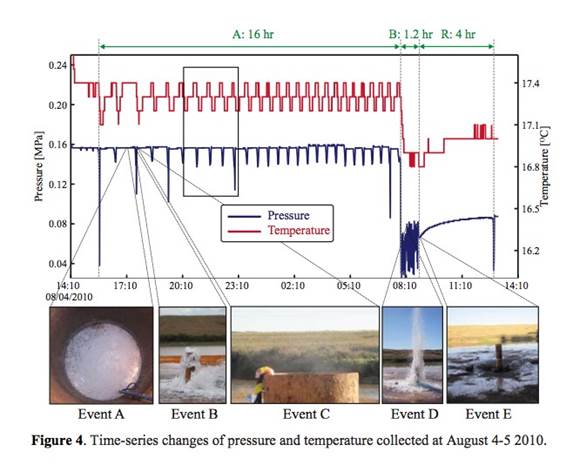
| 8/4-8/6 | 8/4 15:30 pm | 8/5 7:30 am | A-type | 16 hour | |
| | 8/5 7:30 am | 8/5 8:40 am | B-type | 1.2 hour | |
| | 8/5 8:40 am | 8/5 12:40 pm | Recharge | 4 hour | Because the levels of static pressure and temperature were changed, we stopped the recording at the piezometer. |
| | 8/5 12:40 pm | 8/5 18:15 pm | C-type | 5.6 hour | We began to record manually. |
| | 8/5 18:15 pm | 8/6 over 1:00 am (?) | Probably D-type | Over 6.8 hour (?) | Eruptions continued past 1:00 am on 8/6. We do not know the exact time when it stopped. |
| | 8/6 over 1:00 am (?) | 8/6 10:00 am | Recharge | | At 10:00 am on August 6, the geyser was completely drained as shown in Event D of Figure 4. |
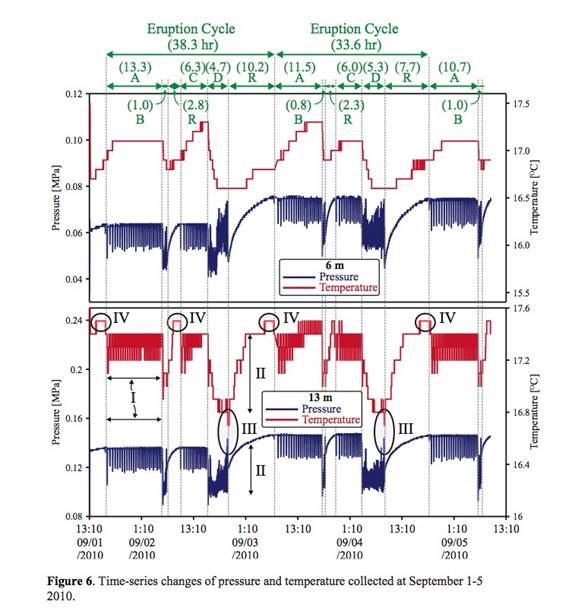
| 9/1-9/5 | 9/1 16:50 pm | 9/2 6:10 am | A-type | 13.3 hour | |
| | 9/2 6:10 am | 9/2 7:10 am | B-type | 1 hour | |
| | 9/2 7:10 am | 9/2 10:00 am | Recharge | 2.8 hour | |
| | 9/2 10:00 am | 9/2 16:20 pm | C-type | 6.3 hour | |
| | 9/2 16:20 pm | 9/2 21:00 pm | D-type | 4.7 hour | PVC pipe attached to piezometers is broken. The level of static pressure and temperature was changed slightly. |
| | 9/2 21:00 pm | 9/3 7:10 am | Recharge | 10.2 hour | |
| | 9/3 7:10 am | 9/3 18:40 pm | A-type | 11.5 hour | |
| | 9/3 18:40 pm | 9/3 19:30 pm | B-type | 0.8 hour | |
| | 9/3 19:30 pm | 9/3 21:50 pm | Recharge | 2.3 hour | |
| | 9/3 21:50 pm | 9/4 3:50 am | C-type | 6 hour | |
| | 9/4 3:50 am | 9/4 9:10 am | D-type | 5.3 hour | |
| | 9/4 9:10 am | 9/4 19:50 pm | Recharge | 7.7 hour | |
| | 9/4 19:50 pm | 9/5 6:30 am | A-type | 10.7 hour | |
| | 9/5 6:30 am | 9/5 7:30 am | B-type | 1 hour | |
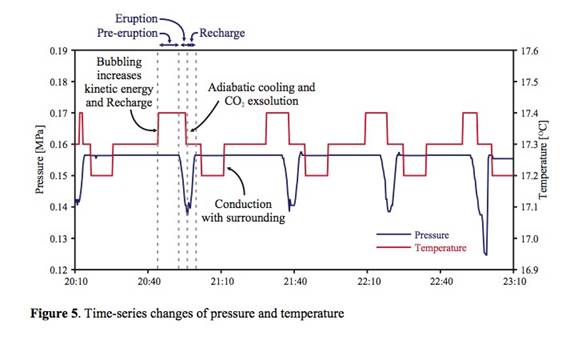
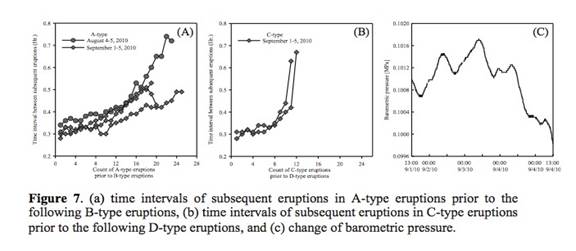
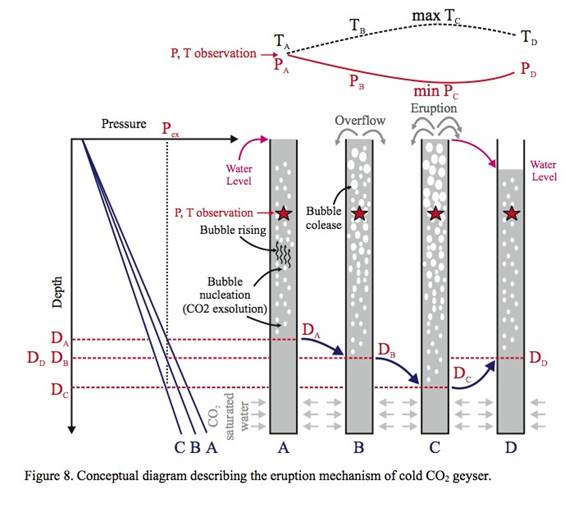
Single Large-Scale Eruption: Single large-scale eruption, characterized by large reduction of both pressure and temperature and, at the same time, longer eruption duration, occurred from 7:30 am to 8:40 am on August 5 after a series of multiple small- scale eruptions (Figure 4). In detail, we differentiated between large- and small-scale eruptions based on three major components such as intensity, duration, and drainage period. First, the intensity of the large-scale eruption was strong and, due to the power of instantaneous CO2 gas emission, the water column reached approximately over 10 m above the surface (Event D in Figure 4) but it only reached the casing top in the small-scale eruption (Event B in Figure 4). Due to the intensity difference, both the pressure and temperature drop was significantly larger in the large-scale eruption than in the small-scale eruption. Second, the average duration of the small-scale eruptions was approximately 0.12 hour (7.2 minutes) but that of large-scale eruption was 1.2 hours. Finally, we did not observe any change of height in the water pool around the geyser immediately after the small-scale eruption although a slight decrease of hydrostatic pressure was measured. However, after the large-scale eruption of 1.2 hours ceased, the water pool was completely drained into the geyser (Event E in Figure 4), and the water level inside the geyser well was approximately 2.5 to 3 m below the surface.
- The Gordon Creek field in north-central Utah, just 25 miles from the original Farnham Dome site (and thus our baseline geologic and hydrologic knowledge is very solid). This site has never been subjected to CO2 injection, and will not begin injection for at least 1 year from now.
- Our SWP (Phase 2) field site in the Permian Basin in western Texas; we started CO2 injection at this site in October 2008, and injection is not slated to cease until 2010 at the earliest.
- Our SWP (Phase 2) field site in the San Juan Basin in northern New Mexico; we started CO2 injection at this site in July 2008, and injection is ceasing this week (the week of August 17, 2009).
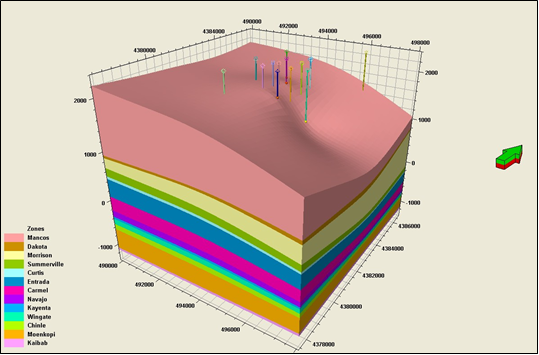
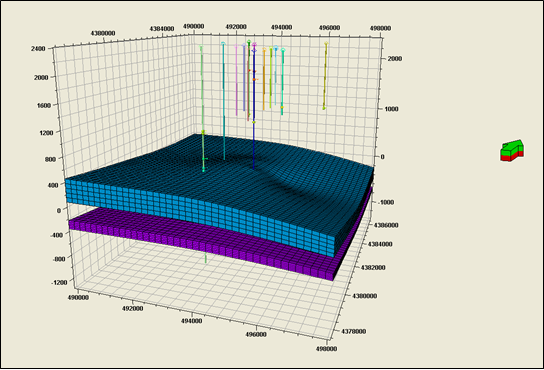
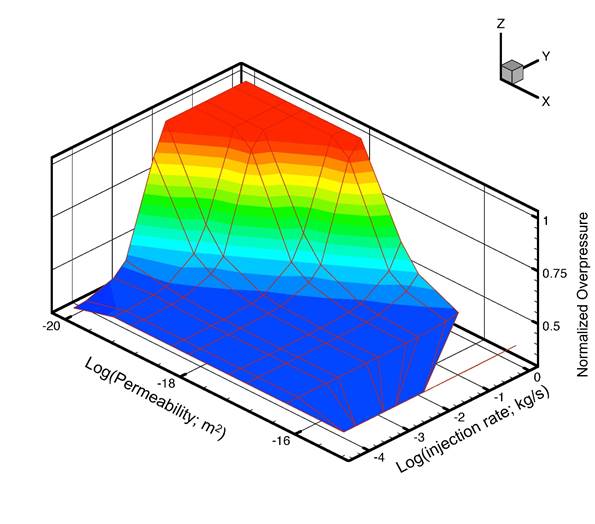
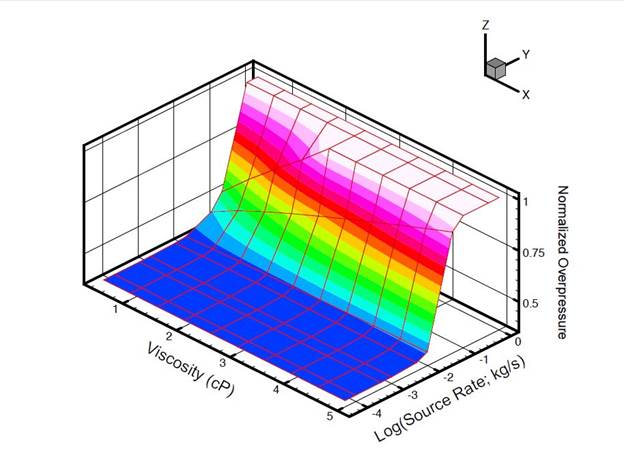
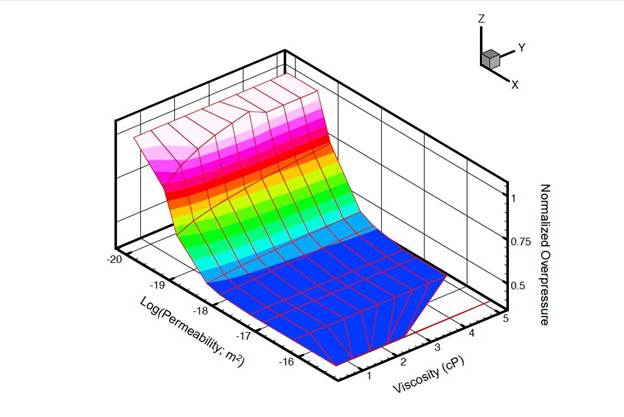

- Development of a methodology for calculating the probability of leakage of CO2 into USDWs, especially via known (or unknown) fault pathways;
- A study of the impact of the fault structure on possible leakage of CO2 into overlying formations.


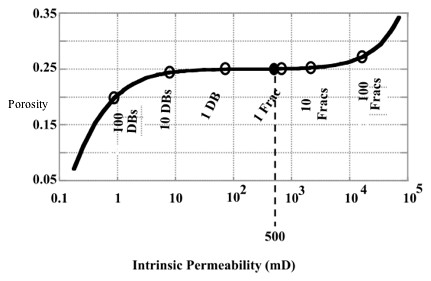
Future Activities:
- Summary report of leakage mechanisms and processes for natural analogue leakage sites;
- Summary report of characterization of a natural CO2 storage site (non-leaking) site; the planned site for this analysis and report is the Farnham CO2 Dome in central Utah.
- Summary report of Gordon Creek model simulation analyses for basic CO2 migration and leakage assessment (these simulations will be the primary tool for developing probability density functions associated with selected risks).
- If time affords, we also will develop summary reports of analyses of the other two field sites (San Juan Basin, NM, and SACROC field, TX).
- Summary report of probability density functions (development method and outcomes) for an engineered storage site. Given the presence of a major fault located 500 meters from the planned injection site, we will develop probability density functions (PDFs) for risk of (a) leakage through this fault, and (b) induced slip on this fault, following CO2 injection. We will deliver these PDFs and a detailed description of the process and algorithms used to develop these PDFs.
- Generalized Risk Registry (tabulation of risks) for sites with USDWs present.
- Detailed (specific) risk registry for the Gordon Creek field site.
- Summary report of methods and associated validation approach for tracer, microbiologic, chemical and physical field data used for calibration of quantitative risk assessment.
- Brief summary report of approach for application of tracer, microbiologic, chemical and physical field data in reservoir models.
- Brief summary report of approach for application of tracer, microbiologic, chemical and physical field in developing risk estimates (in the form of PDFs).
- Summary report of efficacy of gathered data for refining risk estimates in the form of PDFs.
- A website targeting K-12 students for education regarding geologic CCS, USDWs, potential risks to USDWs, and plans to mitigate those risks.
- Presentations at annual project review meetings.
- Presentations at national and regional conferences to describe the outcomes of this research to colleagues and other stakeholders.
References:
Journal Articles:
No journal articles submitted with this report: View all 3 publications for this projectProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
