Grantee Research Project Results
2009 Progress Report: Clinically Relevant IgE-Cross-Reactivity of Nut Allergens
EPA Grant Number: R834066Title: Clinically Relevant IgE-Cross-Reactivity of Nut Allergens
Investigators: Schein, Catherine H. , Maleki, Soheila , Teuber, Susanne
Current Investigators: Schein, Catherine H. , Teuber, Susanne , Maleki, Soheila
Institution: The University of Texas Medical Branch - Galveston , USDA , University of California - Davis
Current Institution: The University of Texas Medical Branch - Galveston , University of California - Davis , USDA
EPA Project Officer: Aja, Hayley
Project Period: December 1, 2008 through November 30, 2011
Project Period Covered by this Report: December 1, 2008 through June 1,2010
Project Amount: $409,927
RFA: Exploratory Investigations in Food Allergy (2007) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Progress Summary:
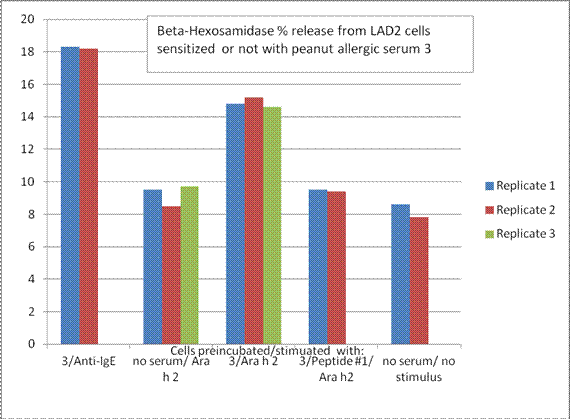
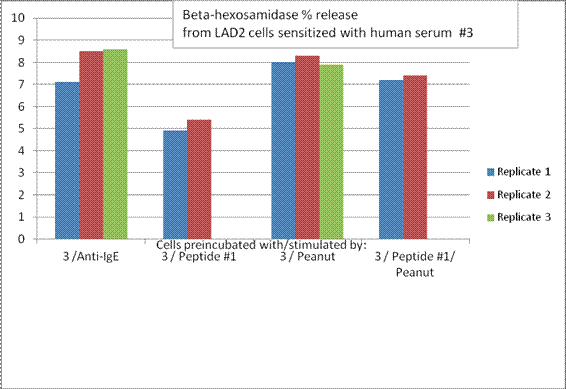
Future Activities:
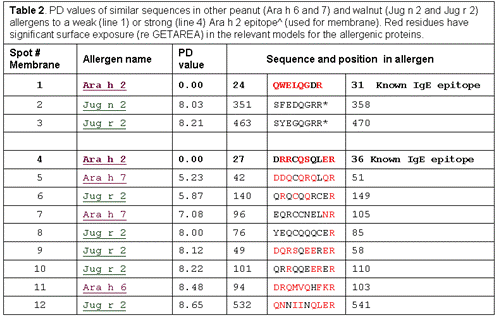
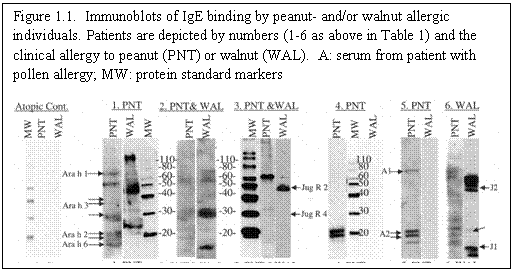
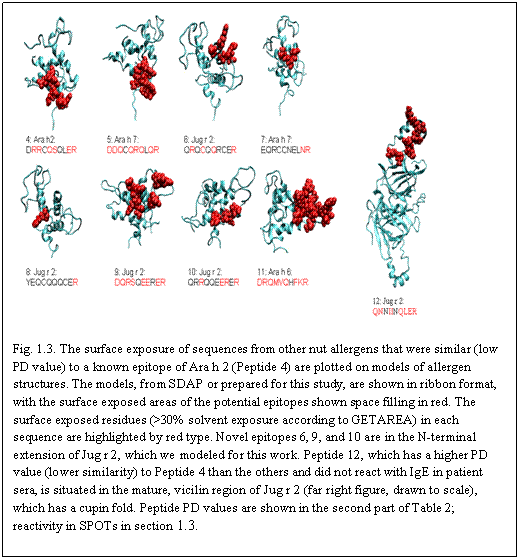
Aim 1: The original characterization was done with 6 patients, and one control selected to have high serum IgE but primary allergy to pollen, rather than nuts. We are expanding the patient pool, and will characterize the new group of sera as below, using strips of selected sequences on spot assays, Western blotting, and, where appropriate, competition assays.
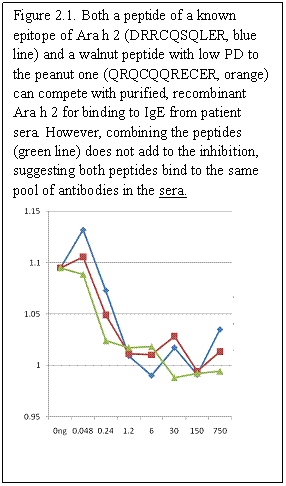
Aim 2: Other characterized sera will be tested in these ELISA competitions.
Aim 3: The finding that the Peptide 1, a known IgE binding sequence from Ara h 2, could inhibit basophil degranulation stimulated by Ara h 2, suggests that this might aid in a novel form of de-sensitization (or antibody blocking) therapy using peptides that can bind to the IgE but not trigger degranulation. Dr. Teuber’s group is now trying the other peptides, in combination with different sera, to see how specific the effect is for the chosen peptide.
References:
Journal Articles:
No journal articles submitted with this report: View all 14 publications for this projectProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
