Grantee Research Project Results
2011 Progress Report: Near-real Time, Highly Sensitive and Selective Field Deployable Biosensor for Cyanotoxins and Cyanobacteria Using both Antibodies and DNA-signatures
EPA Grant Number: R833829Title: Near-real Time, Highly Sensitive and Selective Field Deployable Biosensor for Cyanotoxins and Cyanobacteria Using both Antibodies and DNA-signatures
Investigators: Mutharasan, R.
Institution: Drexel University
EPA Project Officer: Aja, Hayley
Project Period: June 10, 2008 through March 31, 2011 (Extended to September 30, 2013)
Project Period Covered by this Report: October 1, 2009 through September 30,2012
Project Amount: $599,999
RFA: Development and Evaluation of Innovative Approaches for the Quantitative Assessment of Pathogens and Cyanobacteria and Their Toxins in Drinking Water (2007) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
The overall goal of this research is to develop piezoelectric-excited millimeter-sized cantilever sensors (PEMC) for cyanotoxins in source, The overall goal of the proposed research is to develop piezoelectric-excited millimeter-sized cantilever sensors (PEMC) for cyanotoxins in source, finished and system waters that measures in a field-deployable format and rapidly in 15 minutes so that cyanotoxin(s) hazard and management decisions can be made in a timely fashion. The cantilever sensors are of unique design and are the result of development in the PI’s lab over the past 5 years. PEMC’s significant advantage is the use of label-free reagents and a proposed simple measurement format. PEMC sensors exhibit high sensitivity such that greater than femtograms per liter (10-15 g/L; parts per quintillion) cyanotoxin concentration would be measurable directly by dipping into a few milliliters water samples. PEMC sensors are not the fragile AFM-like microcantilevers but are mechanically robust millimeter-sized resonant mode cantilevers that exhibit a high-order mode that measures mass changes at the femtogram (10-15 g) level under liquid immersion and flow conditions.
Progress Summary:
1.0 Research Method and Results—Microcystin Detection
1.1 Experimental apparatus and methods
1.2 Sensor mass calibration
1.3 Water sample experiments
1.4 Sensor response to MCLR in three water media
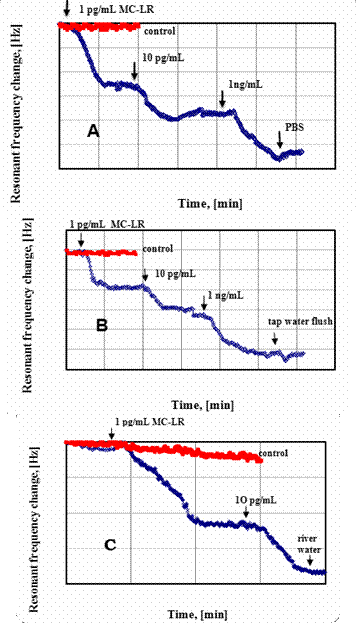
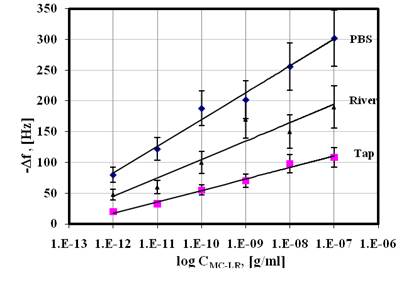
1.5 Estimates of detection limit and dynamic range
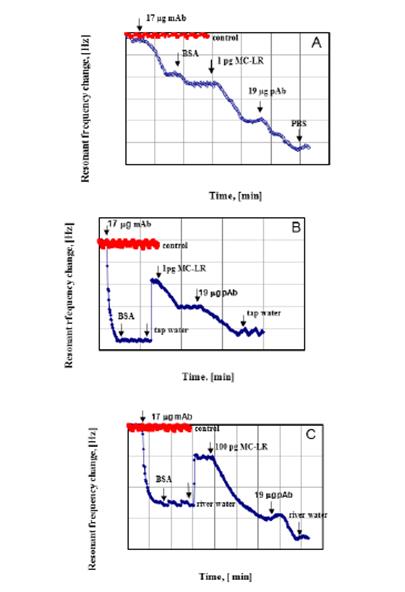
1.6 Confirmation of detection using sandwich assay
2.0. Research Method and Results – Cyanobacteria detection


- Centrifuge cell suspension at 14,000 g for 15 min to get a cell pellet.
- Remove supernatant liquid.
- Resuspend cell pellet in 200 µL of TE buffer.
- Add 100 µL of 10% SDS in DI water.
- Incubate cell suspension at 100°C for 15 min.
- Immediately incubate the cell suspension at 2–4°C for 10 min.
- Add 600 µL of chloroform and mix well by inverting for 1–2 min.
- Centrifuge the cell suspension at 14,000 g for 2 min.
- Take the aqueous supernatant in to a separate vial without disturbing the aqueous-organic interface.
- Add 50 µL of TE buffer with 1M NaCl and mix well.
- Add 800 µL of cold ethanol and mix well.
- Incubate the vial at −20°C for 8–12 hr for DNA precipitation.
- Centrifuge the vial at 14,000 g for 30 min.
- Discard the supernatant.
- Wash the DNA pellet with cold ethanol.
- Let the ethanol evaporate at room temperature.
- Suspend the DNA pellet in TE buffer.
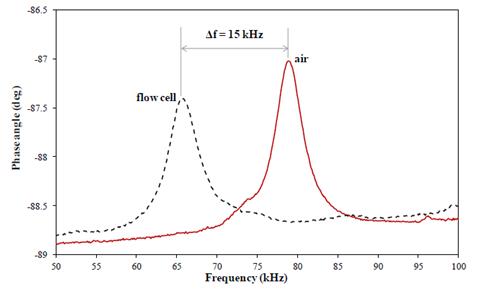
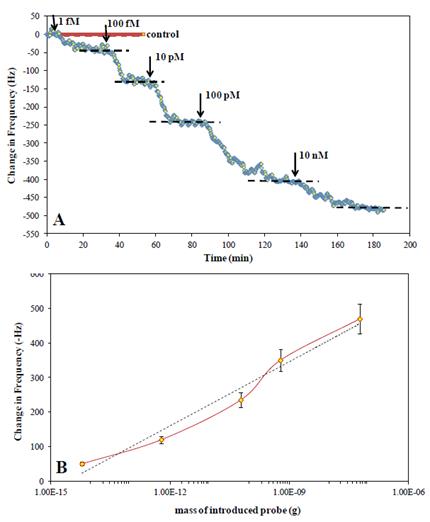
Future Activities:
We are in the process of conducting DNA-based detection experiments with a model cyanobacterium, Microcystis aeruginosa.We are in the process of conducting DNA-based detection experiments with a model cyanobacterium, Microcystis aeruginosa.
Having established DNA extraction protocol, and sensor sensitivity we are currently conducting hybridization experiments with synthetic DNA strands. Initial results indicate that we can measure easily ssDNA at a few femtograms. During coming 3 months we are planning to complete hybridization experiments followed by genomic DNA extracts and finally during the summer complete the sensing done in river water.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 2 publications | 2 publications in selected types | All 2 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Ding Y, Mutharasan R. Highly sensitive and rapid detection of microcystin-LR in source and finished water samples using cantilever sensors. Environmental Science & Technology 2011;45(4):1490-1496. |
R833829 (2011) R833829 (Final) |
Exit |
Supplemental Keywords:
resonant frequency, ultrasensitive, immuno-bindingProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
