Grantee Research Project Results
2009 Progress Report: Comparative Toxicity of Coarse Particles
EPA Grant Number: R833742Title: Comparative Toxicity of Coarse Particles
Investigators: Gordon, Terry , Lippmann, Morton , Chen, Lung Chi , Ito, Kazuhiko
Institution: New York University School of Medicine
EPA Project Officer: Chung, Serena
Project Period: March 1, 2008 through February 28, 2012
Project Period Covered by this Report: March 1, 2009 through February 28,2010
Project Amount: $1,199,927
RFA: Sources, Composition, and Health Effects of Coarse Particulate Matter (2006) RFA Text | Recipients Lists
Research Category: Particulate Matter , Air Quality and Air Toxics , Air
Objective:
The objective of this study is to determine the contribution of coarse particles to the adverse health effects associated with exposure to ambient PM. The aims and objectives have not changed from the original submission. We hypothesized that differences in the toxicity of coarse PM (PM10-2.5) samples are due to the source contributions to the particles, and in testing this hypothesis, we are: 1) measuring the differential toxicity of coarse particles both in vitro and in vivo; and 2) identifying whether coarse particles from urban and rural sources differ in toxicity. A number of investigators have clearly demonstrated that PM toxicity in the mammalian lung is governed, in part, by particle size, but little research has been published on whether the physicochemical properties of coarse particles influence their toxicity in mammalian cells. In the proposed studies, a group of particle toxicologists are collaborating with a source apportionment epidemiologist to explore the toxicity of a variety of urban and rural coarse particles in established models of mammalian cell toxicity.
Progress Summary:
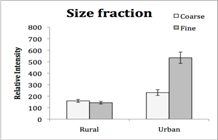
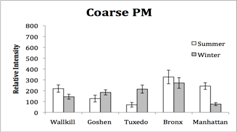
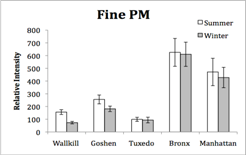
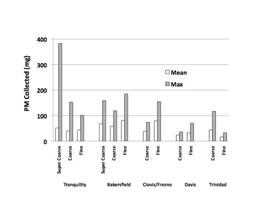
1) Overview of work status and preliminary results: The work on the project aims has proceeded smoothly but at a slower pace than anticipated. The delays have primarily been due to delays in the digestion of PM samples and the trace element analyses of PM samples. A microwave digestion system was purchased and a protocol developed to more rapidly address the preparation of hundreds of PM samples for ICP-MS analyses of trace elements. The in vitro bioassays have been completed for the urban and rural NY samples. The sampling of San Joaquin Valley, CA PM is complete and the preparation for PM samples for chemical analyses and bioassays is scheduled for Year 3.
|
Mass Concentration (µg/m3) of PM Collected
|
|||||
|
|
Super Coarse
(> PM10)
|
Coarse
(PM10-2.5)
|
Fine
(PM2.5)
|
||
|
Bronx
|
Urban
|
7.18
|
7.74
|
10.69
|
|
|
Manhattan
|
Urban
|
ND
|
9.63
|
10.88
|
|
|
Goshen
|
Rural
|
ND
|
5.99
|
8.30
|
|
|
Tuxedo
|
Rural
|
ND
|
4.91
|
7.06
|
|
|
Wallkill
|
Rural
|
2.94
|
4.80
|
7.15
|
|
|
Mass Concentration (µg/m3) of PM Collected
|
|||||
|
|
Coarse
|
Coarse
|
Fine
|
Fine
|
|
|
Summer
|
Winter
|
Summer
|
Winter
|
||
|
Bronx
|
Urban
|
7.25
|
8.34
|
9.21
|
12.47
|
|
Manhattan
|
Urban
|
8.02
|
11.55
|
9.50
|
12.53
|
|
Goshen
|
Rural
|
5.54
|
7.06
|
6.35
|
10.65
|
|
Tuxedo
|
Rural
|
5.06
|
4.72
|
5.85
|
8.51
|
|
Wallkill
|
Rural
|
5.23
|
4.77
|
5.46
|
8.99
|
Future Activities:
The major objective for the remainder of the support period is to complete the bioassays for the California samples and complete the trace element analyses.
Journal Articles:
No journal articles submitted with this report: View all 12 publications for this projectSupplemental Keywords:
RFA, Air, Scientific Discipline, Health Risk Assessment, particulate matter, Biology, bioavailability, sensitive populations, sensitive subgroups, atmospheric particles, cardiopulmonary responses, cardiovascular vulnerability, cardiotoxicity, atmospheric particulate matter, chemical speciation sampling, human health effectsProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
