Grantee Research Project Results
Final Report: The Bioavailability, Toxicity, and Trophic Transfer of Manufactured ZnO Nanoparticles: A View from the Bottom
EPA Grant Number: R832530Title: The Bioavailability, Toxicity, and Trophic Transfer of Manufactured ZnO Nanoparticles: A View from the Bottom
Investigators: Bertsch, Paul M. , Neal, Andrew , Jackson, Brian , Williams, Phillip , Glenn, Travis
Institution: Savannah River Ecology Laboratory , University of Georgia , Dartmouth College
Current Institution: Savannah River Ecology Laboratory , Dartmouth College , University of Georgia
EPA Project Officer: Hahn, Intaek
Project Period: October 1, 2005 through September 30, 2008 (Extended to September 30, 2009)
Project Amount: $363,680
RFA: Exploratory Research: Nanotechnology Research Grants Investigating Environmental and Human Health Effects of Manufactured Nanomaterials: A Joint Research Solicitation - EPA, NSF, NIOSH (2005) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
The overall objectives of our research are to evaluate: 1) the bioavailability and toxicity of manufactured nanoparticles (ZnO-np) as a function of particle size to the model bacteria, Burkholderia vietnamiensis PR1301 and the model detritivore Caenorhabditis elegans as referenced against aqueous Zn2+ (Zn2+ aq), 2) the ability of manufactured ZnO-np to be transferred from one trophic level to the next as assessed in the simple food chain consisting of pre-exposed PR1 and C. elegans, and 3) the synergistic or antagonistic effects of manufactured ZnO-np on the toxicity of Cu2+ to PR1 and C. elegans. These three overall objectives will be approached in the context of the following four hypotheses:
Hypothesis 1: The bioavailability and toxicity of manufactured ZnO-np increases with decreasing particle size (i.e., 3 nm vs. 80 nm).
Hypothesis 2: The toxicity of ZnO nanoparticles to PR1 and C. elegans is lower than an equivalent concentration of dissolved Zn2+.
Hypothesis 3: The bioavailability and toxicity of ZnO-np introduced via trophic transfer differs from direct exposure.
Hypothesis 4: ZnO-np alter the bioavailability and toxicity of dissolved metals
Summary/Accomplishments (Outputs/Outcomes):
GRANT UPDATES
The research designed to address the overall objectives and test the proposed hypotheses was separated into identifiable tasks and spread over the project’s more than three year span (Fig 1). While the overall aim of the project did not change, the trophic transfer research on pre-exposed B. vietnamiensis consumed by C. elegans was analytically challenging and experiments were expanded to include an additional detritivore, the model earthworm Eisenia fetida. Also, based on initial results on the importance of acetate, a constitutive acetate utilizing metal sensitive bacteria Cupriavidus necator was included starting in year 2.
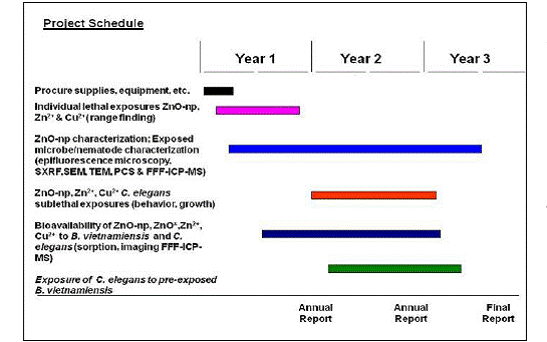
Figure 1. Project schedule delineating specific tasks spread over the project’s three year span.
The grant was transferred from the University of Georgia to the University of Kentucky (UK) in June-July 2008. The personnel originally hired on the grant were kept to ensure continuity of the project. Nevertheless, de-commissioning, moving and reestablishing laboratories at UK as well as personnel transfer resulted in dead-time and delayed our projected schedule of work. As a result of the downtime associated with decommissioning and reestablishing laboratory facilities, a no-cost extension through September 30, 2009 was requested and granted. This ensured that all research objectives were met as well as provided an appropriate time frame for results to be published in journals and the final project report to be submitted.
PERSONNEL
As specified in the proposal, an interdisciplinary post doctoral associate was hired to oversee the day-to-day research activities of the project. Also, two of the PIs served as advisors to graduate students involved with different aspects of the project. One of the students (B.A. Neely) received an EPA STAR Fellowship, partially focused on the ZnO nanoparticle work.
Post doctoral associate: Nadine J. Kabengi
Students: Hongbo Ma. Major: Toxicology
Department: Environmental Health Science, The University of Georgia
Advisor: Phillip William
Benjamin A. Neely. Major: Environmental Microbiology
Department: Marine Biomedicine and Environmental Science Program,
Medical University of South Carolina
Advisors: Paul M. Bertsch and Pamela J. Morris
Collaborators: Art Grider (University of Georgia), Pamela J. Morris (Medical University of South Carolina), John Shields (University of Georgia), Jason Unrine (University of Kentucky), and Jinsong Wu (Northwestern University)
As of September 2009, both students (Hongbo Ma and Benjamin Neely) successfully completed their Ph.D. degrees.
RESULTS
Research activities in this project focused on: 1) characterization of smaller (3 nm- s-ZnO-np) and larger (80 nm-l-ZnO-np) ZnO-np under physicochemical conditions of the exposure experiments, 2) initial range finding experiments examining the bioavailability and toxicity of s-ZnO-np to B. vietnamensis, C. elegans and E. fetida as referenced to dissolved Zn2+aq, 3) understanding the bioavailability and toxicity of l-ZnO-np to C. elegans, 4) deciphering differences in toxicity mechanisms of s-ZnO-np and aqueous zinc acetate to C. necator using proteomics approaches, 5) delineating the effects of s-ZnO-np and ZnCl2 on Cu toxicity, and 6) evaluating the bioavailability and toxicity of s-ZnO-np introduced via trophic transfer as opposed to direct exposure.
1. Characterization
s-ZnO-np:
The 3 nm (± 2-6 nm) s-ZnO-np suspsensions were obtained from Applied Nanoworks (http://www.appliednanoworks.com). Ion chromatography indicated that the sole counter anion used to stabilize the suspensions was acetate although the concentrations of acetate were somewhat higher (3.07 M) than reported by the manufacturer. Other studies demonstrated that the stability of the s-ZnO-np suspension was dependent on acetate concentration and pH. High resolution thermogravimetric analysis (HR-TGA) coupled with traditional chemical methods suggested the existence of multiple populations of acetate associated with the s-ZnO-np, which affect both their stability and reactivity. Characterization data obtained with a variety of techniques including TEM, XRD, FTIR and FT-Raman spectroscopies reveals that the ZnO primary particles undergo irreversible structural transformations accompanying acetate removal (Fig 2), with the resulting ZnO aggregates exhibiting surface reactivity similar to that of bulk ZnO, having a mean particle size of 1.5 µm.

Figure 2. TEM of s-ZnO-np A) dried at 60°C and B) after removal of all acetate by heating to 450°C
Additional characterization of s-ZnO-np revealed discrepancies in the particle size determined by TEM imaging compared to dynamic laser light scattering (DLS). Sample observations with TEM resulted in crystal growth transformations of s-ZnO-np. While some growth transformations appear to be induced by sample heating; other growth seems to be induced by additional e-beam effects on the nanoparticles surface chemistry. Cooling samples with a cryo stage during analysis compensated for the beam-induced heating effects.
Surface reactivity assays using methylene blue degradation under UV irradiation revealed that the s-ZnO nanoparticle surfaces were passivated, presumably from strong acetate binding to O unsaturated crystal planes (see Fig. 3). As discussed above, the removal of acetate by heating resulted in crystal growth but restored surface reactivity as assessed by the methylene blue assay (activity similar to ZnO bulk-data not shown).

Figure 3. Left: Transmission Electron Micrograph of l-ZnO-np sample; Right: Methylene Blue (MB) assay showing the degradation of MB after exposure to UV lights in presence of s-ZnO-np, l-ZnO-np and ZnO bulk. Increased discoloration is an indication of higher surface reactivity
l-ZnO-np:
Powdered nanoparticulate ZnO (NanoGard® zinc oxide) was purchased from Alfa Aesar (Ward Hill, MA, USA) with a stated size of 40-100 nm. The l-ZnO-np were found to be 75-80% zinc. While acetate controlled the surface reactivity through passivation of surface sites for the s-ZnO-np, this was not the case for l-ZnO-np which also had greater stability under TEM and a much greater surface reactivity as determined by the Methylene Blue degradation assay (Fig 3). Significant particle aggregation was observed under experimental conditions; hence all test solutions for further experiments were freshly diluted from the stock, which was sonicated for 2 h prior to making dilutions to ensure proper dispersion of the materials.
2. Bioavailability and Toxicity of s-ZnO-np
Nanoparticle-bacteria interactions: The presence of acetate in s-ZnO-np suspension was shown to affect Burkholderia vietnamiensis PR1301 (PR1) growth. At neutral pH, the acetate anion can serve as a non-specific substrate while at acidic pH, the undissociated acid can be cytotoxic. The microbial acetate utilization may influence the stability of s-ZnO-np during the course of exposure. With acetate concentration normalized to 10.7 mM, PR1 grew at 25, 50, 75, 100, and 250 mg L-1 Zn as s-ZnO-np or as aqueous ZnCl2 at both pH 6 and pH 7. A pH dependent toxicity was observed for PR1, with toxicity observed at lower concentrations at pH 7 vs. pH 6. This pH dependent toxicity was observed with both aqueous ZnCl2 and s-ZnO-np. The EC50 values derived from growth curves (Fig 4) of s-ZnO-np and aqueous ZnCl2 at pH 6 were significantly different (α=0.05) at 188.6 ± 8.5 mg L-1 and 163 ± 9.6 mg L-1, respectively. At pH 7, the EC50 of s-ZnO-np and aqueous ZnCl2 values were similar at 104.4 ± 13.5 mg L-1 and 96.6 ± 14.1 mg L-1, respectively. While the overall trends of toxicity were very similar, the underlying mechanisms are likely different. At 250 mg L-1 s-ZnO-np (pH 6), significant flocculation of primary particles was observed following 24 hours of cell growth. Aggregates do not form at 250 mg L-1 s-ZnO-np in the absence of PR1 suggesting a biologically controlled induction. Aggregates could result from acetate degradation and/or exopolymer secretion.
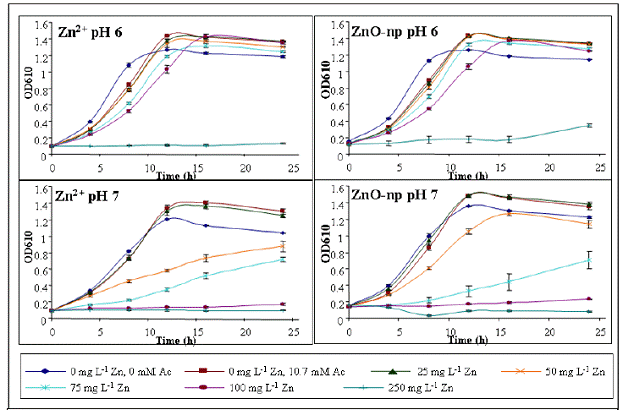
Figure 4. PR1 growth curves with equivalent Zn concentrations as aqueous Zn and ZnO-np and acetate concentrations normalized at 10.7 mM.

Figure 5. Fluorescence micrograph of mtl2::GFP (pseudo-color representation) expression in C. elegans exposed to s-ZnO-np. Inset shows Zn distribution determined from ZnKα fluorescence
Nanoparticle-nematode interactions: The effects of s- ZnO-np and aqueous ZnCl2 to Caenorhabditis elegans were evaluated by conducting lethality tests and by examining the spatial distribution of Zn from s-ZnO-np and ZnCl2 in exposed worms by 2D XRF elemental mapping. Acetate was not toxic to C. elegans. The LC50 values were 789 ± 147 mg and 884 ± 150 mg Zn L-1 for s-ZnO-np and ZnCl2 respectively (Fig 5). No significant difference in general toxicity to C. elegans was found between s-ZnO-np and aqueous ZnCl2, yet, as with the bacteria, the mechanism of toxicity is likely different. The spatial distribution of Zn was determined by synchrotron-based XRF and was found to be localized in the posterior region of control worms. Zn distribution in s-ZnO-np and ZnCl2 exposed nematodes showed similar patterns with the areas of maximum intensity spreading out through the nematode as exposure concentration increases. The maximum intensities in ZnCl2 exposed nematode are approximately twice as high as in s-ZnO-np exposed nematode, which may suggest their different bioavailability. Other imaging experiments demonstrated that while the Zn spatial distribution and mtl2::GFP expression in control nematodes is not correlated, a co-localization of Zn and mtl2::GFP expression exist in worms exposed to s-ZnO-np and ZnCl2 (fig 5). There was also evidence for accumulation of the “aging” pigment, lipofucin, which has been used as a biomarker for stress in previous studies in exposed worms and suggests that worms exposed to Zn2+ and s-ZnO are experiencing stress that results in the accumulation of lysosomal debris (fig 6).
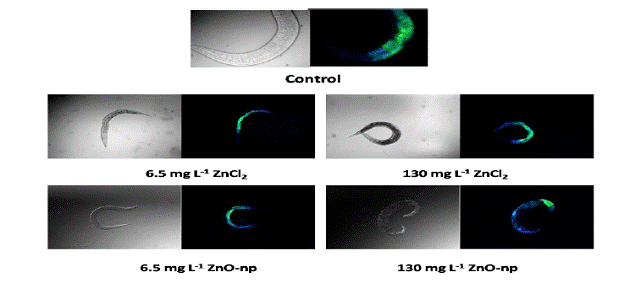
Figure 6. Differential interference contrast and corresponding fluorescence micrographs of Caenorhabditis elegans exposed to two concentrations of ZnCl2, a commercial ZnO nanoparticle suspension, or control medium. Green fluorescence corresponds to green fluorescent protein, a tag for the metallothionein II promoter. Blue fluorescence likely corresponds to lipofuscin or “age pigment”.
Nanoparticle-earthworm interactions: The effects of s-ZnO-np and aqueous ZnCl2 to Eisenia fetida were evaluated by exposing worms to s-ZnO-np and ZnCl2 following the US-EPA earthworm toxicity protocol (OPPTS 850.6200) and by examining the spatial distribution of Zn from s-ZnO-np and ZnCl2 by 2D XRF elemental mapping. After 14 days of exposure to artificial soil containing 1000 µg g-1 Zn (Fig 7-A), there was significantly more Zn accumulated in s-ZnO-np exposed worms. Zn-rich granules were detected in control worms but not in exposed worms. The Zn-rich granules in control worms were accompanied by an elevated concentration of Zn-bound metallothienin in earthworm tissues and are believed to represent a Zn storage mechanism in response to Zn deficient conditions in the artificial soil. Evidence for Zn stress in the controls was provided by total Zn analysis of the soils prior to and following the 14 day exposure, where 85-95% of the total Zn was removed from control soils. The zinc tissue and molecular distribution in s-ZnO-np and ZnCl2 exposed earthworms were similar, with high intensity in gut area likely to be associated with ingested sediments (Fig 7-B). The absence of granules and Zn-MT in s-ZnO-np treatment suggests bioavailability of Zn.
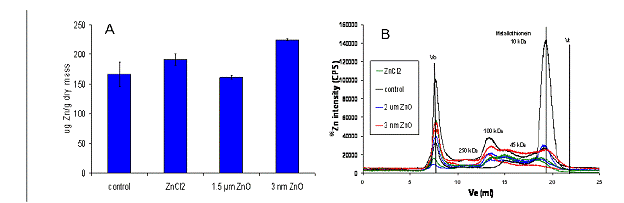
Figure 7. A) Bioaccumulation and B) Molecular distribution of Zn in Eisenia fetida after 14 days of exposure to artificial soil containing 1000 µg g-1 Zn
3. Bioavailability and Toxicity of l-ZnO-np to nematodes:
The toxicity of l-ZnO-np to C. elegans was evaluated by conducting lethality tests under natural sunlight illumination; and compared it to toxicity under ambient artificial laboratory light. Bulk ZnO (~1.5 µm primary particle diameter) and aqueous ZnCl2 were used as reference toxicants. Particle aggregation for both l-ZnO-np and bulk ZnO was significant in the test medium as revealed by transmission electron microscopy, dynamic light scattering, and differential interference contrast microscopy (Fig 8).
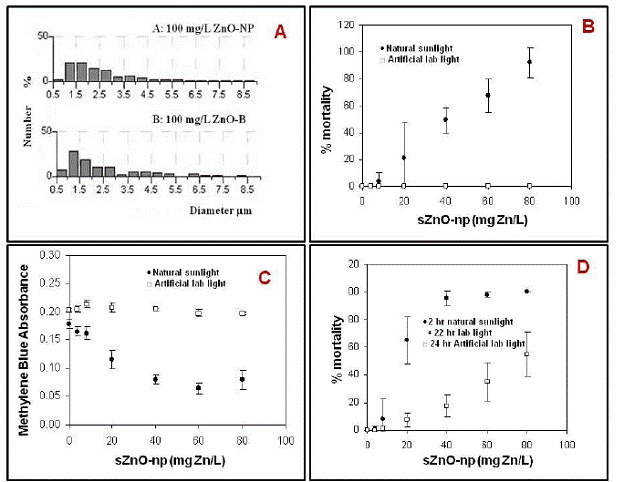
Figure 8. A: Size distribution of l-ZnO-np and bulk ZnO suspensions using differential interference contrast microscopy. B: Two-hour lethal toxicity of l-ZnO-np (LC50 = 39(±8) mg Zn/L) to the nematode C. elegans ; C: Two-hour methylene blue degradation by l-ZnO-np ; D: Twenty-four-hour lethal toxicity of l-ZnO-np to the nematode C.elegans. In all graphs, error bars represent standard errors (n=3).
Both l-ZnO-np and bulk ZnO exhibited significant phototoxicity with 2-h LC50s of 39±8 (mean±SEM, n=3) and 67±8 mg Zn/L, respectively (Fig 8-B).
Photocatalytic activity of l-ZnO-np and bulk ZnO measured by methylene blue degradation (Fig 8-C) showed strong positive correlation with mortality in the nematodes, suggesting the mode of action of the phototoxicity is closely associated with photo-catalytic activity of the metal oxide. Toxicity in the presence of ambient artificial laboratory light was much less than in the presence of sunlight for both l-ZnO-np and bulk ZnO. The 24-h exposure under ambient laboratory light caused less lethality than 2-h exposure under natural sunlight illumination (Fig 8-D); LC50 (l-ZnO np, sunlight plus laboratory) = 17(±4) mg Zn/L, LC50 (ZnO bulk, sunlight plus laboratory) = 38 (±6) mg Zn/L. Toxicity in the presence of ambient light was probably not related to photocatalytic ROS generation as neither nano-ZnO nor bulk ZnO degraded methylene blue. Aqueous Zn2+ showed no lethal toxicity over the concentration range tested, demonstrating that the mechanism was not extra- or intracellular dissolution of the l-ZnO-np. These results suggest that phototoxicity of l-ZnO-np may occur under natural sunlight illumination but that there are other particle specific toxicity mechanisms operating.
4. Bioavailability and Toxicity of s-ZnO-np to C. necator: proteomics analysis
Since initial experiments with the metal tolerant B. vietnamiensis demonstrated that removal of acetate resulted in the flocculation and aggregation of s-ZnO-np primary particles, experiments with a metal sensitive, constitutive acetate utilizing bacteria, C. necator were conducted. As with previous experiments, no significant difference in the growth rate of C. necator following exposure to s-ZnO-np and aqueous zinc was observed; however, C. necator displays higher acetate utilization rates with aqueous zinc compared to s-ZnO-np, suggesting differences in bioavailability of the s-ZnO-np and free Zn ions. Additionally, use of membrane permeable (DAPI) and impermeable (SYTOX orange) dyes, demonstrated a higher incidence of compromised cells associated with sZnO-np exposure compared to the free ion. Different protein expression profiles (2D-SDS-PAGE) were obtained from C. necator exposed to s-ZnO-np and Zn2+ with the statistically significant and at least two-fold down regulation of six key proteins observed for ZnO-np compared to Zn2+ exposed bacteria (Fig 8).
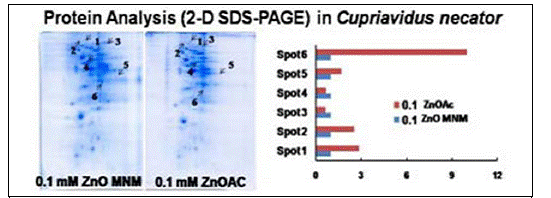
Figure 8. Proteomic responses of the bacteria Cupriadus necator to s-Zno-np differ from responses to aqueous zinc acetate.
5. Effect of s-ZnO-np and Zn2+ on Cu toxicity:
There was no significant difference in the impact of s-ZnO-np and Zn2+ on Cu toxicity to the nematode C. elegans when they are at relatively low concentration (25 and 50 ppm Zn). However, at higher zinc concentrations (> 100 mg L-1), s-ZnO-np decreases copper toxicity compared to aqueous zinc (Fig 9).
The presence of 500µM Zn (as either s-ZnO-np or ZnCl2) increased GFP transgene expression response to Cu exposure in mtl2::GFP transgenic C. elegans. Yet, there is no significant difference in this effect between s-ZnO-np and Zn2+. For most of the Cu concentration tested, elevated GFP response with the presence of 500µM s-ZnO-np or Zn2+ was greater than the GFP response induced by 500µM s-ZnO-np alone, suggesting there might be a synergistic interaction between Cu and s-ZnO-np/ Zn2+ (Fig 9).
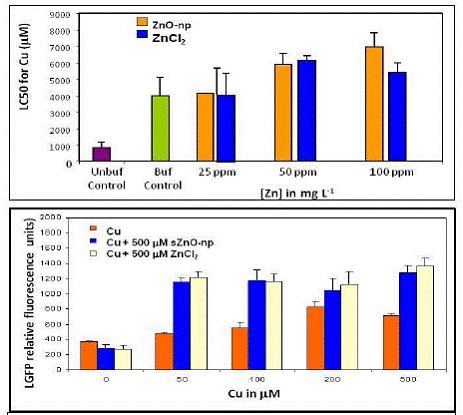
Figure 9. Top: effect of s-ZnO-np or ZnCl2 on Cu toxicity to C. elegans; Bottom: GFP transgene expression response to Cu and s-ZnO-np or ZnCl2 exposure in mtl2::GFP transgenic C. elegans.
6. s-ZnO-np trophic transfer:
There was no evidence for significant trophic transfer in the bacterial-nematode model. However, this may be more related to experimental challenges since the GFP expression profiles are not sensitive to very low concentrations of Zn or s-ZnO-np (Fig 10).
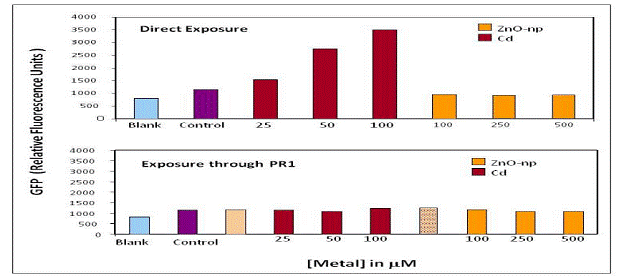
Figure 10. Trophic transfer of s-ZnO-np and Cd in B. vietnamiensis PR1301 and C.elegans model.
Conclusions:
SUMMARY AND MAJOR CONCLUSIONS
The major findings of this research are summarized as follows:
Characterization
-
Size determination and surface chemistry is a critical issue
-
TEM may not be the best method for size determination for small metal oxide nanomaterials
-
Acetate controls s-ZnO-np reactivity, passivates surface sites; not so for bulk (1.2 mm) intermediate for larger (~80 nm) particles
-
Removal of acetate leads to flocculation/ aggregation of s-ZnO-np primary particles but promotes surface reactivity
Interactions with Bacteria
-
No difference in growth rate between s-ZnO-np & Zn2+(aq) for C. necator and B. vietnamiensis PR1301 but both inhibit growth
-
Higher OAc utilization rates with Zn2+ aq compared to s-ZnO-np in Cupriavidus necator
-
Evidence for Zn bioavailability from Zn ion, but not s-ZnO-np
-
More cells with compromised membranes associated with s-ZnO-np exposure compared to free ion
-
Different mechanism(s) of toxicity suggested and supported by different protein expression profiles between Zn2+ aq and s-ZnO exposed microbes.
Interactions with Nematodes
-
s-ZnO-np LC50/EC50 not significantly different from Zn2+(aq) . The behavior and reproduction endpoints are respectively 8-10 and 75-100 times more sensitive than lethality. l- ZnO-np display no toxicity in same concentrations range .
-
Different mechanism (s) of toxicity suggested.
-
At [Zn]>100 mg.L-1, s-ZnO-np decreases Cu toxicity as compared to Zn2+(aq)
-
No significant GFP was induced either in 100uM Cd or 500uM ZnO-np through PR1 exposure (trophic transfer).
-
GFP mtl2 (and mtl1) expression is induced locally but is tissue specific (not necessary related to total Zn)
-
24 hours LC50 for l-ZnO-np and bulk ZnO were 17 ± 4 and 38 ± 6 (mean ± SEM, n = 3) mg Zn L-1, respectively.
-
The phototoxicity of l-ZnO-np is substantially enhanced under natural sunlight illumination as compared to ambient laboratory light illumination or in the dark.
-
Concurrent photodegradation of methylene blue under natural sunlight illumination confirms that this phototoxicity is associated with photoactivation/ROS generation of the nanoparticles.
-
Particle aggregation for l-ZnO-np is significant in the test medium
-
Primary particle size was more important than aggregate size in determining phototoxicity of l-ZnO-np.
-
C. elegans is a good model ecoreceptor for examining nanoparticle toxicity
Additional Remarks
-
Trophic transfer model consisting of pre-exposed bacteria to consuming nematode is challenging since the GFP expression of transgenic nematodes is not a sensitive enough assay
-
s-ZnO-np are bioavailable from soils as demonstrated in earthworms experiments.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 24 publications | 3 publications in selected types | All 2 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Ma H, Bertsch PM, Glenn TC, Kabengi NJ, Williams PL. Toxicity of manufactured zinc oxide nanoparticles in the nematode Caenorhabditis elegans. Environmental Toxicology and Chemistry 2009;28(6):1324-1330. |
R832530 (2008) R832530 (Final) |
Exit |
|
|
Ma H, Kabengi NJ, Bertsch PM, Unrine JM, Glenn TC, Williams PL. Comparative phototoxicity of nanoparticulate and bulk ZnO to a free-living nematode Caenorhabditis elegans: the importance of illumination mode and primary particle size. Environmental Pollution 2011;159(6):1473-1480. |
R832530 (2008) R832530 (Final) |
Exit Exit |
Supplemental Keywords:
Health, Scientific Discipline, Water, Health Risk Assessment, Risk Assessments, Environmental Chemistry, Engineering, Chemistry, & Physics, Biochemistry, nanomaterials, bioavailability, human exposure, trophic transfer, engineered nanomaterials, environmental contaminants, fate and transport, nanotechnology, ambient particle health effects, food chain, human health risk, particle exposure, biochemical researchProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
