Grantee Research Project Results
Final Report: Linking Impacts of Climate Change to Carbon and Phosphorus Dynamics Along a Salinity Gradient in Tidal Marshes
EPA Grant Number: R832222Title: Linking Impacts of Climate Change to Carbon and Phosphorus Dynamics Along a Salinity Gradient in Tidal Marshes
Investigators: Vile, Melanie A. , Neubauer, Scott C. , Weston, Nathaniel
Institution: Villanova University , University of South Carolina at Columbia
EPA Project Officer: Chung, Serena
Project Period: April 14, 2005 through April 13, 2008 (Extended to April 13, 2010)
Project Amount: $705,211
RFA: Effects of Climate Change on Ecosystem Services Provided by Coral Reefs and Tidal Marshes (2004) RFA Text | Recipients Lists
Research Category: Aquatic Ecosystems , Ecological Indicators/Assessment/Restoration , Watersheds , Water , Climate Change
Objective:
Tidal freshwater marshes are often located in areas experiencing intense urbanization pressure, yet provide valuable services to coastal ecosystems. A climate change stressor that is unique to tidal freshwater marshes is the intrusion of salt water into previously freshwater zones. Marshes must accrete to keep pace with rising sea levels, and accretion rates depend on the balance between accumulation and decomposition of sediments. In tidal freshwater marshes, organic carbon (C) accumulation is a major mechanism of marsh accretion, and understanding how changes in salinity will alter pathways of microbial metabolism of marsh C is critical. Our overall objective was to understand how salt water intrusion affects the biogeochemical cycling of C, S, N and P, which in turn affects the balance between C accretion rates, and gaseous C losses from tidal freshwater marshes.
Over the past four years, we have undertaken an extensive effort to determine the impact of climate‐change induced, salt‐water intrusion on tidal freshwater marsh ecosystems in the Delaware Estuary. Our goal was to implement a novel, three‐phase approach to determine changes in tidal marsh metabolism (e.g., CO2 and CH4 gas fluxes and SO4 2‐ reduction), C and P sequestration (sediment deposition and burial), and changes in rates of organic matter decomposition at sites along a low‐salinity transitional gradient in the Delaware Estuary. All three phases of the proposed project were implemented successfully with three complete field seasons of data for field components and one full year of data collection for the lab experiment.
Phase 1 involved finding suitable field sites in the spring and summer of 2005 & 2006 by making appropriate biological (vegetation) and chemical (e.g., salinity) determinations. We spent several months on selecting sites in tributaries of the Delaware estuary with appropriate salinity levels and vegetation (given urbanization pressures, Phragmites invasions to large areas of the Delaware estuary made it difficult to find sites). We established 4 sites in the DE estuary that spanned a range in salinity.
Phase 2 consisted of two components: a laboratory manipulation experiment and field‐based gas flux measurements. We have initiated a longterm laboratory experiments on cores collected from a site representing a freshwater endmember (i.e., Woodbury) of our salinity gradient (Results from the lab study are in press in the peer‐reviewed journal, Biogeochemistry; see attached manuscript). To complement our lab study, we set up field plots in 2007 at three sites along our established salinity gradient, and measured net ecosystem production (NEE) over the field season.
Phase 3 was initiated in 2007, and involves a large‐scale field manipulation (reciprocal transplanting of cores as a space for time substitution) to examine longer‐term, ecosystem‐level responses of marshes to elevated salinity. Since April 2007, we have measured Net Ecosystem Exchange (the balance in C production and consumption), monthly and in some cases bi‐monthly, over the duration of the field season, for two seasons in both permanent and reciprocally transplanted plots). Phase 3 continued through the Fall of 2009.
Results of both the lab and field experiments from this funded research have provided us a stronger foundation to understand the response of Tidal Freshwater Marshes (TFMs) to climate change and salt water intrusion. The balance between marsh accretion and subsidence, and ultimately the ability of TFMs to outpace rising sea levels involves a complex interaction of the processes that drive plant production, microbial decomposition, sediment deposition and, ultimately, marsh accretion. The results of our work suggest that salt water intrusion will increase microbial decomposition and, together with declines in plant production, may put TFMs at risk of permanent inundation and create a positive feedback to the global C cycle.
Summary/Accomplishments (Outputs/Outcomes):
SITE SELECTION
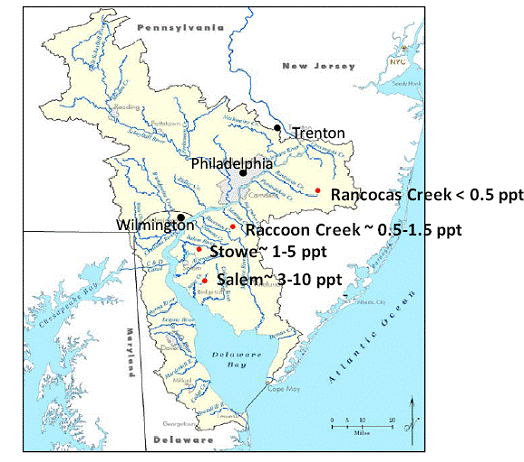
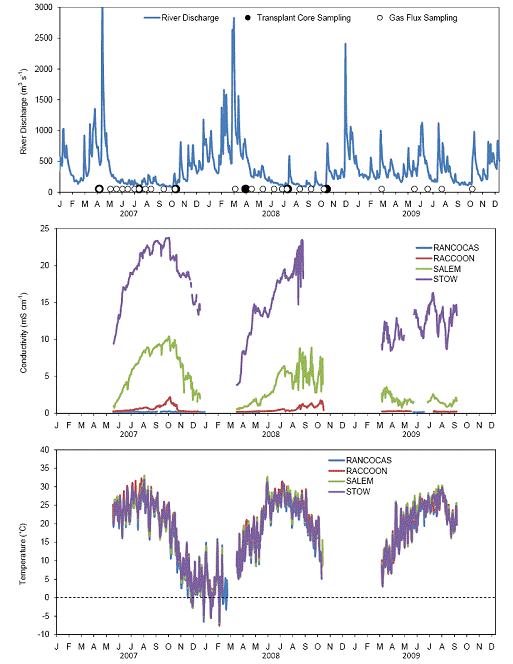
The Delaware River basin covers approximately 33,061 km2 in DE PA, NJ and NY, and is one of the most populated and ecologically important areas of the mid‐Atlantic region in the U.S. The Delaware River is the longest free‐flowing [un‐dammed] river east of the Mississippi, and extends 530 km from the confluence of its east and west branches in New York, and is a tidal estuary for 190 km before entering the Atlantic Ocean at the mouth of Delaware Bay. The Delaware River Basin is highly urbanized, especially the tidal portion. We have established four sites in the tidal portion of the Delaware River (Figure 1). Rancocas Creek is our freshwater end‐member receiving no salt (negligible conductivity; Figure 2) for the duration of the growing season and beyond (pore water analysis of cores collected from Rancocas have verified the lack of salt water intrusion to a depth of 25 cm, Raccoon Creek is largely fresh with salinities ranging from 0 to ~ 0.8 ppt (conductivity typically less than 2.0 mS cm‐1 even during the driest months of the year; Figure 2), and Salem in Mannington Meadows receives salinity in the range of 1‐5 ppt. Stow Creek is our salt water end‐member with salinities ranging from 5 ‐12 ppt, and highest conductivity (Figure 2). We have set up boardwalks at all four sites, permanent square collars for gas fluxing (0.5 m x 0.5 m; Figure 3 left), and transplanted cores with collars that measure (30 cm in diameter; Figure 3 right). The transplanted cores were collected from Rancocas in 2007, and transplanted to each of the four sites (back transplanted to Rancocas, Raccoon, Salem, and Stow).
ACCOMPLISHMENTS
LAB EXPERIMENT‐SUMMER 2007 & 2008
We completed a long‐term, salinity‐manipulation lab experiment that began in 2007. From both control and salinity‐amended cores, we have approximately 14 months of CO2 and CH4 flux measurements, depth integrated concentrations of chloride, sulfate, dissolved inorganic carbon (DIC), ammonium, dissolved organic carbon (DOC), acetate, sediment organic C and methane, and depth specific rates of sulfate reduction, hydrogenotrophic methanogenesis, and acetoclastic methanogenesis. The bulk of these data are in press in the peer‐reviewed journal, Biogeochemistry. Please see attached manuscript (Weston et al.) for figures and full interpretation. Major Findings from the lab experiment include: (1) Salt water intrusion into tidal freshwater marshes (TFM) can significantly increase rates of microbial C mineralization. (2) The total amount of C mineralized as CO2 and CH4 from salt - water amended cores was ~ 37% greater than freshwater amended cores over the one year duration of the experiment. (3) Salinity intrusion increased rates of both sulfate reduction and, surprisingly, methanogenesis, resulting in increased CO2 and CH4 emission (as the product of these decomposition processes) from the TFM sediments undergoing salinity intrusion. (4) This increase in organic matter decomposition and carbon gas emission indicates that the vertical accretion potential of TFM experiencing salinity intrusion may be decreased, with negative implications for the ability of TFM to keep pace with rising sea levels and feedbacks to the global C cycle.
FIELD EXPERIMENTS: PERMANENT PLOTS AND TRANSPLANTS
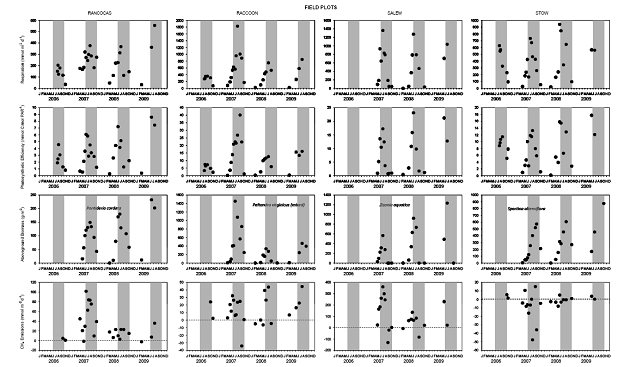
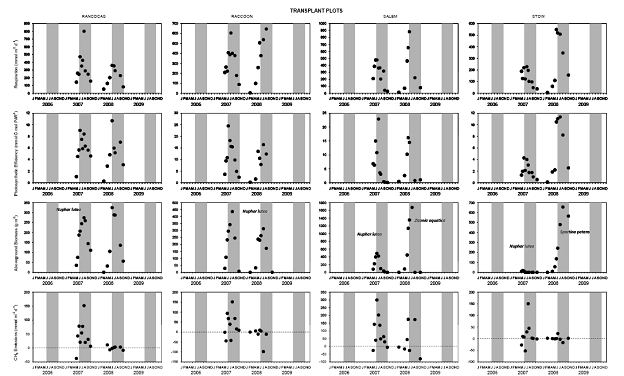
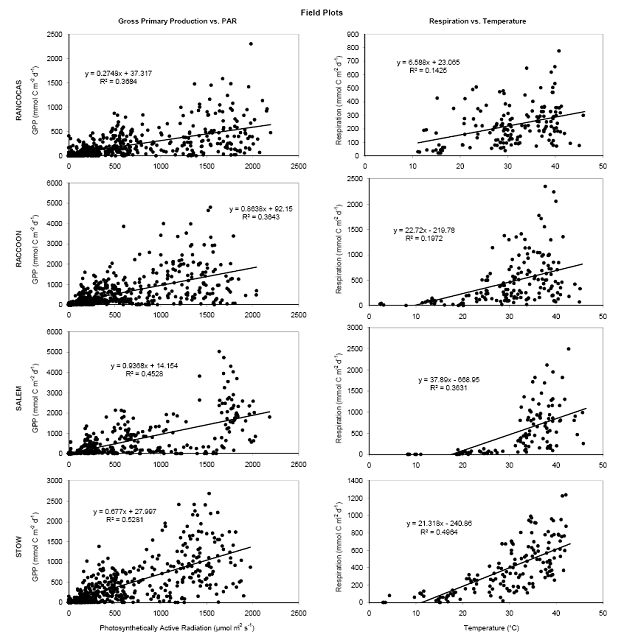
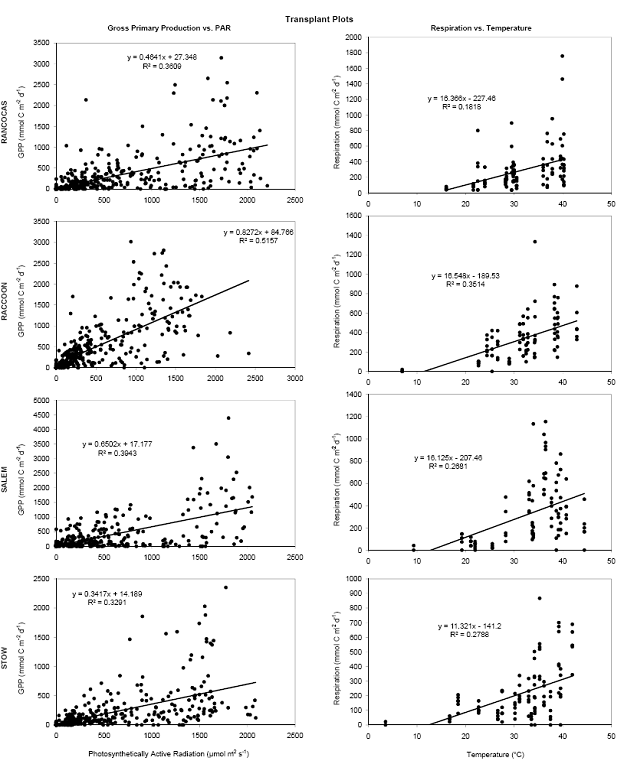
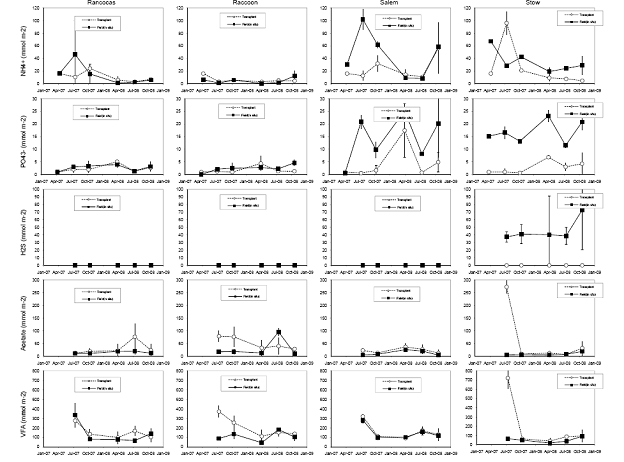
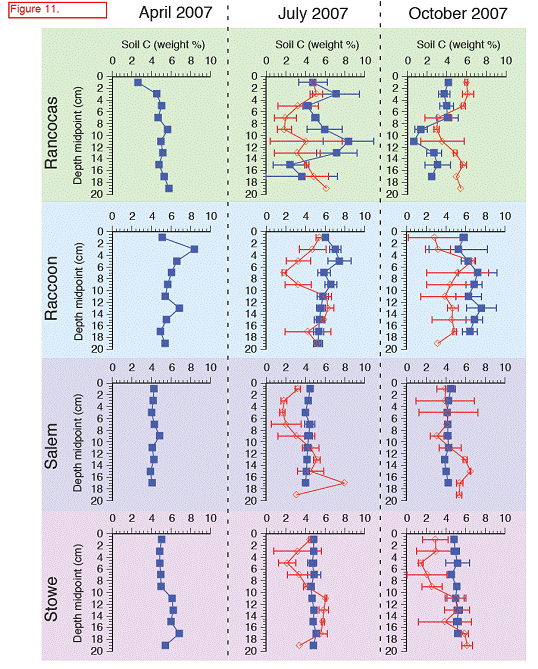
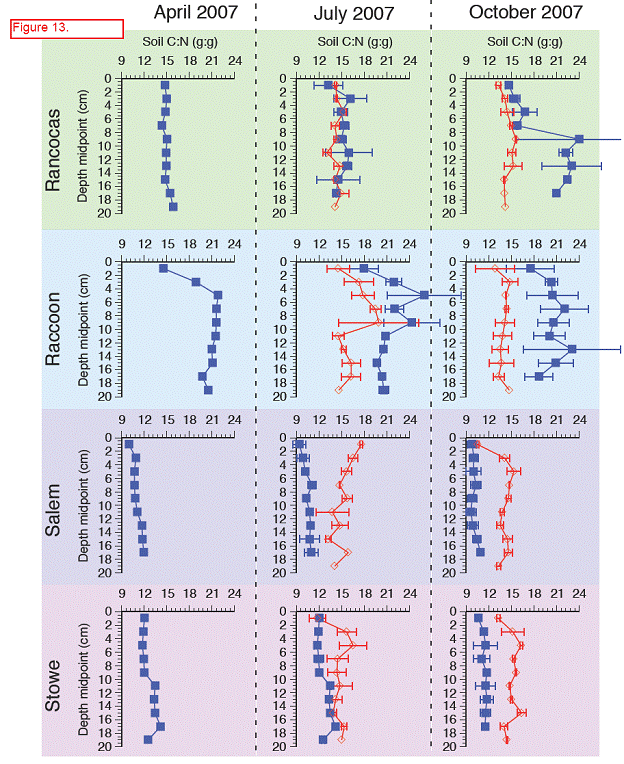
For both the permanent plots and transplanted cores we have three complete field seasons of biomass, photosynthetic efficiency, respiration rates, and methane fluxes (Figures 4‐7). These data sets constitute the bulk of the project, and will likely be published as two, perhaps three manuscripts. Not surprisingly, transplants behaved differently than permanent plots early on (2007), especially in terms of biomass, but by the second field season, the native plants had grown into the transplanted cores, which initially contained the dominant vegetation at Rancocas (Pontederia cordata), and behaved more like the host site, biologically and chemically (Figures 4‐7). By the second year post‐transplant, plant species reflected that of the host site and estimates were comparable to those in the permanent plots. Respiration rates, CH4 Flux rates, photosynthetic efficiency, and aboveground biomass were all highest at Raccoon and Salem in the permanent field plots (Figure 4). During the last field season, patterns in the transplant plots were similar to field plots in that respiration rates and photosynthetic efficiency were highest at Raccoon and Salem, while above ground biomass was greatest at Salem and Stow. Interestingly, CH4 fluxes were greatest at all sites in 2007, when first transplanted, but rates were typically low for all sites except Salem (Figure 5). Gross Primary Productivity (GPP) was greatest at Raccoon and Salem in the field plots while rates were similar for transplanted cores at all sites (Figure 6 & 7). Plants at Raccoon and Salem were more efficient photosynthetically (slope of relationship for GPP vs. PAR (photosynthetically active radiation) than the dominant vegetation at Raccoon and Stow in both field and transplant plots (Figures 4‐7). The C:N ratio was highest at Raccoon, due to greater %C throughout the profile (Figures 11‐14).
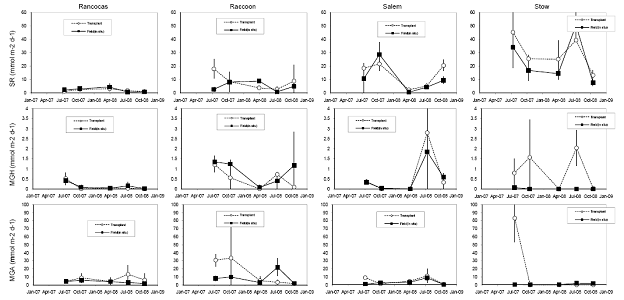
We also measured microbial rates of sulfate reduction and methanogenesis; rates of methane production through both the acetoclastic and hydrogenotrophic pathway also were measured (Figure 10). Rates of hydrogenotrophic methanogenesis are generally low, and acetoclastic methanogenesis is very low at Stow in situ, which is what we expected, but also supports the results we obtained in the lab experiment (see attached manuscript, Weston, et al., 2010 in press in Biogeochemistry). We expected to find rates of acetoclastic methanogenesis highest at Rancocas, our freshwater end‐member site, but rates were highest at Raccoon. Rates of acetoclastic methanogenesis may be lower at Rancocas because of site placement (plots were situated on bank instead of higher in the marsh) and type of soils at this site (lower organic C and higher mineral content; Figures 11‐14). Acetoclastic methanogenesis is higher in transplants at Raccoon (at 3 and 6 months) and Stow (at 3 months) than in field cores, with no significant difference at Rancocas, which we expected (Figure 10); rates were slightly higher at Salem at 3 months.
CARBON, NITROGEN AND PHOSPHORUS
CARBON
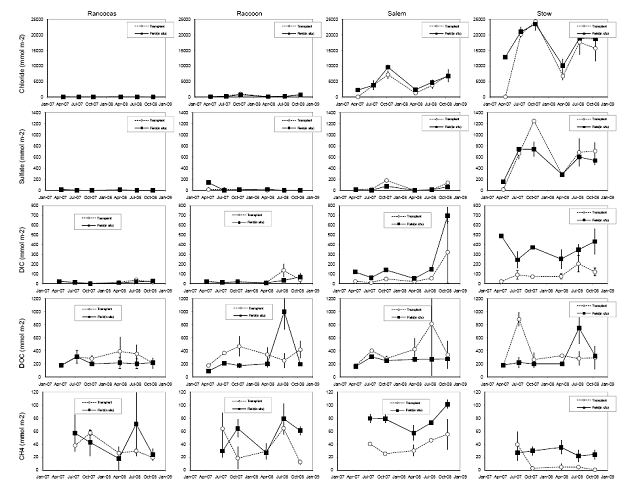
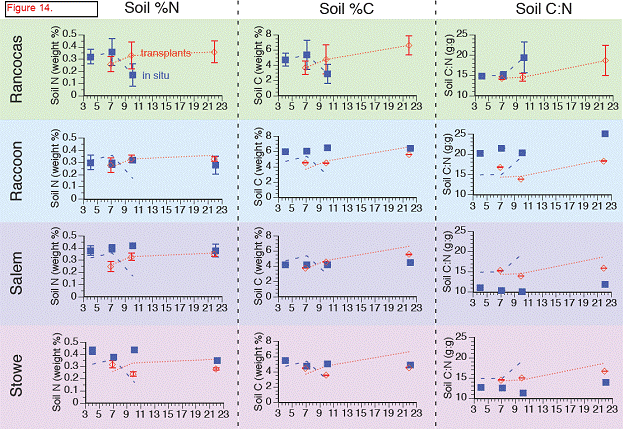
Carbon (C) varied between 2 and 8 % in cores taken in situ with lowest values at Rancocas, highest values at Raccoon, both are freshwater sites. The lower %C found at Rancocas is due to the higher mineral content found there relative to the other sites, and is likely due to where the cores were taken (creek bank versus higher elevation in the marsh) (Figure 11). Total %C did varied with depth at both Rancocas and Raccoon, but varied little with depth for Stow and Salem (Figure 11). Interestingly, when comparing cores in situ collected to cores collected from transplant plots, the transplanted cores showed lower % C, which supports higher rates of C metabolism (sulfate reduction, methanogenesis) measured in transplants, at least in the initial months following transplantation (Figure 10).
NITROGEN
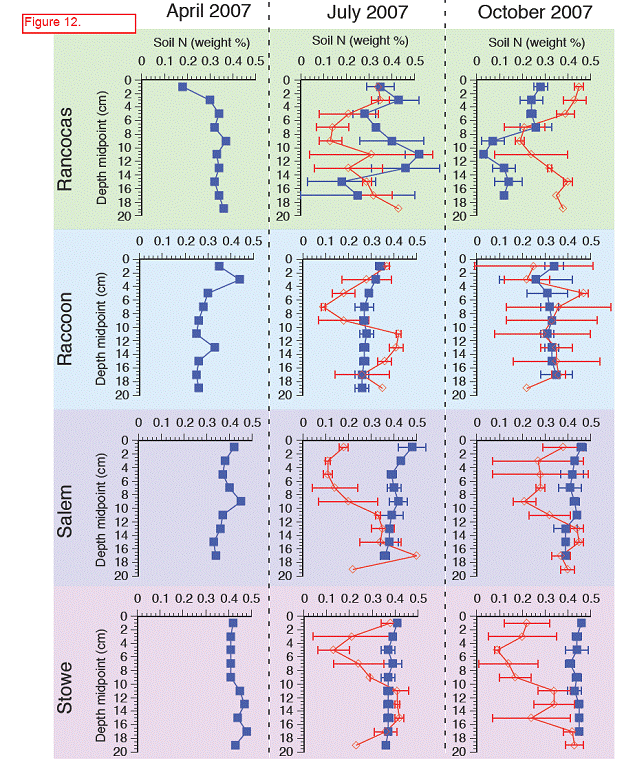
Nitrogen ranged from 0.1 to 0.5 % with higher %N found in the more saline sites (Salem and Stow), and as we saw with %C, variation with depth was seen primarily in the freshwater sites, Rancocas and Raccoon (Figure 12). %N in transplant cores tended to be lower when compared to in situ depth profiles (Figure 12). C:N ratios ranged from 9 to 21, with higher ratios found in the freshwater sites than the more saline sites (Figure 13). Interestingly, at the more saline sites, the C:N ratio was higher in the transplanted cores than cores collected in situ, while in the freshwater site Raccoon, C:N ratios were higher in cores collected in situ than in transplanted cores.
PHOSPHORUS
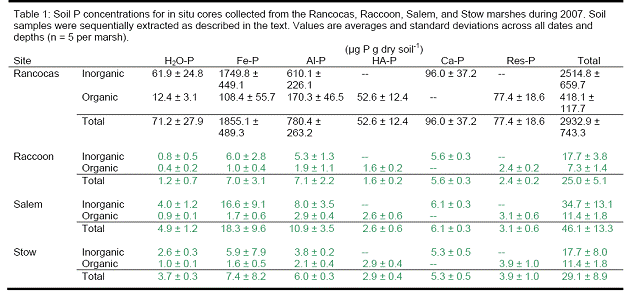
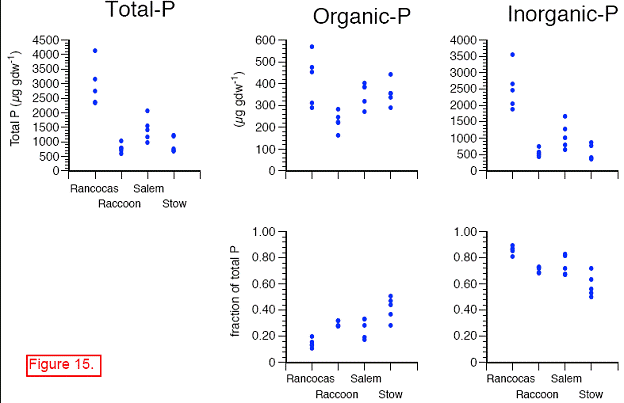
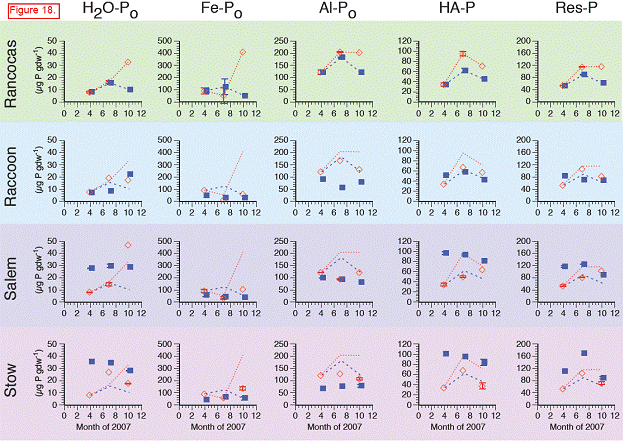
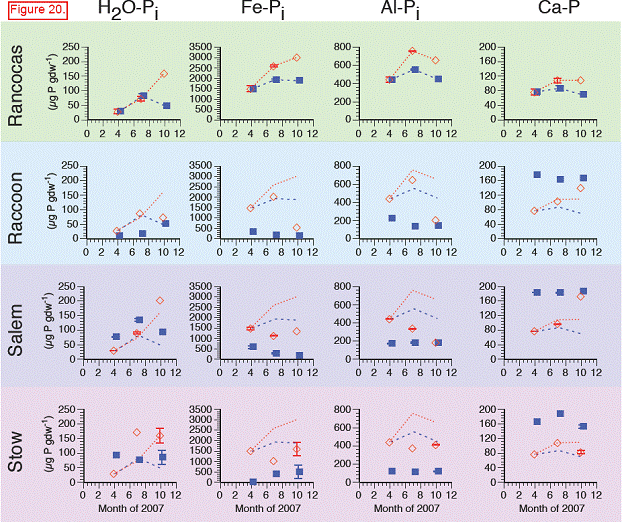
There were significant differences in the soil P pools from the marshes along the salinity gradient. Total soil P, calculated as the sum of the P fractions from the sequential extraction, ranged from 600 to 4100 μg P gdw‐1 across all sites, dates, and depths. Concentrations were highest at the freshwater end‐member site, Rancocas (Figures 15‐17). Concentrations at the three downstream sites were generally similar to each other but lower than at Rancocas. There were no repeatable changes in total P with depth or time across the four sites (data not shown).
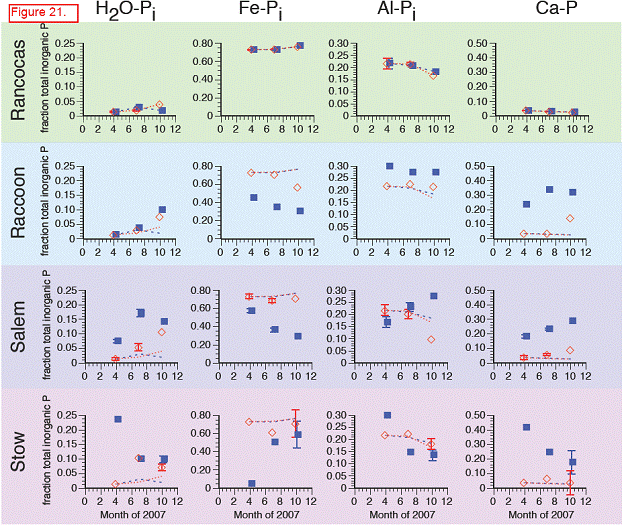
The trends in inorganic P (sum of H2O‐Pi, Fe‐Pi, Al‐Pi, and Ca‐P) paralleled those of total soil P, with concentrations highest at Rancocas and lower (but similar to each other) at Raccoon, Salem, and Stow. Inorganic P accounted for > 80 % of total P at Rancocas and decreased to ~60 % of total P at Stow (Figure). The concentrations of Fe‐Pi and Al‐Pi were highest at Rancocas, where Fe‐Pi accounted for 60‐70% of total inorganic P (vs. only 20‐50 % at the downstream sites, except for the April 2007 samples from Stow where Fe‐Pi was <5% of total Pi). The contribution of Al‐Pi to total Pi was ~10‐30% at all sites. Ca‐P had a similar contribution to total Pi, except at Rancocas where Ca‐P accounted for 3‐5% of total Pi. Water‐extractable inorganic P (H2O‐Pi) made the smallest contribution to soil inorganic P, accounting for 1‐3% of total Pi at Rancocas and increasing steadily to Stow (9‐24 % of total Pi).
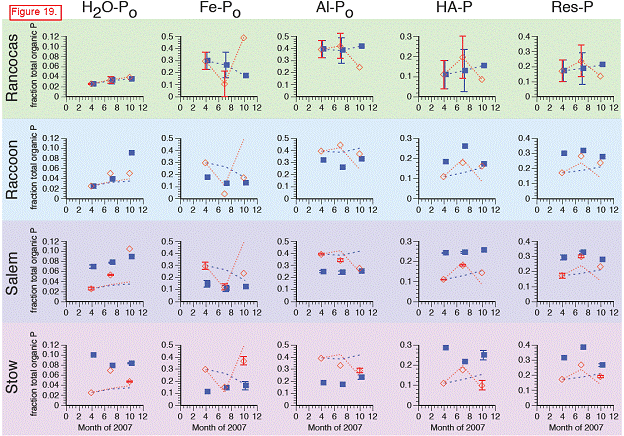
In contrast with total Pi, soil organic P concentrations (Po, sum of H2O‐Po, Fe‐Po, Al‐Po, HAP, and Res‐P) were generally similar at all four sites (Rancocas: 290‐570 μg gdw‐1; Raccoon: 160‐280 μg gdw‐1; Salem and Stow: 270‐440 μg gdw‐1). Across the salinity gradient, organic P accounted for 11‐19% of total soil P at Rancocas and increased to 28‐50 % at Stow. Organic P associated with metals (Fe‐Po and Al‐Po fractions) decreased in concentration and significance to the total organic P pool when moving downstream from Rancocas to Stow. In contrast, the concentrations and importance of the H2O‐Po, HA‐P, and Res‐P organic P fractions increased from Rancocas to Stow.
TRANSPLANTS:
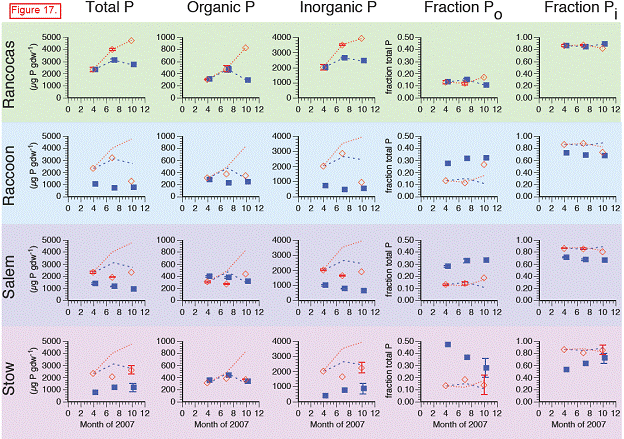
Total soil P: Cores transplanted to Raccoon, Salem, and Stow had lower total soil P concentrations in both July and October 2007 than did the donor marsh (Rancocas). At the three downstream sites, total soil P in the transplants was converging toward levels in the in situ soils at these sites. This appeared to be largely driven by decreases in inorganic P at all sites. There were also decreases in soil organic P in the Raccoon, Salem, and Stow transplants relative to the cores transplanted back into the donor marsh but the absolute magnitude of the decrease in organic P was less than the decreases in inorganic P (100‐500 μg P gdw‐1 vs. >1000‐2000 μg P gdw‐1)
ORGANIC P POOLS IN TRANSPLANTS:
H2O‐Po: concentrations rise throughout growing season in all transplanted cores … but not at any sites except Raccoon.
Fe‐Po: With the exception of the October 2007 data point at Rancocas, which was (anomalously) high, all transplants showed the same temporal trends in Fe‐Po concentrations, suggesting that Fe‐Po concentrations are not significantly influenced by salinity/sulfate.
Al‐Po: Concentrations of Al‐Po in the transplants at Raccoon, Salem, and Stow were generally lower than in the transplants at Rancocas.
HA‐P: Concentrations of HA‐P generally showed the same temporal patterns in all transplanted cores, regardless of site. Interestingly, concentrations of HA‐P in the transplanted cores at Raccoon, Salem, and Stow were generally lower than the cores transplanted at Rancocas, even though HA‐P concentrations in the in situ samples from these downstream marshes were higher than those from the donor marsh (i.e., HA‐P increased as salinity increased in the natural marshes but not in the transplants).
Res‐P: Very few changes in Res‐P in the transplants, regardless of site.
INORGANIC P POOLS IN TRANSPLANTS:
H2O‐Pi: Like H2O‐Po, concentrations of H2O‐Pi generally rose throughout the growing season in the transplants, regardless of site. H2O‐P may be more a function of soil type (see Sundareshwar and Morris, 1999).
Fe‐Pi: Relative to cores harvested and re‐transplanted at Rancocas, Fe‐Pi concentrations in the cores that were transplanted to Raccoon, Salem, and Stow were lower and closer to Fe‐Pi concentrations in the natural marshes adjacent to the transplanted cores. This may reflect interactions between the Fe, S, and P cycles that also contribute to lower Fe‐Pi concentrations in more saline sites.
Al‐Pi: Similar trends as Fe‐Pi. Some of this may be an ionic interaction since Al‐Pi fraction includes some P sorbed to clays. Additionally, it may reflect Fe‐S‐P interactions, although the Al‐P generally contains P associated with more‐recalcitrant Fe minerals (vs. more‐labile Fe minerals in the Fe‐Pi pool).
Ca‐P: At Raccoon and Salem, Ca‐P in the transplants was higher than in the transplants at the donor marsh, Rancocas. However, there were no real differences in Ca‐P between the donor marsh transplants and the transplants at Stow, the most saline site, even though Ca‐P in the in situ cores was roughly 2x higher at Stow than at Rancocas.
MICROBIAL COMMUNITY COMPOSITION‐LAB STUDY
The ability of marshes to keep pace with rising sea level depends upon accretion of C, and the accretion and decomposition of C is dependent on which microbes are dominant. This finding has important implications for microbial populations and what controls their abundance, population and community dynamics. To gain a mechanistic understanding of how and why the dominant microbial processes responded in the manner they did in the lab experiment described above (higher), we wanted to know how the community composition of sulfate reducing and methanogenic microbes responded to salinity intrusion. Weston incorporated a new component of the project (not initially proposed in the EPA grant) that links process‐based biogeochemical rates with quantitative determinations of key functional genes for sulfate reducers and methanogens. Key populations of anaerobic microbes mediating the oxidation of organic matter were targeted using functional gene primers: Sulfate Reducers (dissimilatory sulfite reductase, dsrAB), Methanogens (methyl co‐enzyme M reductase, mcrA), Denitrifiers (nitrite reductase, nir). Population sizes were determined using q‐PCR techniques, and community composition was determined by selective cloning and sequencing. Weston extracted DNA from freshwater and saline marsh sediments, functional genes were PCR amplified using functional gene‐specific primers, and dsrAB and mcrA products of appropriate size were obtained. Qualitatively, we found more mcrA functional gene products in freshwater sites and more dsrAB in saline sites. Preliminary data collected by Weston were promising enough to explore further. Over the summer of 2007 and 2008, Tanja Přsa, a senior thesis research student, with funding from the Biology Department added a molecular component to take advantage, and complement our biogeochemical process rate data to further understand the impact of salinity intrusion on C mineralization pathways in TFM. We examined the impacts of rising sea‐level on the structure and metabolic activity of SRB in TFM sediments undergoing salinity intrusion in the field transplant experiment described above. In the spring of 2007, we transplanted 6 intact sediment cores (30 cm diameter, 25 cm deep) from a TFM to the same TFM (Rancocas) and to a down‐estuary brackish marsh (Stow, salinity ~11 ppt). Sediment subcores were collected from transplants at both sites at the time of transplant (t=0) as well as 3 and 6 months post‐transplant (mid‐summer and early fall). Tanja Přsa has used this approach to examine the community composition in the laboratory experiment, and on the transplanted sediment cores. We determined rates of dissimilatory sulfate reduction and examined the community composition of SRB by targeting the dissimilatory sulfite reductase alpha subunit (dsrA) functional gene. We constructed dsrA clone libraries for both control and salinityimpacted sediments at 0, 3, 6 months post‐transplant and used phylogenetic analyses to determine changes in SRB community composition between the TFM and down‐estuary brackish marsh over time. Rates of sulfate reduction rates were significantly higher at Stow than Rancocas (35.6 ± 17.9 and 2.5 ± 0.9; mean ± std dev, respectively; p=0.0141, ANOVA). Phylogenetic analyses of sulfate reducing bacteria show that the community composition of sulfate reducing bacteria at Stow was significantly different from Rancocas 3 and 6 months post‐transplant (p < 0.05; ∫‐LIBSHUFF). These results suggest that salinity intrusion into TFMs will result in increased sulfate reduction rates and changes in microbial SRB populations. These changes will alter C dynamics in TFMs, potentially altering accretion rates and putting TFMs in jeopardy as sea levels rise.
Ms. Přsa has presented this work at the Society of Wetland Scientists meeting in Washington D.C. in May 2008, where she won honorable mention for best student poster. Ms. Přsa also presented her work at the Partnership for the Delaware River Estuary Science and Environmental Summit, held in Cape May, NJ in Janauary 2009; At this meeting, Tanja Přsa won an award for best student poster presentation, and was invited to submit an article to Estuary News, a publication of the Partnership for the Delaware River Estuary (see attached pdf of the newsletter or view the following link: http://www.delawareestuary.org/pdf/EstuaryNews/2010/Winter News10.pdf). Weston and Prsa plan to publish the results of this study in a peer reviewed journal later this fall.
We have several papers in various stages of publication. We expect a total of five peer‐reviewed publications from the funded work.
Figure 1. Field sites in along a salinity gradient in the DE Estuary.
Figure 2. Delaware River mean daily discharge at Trenton, NJ (top, data from USGS site 01463500), mean daily conductivity (middle) and air temperature (bottom) at the four field sites in the Delaware River Estuary.
Figure 3. Picture illustrating permanent square collars In the field (left) and transplanted cores (right).
Figure 4. Average rates of ecosystem respiration and photosynthetic efficiency, aboveground biomass and methane emissions over 3-4 years in permanent field plots at fours sites in the Delaware River Estuary. The dominant plant species is indicated in the biomass plots, and the growing season (June - September) is indicated by shaded regions.
Figure 5. Average rates of ecosystem respiration and photosynthetic efficiency, aboveground biomass and methane emissions over 2 years in tidal freshwater marsh plots transplanted from the Rancocas site to four sites in the Delaware River Estuary. The dominant plant species is indicated in the biomass plots, and the growing season (June - September) is indicated by shaded regions.
Figure 6. Relationship between gross primary production and photosynthetically active radiation (left) and between ecosystem respiration and temperature (right) in the permanent field plots at the four sites in the Delaware River Estuary over 4 years.
Figure 7. Relationship between gross primary production and photosynthetically active radiation (left) and between ecosystem respiration and temperature (right) in the transplanted plots at the four sites in the Delaware River Estuary over 4 years.
Figure 8. Soil porewater inventories (to a depth of 15 cm) of chloride, sulfate, dissolved inorganic carbon, dissolved organic carbon, and methane from four field sites in the Delaware River Estuary, and cores transplanted from the Rancocas tidal freshwater marsh site to the four field sites over a two-year period.
Figure 9. Soil porewater inventories (to a depth of 15 cm) of ammonium, phosphate, hydrogen sulfide, acetate, and total volatile fatty acids from four field sites in the Delaware River Estuary, and cores transplanted from the Rancocas tidal freshwater marsh site to the four field sites over a two-year period.
Figure 10. Rates of microbial sulfate reduction, hydrogenotrophic methanogenesis, and acetoclastic methanogenesis in soils (integrated to a depth of 15 cm) from four field sites in the Delaware River Estuary, and cores transplanted from the Rancocas tidal freshwater marsh site to the four field sites over a two-year period.
Values represent the average %N, %N, or C:N ratio in the top 20 cm, averaged over two replicate soil cores (except for April in situ cores where n = 1 core). Error bars are ± 1 standard deviation and indicate variability of each parameter with depth. Dashed lines show the values of each parameter from Rancocas, the donor marsh for the transplant cores.
Conclusions:
SIGNIFICANCE OF ACCOMPLISHMENTS
Tidal marshes are highly productive ecosystems that provide ecological services such as habitat for birds, fish and shellfish, storm‐surge buffering and water quality mitigation. The loss of coastal marshes can have devastating ecosystem‐level consequences, as previous hurricane events in the Gulf coast have demonstrated. Given that approximately 50% of the global population lives within coastal regions, both the socioeconomic and environmental impacts of sea‐level rise are far reaching. The response of TFM’s to rising sea levels is a complex interaction of the processes that drive plant production, microbial decomposition, sediment deposition, and marsh accretion. A greater understanding of ecosystem‐level responses of TFM to climate change is a major challenge that is of interest to scientists, land managers/planners, and increasingly, the general public. We have demonstrated how salinity‐intrusion impacts C accumulation in freshwater tidal marshes.
STAKEHOLDERS AND/OR USERS OF RESULTS/DATA/PRODUCTS
We have disseminated our findings through peer‐reviewed publications, conference presentations at national and international meetings, and invited seminars at various academic institutions in the greater Philadelphia area. Both Weston and Vile have worked on various outreach projects at the Academy of Natural Sciences in Philadelphia. Results of this research also have been disseminated to managers and stakeholders in the Delaware River and other coastal regions. Throughout this project, we have demonstrated a solid commitment to educating undergraduate students in the sciences through active involvement in the research. Additional undergraduate student involvement in this research was attained through Senior Thesis research at Villanova University, a RUI (Research at Undergraduate Institutions) institution. Additionally, this project supported an early‐career scientist during his transition from a postdoctoral fellow into a tenure track position in the Department of Geography and the Environment at Villanova University.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 20 publications | 3 publications in selected types | All 3 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Keams P, Weston NB, Bowen J, Zivkovic T, Vile M. Tidal Freshwater Marshes Harbor Phylogenetically Unique Clades of Sulfate Reducers That Are Resistant to Climate-Change-Induced Salinity Intrusion. ESTUARIES AND COASTS 2016;39(4):981-991. |
R832222 (Final) |
Exit Exit |
|
|
Watson N, Neubauer S, Velinsky D, Vile M. Net ecosystem carbon exchange and the greenhouse gas balance of tidal marshes along an estuarine salinity gradient. BIOGEOCHEMISTRY 2014;120(1-3):163-189. |
R832222 (Final) |
Exit Exit |
|
|
Weston NB, Vile MA, Neubauer SC, Velinsky DJ. Accelerated microbial organic matter mineralization following salt-water intrusion into tidal freshwater marsh soils. Biogeochemistry 2011;102(1-3):135-151. |
R832222 (Final) |
Exit Exit Exit |
Supplemental Keywords:
Ecosystem, aquatic, habitat, environmental chemistry, biology, geology, ecology, hydrology, genetics, limnology climate models, northeast, Atlantic coast, midatlantic, ecosystem scaling, metabolism, marine, estuary,, RFA, Air, Scientific Discipline, Ecosystem Protection/Environmental Exposure & Risk, ECOSYSTEMS, Aquatic Ecosystem, Aquatic Ecosystems & Estuarine Research, Ecological Risk Assessment, Terrestrial Ecosystems, Chemistry, Atmosphere, Monitoring/Modeling, Air Pollution Effects, climate change, Environmental Monitoring, Global Climate Change, climate model, global climate models, urbanization, global change, atmospheric chemistry, ecological models, coastal ecosystems, climate models, environmental measurement, climate variability, ecosystem indicators, phosphorus, environmental stress, ecosystem stress, tidal wetlands, wetlands, meteorology, salt water intrusion, aquatic ecosystems, biogeochemcial cycling, climatic influence, carbon supply, salinity stressProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.