Grantee Research Project Results
Final Report: Transformation of Halogenated PBTs with Nanoscale Bimetallic Particles
EPA Grant Number: GR832225Alternative EPA Grant Number: R832225
Title: Transformation of Halogenated PBTs with Nanoscale Bimetallic Particles
Investigators: Zhang, Wei-xian
Institution: Lehigh University
EPA Project Officer: Hahn, Intaek
Project Period: January 1, 2005 through December 31, 2007 (Extended to December 31, 2008)
Project Amount: $325,000
RFA: Exploratory Research to Anticipate Future Environmental Issues: Impacts of Manufactured Nanomaterials on Human Health and the Environment (2003) RFA Text | Recipients Lists
Research Category: Hazardous Waste/Remediation , Nanotechnology , Human Health , Safer Chemicals
Objective:
The goal of this research is to develop nanoscale bimetallic particles (e.g., Fe-Pd, Fe-Ni, Fe-Ag) with sizes in the range of 1-100 nm for treatment of hydrophobic, persistent, bioaccumulative toxic compounds (PBTs) such as polychlorinated biphenyls (PCBs), DDT and lindane.
Summary/Accomplishments (Outputs/Outcomes):
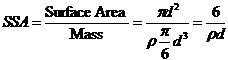 (2)
(2)
2.2 Mobility. Soil and aquifer media impose a filtration effect on groundwater passing through them, which impede the mobility of solid particles carried along by the water. Suspended particles in water are subject to various forces, namely, gravitational, electrostatic, magnetic, and random Brownian motion. For large particles (>> 1 µm), gravity force predominates, resulting in rapid sedimentation and limited transverse mobility. On the other hand, the movement of nano-scale particles is largely governed by random Brownian motion. Constant collisions with water molecules cause arbitrary motion of very fine nanoparticles in water. Attractive electrostatic and magnetic forces (as in the case of iron) are the two main causes contributing to nanoparticle aggregation. Aggregation can be effectively mitigated by controlling the solution pH, ionic strength, and particle size and through the use of stabilizing agents. Laboratory studies have shown that iron nanoparticles stabilized by adding a small amount of naturally occurring polymers during synthesis processes can remain in suspension for extended periods of time (> 6 months) and migrate efficiently through sand-packed columns. The relatively high ionic strength typically found in groundwater also acts to suppress surface electrostatic charges and aids in stable dispersion of the nanoparticles.
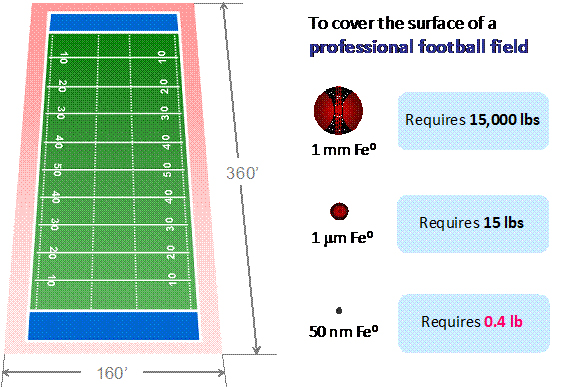
| | 1 mm | ~10 µm | 50 nm |
| Price (dollars/kg) | 3.5 | 22 | 50 |
| Surface area (m2/kg) | 0.77 | 76.92 | 30,000 |
| Unit cost (m2/dollar) | 0.22 | 3.50 | 600 |
Conclusions:
Results from this project have been widely disseminated, including more than 30 peer-reviewed papers and 40 invited presentations and seminars. Key aspects of this research are summarized in this final report.
Journal Articles on this Report : 28 Displayed | Download in RIS Format
| Other project views: | All 68 publications | 28 publications in selected types | All 28 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Cao J, Clasen P, Zhang W-X. Nanoporous zero-valent iron. Journal of Materials Research 2005;20(12):3238-3243. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (Final) |
Exit |
|
|
Cao J, Elliott D, Zhang W-X. Perchlorate reduction by nanoscale iron particles. Journal of Nanoparticle Research 2005;7(4-5):499-506. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (2003) R829625 (Final) |
Exit Exit |
|
|
Cao J, Zhang W-X. Stabilization of chromium ore processing residue (COPR) with nanoscale iron particles. Journal of Hazardous Materials 2006;132(2-3):213-219. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (Final) |
Exit Exit Exit |
|
|
Cao J, Zhang W-X, Brown DG, Sethi D. Oxidation of lindane with Fe(II)-activated sodium persulfate. Environmental Engineering Science 2008;25(2):221-228. |
GR832225 (Final) |
Exit |
|
|
Cao J, Li X, Tavakoli J, Zhang W-X. Temperature programmed reduction for measurement of oxygen content in nanoscale zero-valent iron. Environmental Science & Technology 2008;42(10):3780-3785. |
GR832225 (Final) |
Exit |
|
|
Cheng R, Wang J-L, Zhang W-X. Comparison of reductive dechlorination of p-chlorophenol using Fe0 and nanosized Fe0. Journal of Hazardous Materials 2007;144(1-2):334-339. |
GR832225 (Final) |
Exit Exit Exit |
|
|
Elliott DW, Lien H-L, Zhang W-X. Zerovalent iron nanoparticles for treatment of ground water contaminated by hexachlorocyclohexanes. Journal of Environmental Quality 2008;37(6):2192-2201. |
GR832225 (Final) |
Exit Exit |
|
|
Elliott DW, Lien H-L, Zhang W-X. Degradation of lindane by zero-valent iron nanoparticles. Journal of Environmental Engineering-ASCE 2009;135(5):317-324. |
GR832225 (Final) |
Exit Exit |
|
|
Li S, Elliott DW, Spear ST, Ma LM, Zhang W-X. Hexachlorocyclohexanes in the environment: mechanisms of dechlorination. Critical Reviews in Environmental Science and Technology 2011;41(19):1747-1792. |
GR832225 (Final) |
Exit |
|
|
Li S, Yan W, Zhang W-X. Solvent-free production of nanoscale zero-valent iron (nZVI) with precision milling. Green Chemistry 2009;11(10):1618-1626. |
GR832225 (Final) |
Exit |
|
|
Li X-Q, Zhang W-X. Iron nanoparticles: the core-shell structure and unique properties for Ni(II) sequestration. Langmuir 2006;22(10):4638-4642. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (Final) |
Exit |
|
|
Li X-Q, Elliott DW, Zhang W-X. Zero-valent iron nanoparticles for abatement of environmental pollutants: materials and engineering aspects. Critical Reviews in Solid State and Materials Sciences 2006;31(4):111-122. |
GR832225 (2006) GR832225 (Final) |
Exit Exit |
|
|
Li X-Q, Brown DG, Zhang W-X. Stabilization of biosolids with nanoscale zero-valent iron (nZVI). Journal of Nanoparticle Research 2007;9(2):233-243. |
GR832225 (2006) GR832225 (Final) |
Exit Exit |
|
|
Li X-Q, Zhang W-X. Sequestration of metal cations with zerovalent iron nanoparticles – a study with high resolution X-ray photoelectron spectroscopy (HR-XPS). Journal of Physical Chemistry C 2007;111(19):6939-6946. |
GR832225 (2006) GR832225 (Final) |
Exit |
|
|
Li X-Q, Cao J, Zhang W-X. Stoichiometry of Cr(VI) immobilization using nanoscale zerovalent iron (nZVI): a study with high-resolution X-ray photoelectron spectroscopy (HR-XPS). Industrial & Engineering Chemistry Research 2008;47(7):2131-2139. |
GR832225 (Final) |
Exit |
|
|
Lien H-L, Zhang W-X. Hydrodechlorination of chlorinated ethanes by nanoscale Pd/Fe bimetallic particles. Journal of Environmental Engineering-ASCE 2005;131(1):4-10. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (2003) R829625 (Final) |
Exit Exit |
|
|
Lien H-L, Zhang W-X. Removal of methyl tert-butyl ether (MTBE) with Nafion. Journal of Hazardous Materials 2007;144(1-2):194-199. |
GR832225 (2006) GR832225 (Final) |
Exit Exit Exit |
|
|
Lien H-L, Zhang W-X. Nanoscale Pd/Fe bimetallic particles: catalytic effects of palladium on hydrodechlorination. Applied Catalysis B: Environmental 2007;77(1-2):110-116. |
GR832225 (Final) |
Exit Exit |
|
|
Lien H-L, Elliott DW, Sun Y-P, Zhang W-X. Recent progress in zero-valent iron nanoparticles for groundwater remediation. Journal of Environmental Engineering and Management 2006;16(6):371-380. |
GR832225 (2006) GR832225 (Final) |
Exit Exit |
|
|
Ma L, Zhang W-X. Enhanced biological treatment of industrial wastewater with bimetallic zero-valent iron. Environmental Science & Technology 2008;42(15):5384-5389. |
GR832225 (Final) |
Exit Exit |
|
|
Mace C, Desrocher S, Gheorghiu F, Kane A, Pupeza M, Cernik M, Kvapil P, Venkatakrishnan R, Zhang W-X. Nanotechnology and groundwater remediation: a step forward in technology understanding. Remediation 2006;16(2):23-33. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (Final) |
Exit |
|
|
Martin JE, Herzing AA, Yan W, Li X-Q, Koel BE, Kiely CJ, Zhang W-X. Determination of the oxide layer thickness in core-shell zerovalent iron nanoparticles. Langmuir 2008;24(8):4329-4334. |
GR832225 (Final) |
Exit |
|
|
Ramos MAV, Yan W, Li X-Q, Koel BE, Zhang W-X. Simultaneous oxidation and reduction of arsenic by zero-valent iron nanoparticles: understanding the significance of the core-shell structure. Journal of Physical Chemistry C 2009;113(33):14591-14594. |
GR832225 (Final) |
Exit Exit |
|
|
Sun Y-P, Li X-Q, Zhang W-X, Wang HP. A method for the preparation of stable dispersion of zero-valent iron nanoparticles. Colloids and Surfaces A: Physicochemical and Engineering Aspects 2007;308(1-3):60-66. |
GR832225 (2006) GR832225 (Final) |
Exit |
|
|
Sun Y-P, Li X-Q, Cao JS, Zhang W-X, Wang HP. Characterization of zero-valent iron nanoparticles. Advances in Colloid and Interface Science 2006;120(1-3):47-56. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (Final) |
Exit Exit |
|
|
Yan W, Herzing AA, Li X-Q, Kiely CJ, Zhang W-X. Structural evolution of Pd-doped nanoscale zero-valent iron (nZVI) in aqueous media and implications for particle aging and reactivity. Environmental Science & Technology 2010;44(11):4288-4294. |
GR832225 (Final) |
Exit |
|
|
Zhang W-X, Karn B. Nanoscale environmental science and technology: challenges and opportunities. Environmental Science & Technology 2005;39(5):94A-95A. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (Final) |
Exit Exit |
|
|
Zhang W-X, Elliott DW. Applications of iron nanoparticles for groundwater remediation. Remediation 2006;16(2):7-21. |
GR832225 (2005) GR832225 (2006) GR832225 (Final) R829625 (Final) |
Exit |
Supplemental Keywords:
ground water, nanoparticles, nanotechnology, organics, PBTs, pesticides, remediation, soil, sediments;, Sustainable Industry/Business, RFA, Scientific Discipline, Waste, Remediation, Technology for Sustainable Environment, Sustainable Environment, Environmental Chemistry, Biochemistry, New/Innovative technologies, Environmental Engineering, sustainability, nanoparticle remediation, biotechnology, environmental sustainability, nanocatalysts, innovative technologies, bio-engineering, nanoscale bimetallic particles, remediation technologies, environmentally applicable nanoparticles, biodegradation, nanotechnology, decontamination, bioremediation, bioengineering, nanoparticle based remediationProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
