Grantee Research Project Results
Final Report: Nanostructured Membranes for Filtration, Disinfection, and Remediation of Aqueous and Gaseous Systems
EPA Grant Number: GR832372Title: Nanostructured Membranes for Filtration, Disinfection, and Remediation of Aqueous and Gaseous Systems
Investigators: Kit, Kevin , Davidson, P. Michael , Weiss, Jochen , Zivanovic, Svetlana
Institution: University of Tennessee , University of Massachusetts - Amherst
EPA Project Officer: Hahn, Intaek
Project Period: August 1, 2005 through July 31, 2008 (Extended to July 31, 2009)
Project Amount: $349,200
RFA: Greater Research Opportunities: Research in Nanoscale Science Engineering and Technology (2004) RFA Text | Recipients Lists
Research Category: Hazardous Waste/Remediation , Nanotechnology , Safer Chemicals
Objective:
The objective of this project was to develop electrospun nanofiber chitosan membranes that will have the ability to treat aqueous and gaseous environments by actions of filtration, disinfection, and metal binding. Chitosan is nontoxic and biodegradable, and has been shown to have beneficial antimicrobial and metal binding properties. These beneficial properties will be optimized in a nanofiber structure in which the surface area per mass is very high. The central hypothesis for the proposed research is that the degree to which these nanofiber chitosan membranes effectively filter contaminants, kill microbes, and bind harmful metals will be optimized by minimizing the size of the electrospun fibers and maximizing the available chitosan surface area. The project was originally divided into the following four tasks:
- Determine processing-structure relationships for electrospun chitosan
- Measure filtration properties of electrospun chitosan
- Measure metal binding and physicochemical properties of electrospun chitosan
- Measure antimicrobial properties of electrospun chitosan
However, we found it more logical to divide the project accordingly:
- Determine processing-structure relationships for electrospun chitosan
- Measure metal binding and antimicrobial properties in static conditions
- Measure metal binding, antimicrobial, and filtration properties under dynamic flow conditions
In this way, we were able to determine the most effective fiber structures for metal binding and antimicrobial performance under near equilibrium conditions and then select the most effective for additional dynamic filtration studies.
Summary/Accomplishments (Outputs/Outcomes):
Materials
Electrospinning
Effect of Solvents and Spinning Solution temperature

|
Type of Chitosan (Molecular weight)
|
Solvent
|
Spinning Solution Temperature (°C)
|
Polymer Concentration
|
|
HMW Chitosan
(Mv - 1400 kDa)
|
0.03N HCl
|
25, 40, 70
|
0.6 – 1.5 wt%
|
|
0.1N HCl
|
25, 40, 70
|
0.1 - 2 wt%
|
|
|
0.5N HCl
|
25, 40, 70
|
1.5 wt%
|
|
|
50% TFA
|
25, 40, 70
|
1.5 wt%
|
|
|
90% AA
|
25, 40, 70
|
1.2 wt% + 1.5 wt% Urea
|
|
|
90% AA
|
25, 40, 70
|
1.5 wt%
|
|
|
LMW Chitosan
(Mv - 100 kDa)
|
0.1N HCl
|
25, 40, 70
|
1.7 wt%
|
|
90% AA
|
25, 40, 70
|
5 wt% with addition of salt
|
|
|
30% AA
|
25, 40, 70
|
6 wt% with addition of salt
|
|
|
Hydrolyzed Chitosan
(Mv – 300kDa)
|
80% AA
|
25
|
5 wt %
|
|
Hydrolyzed Chitosan
(Mv – 80kDa)
|
90% AA
|
25
|
4 wt%
|
|
Hydrolyzed Chitosan
(Mv – 20kDa)
|
80% AA
|
25
|
5 wt %, 6 wt%
|
Electrospinning of Chitosan/PEO blends

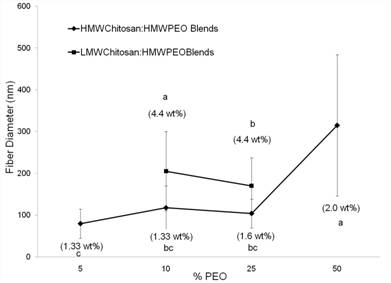
Effect of Spinning Solution Temperature

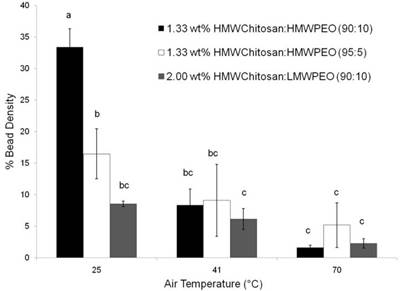

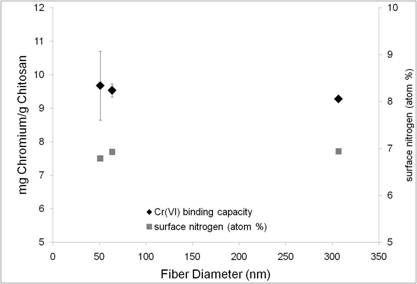
XPS Results
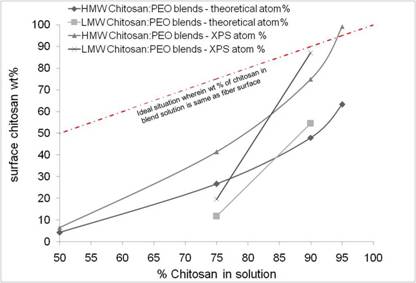
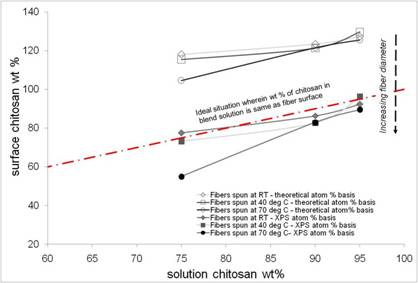
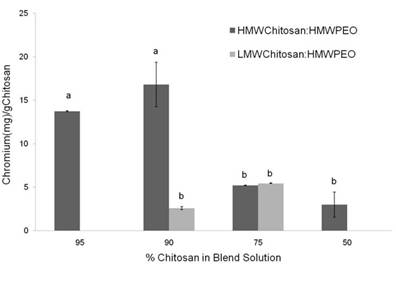
Metal binding - Chitosan/PEO Blends
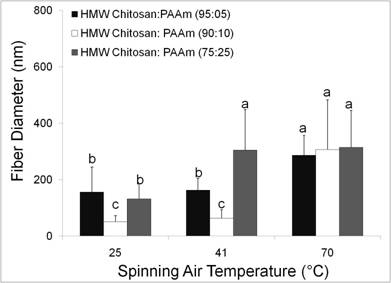
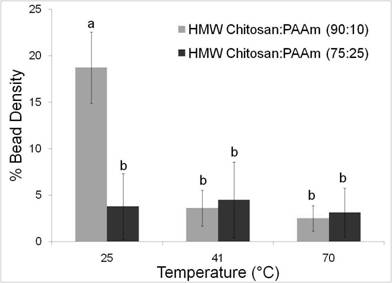
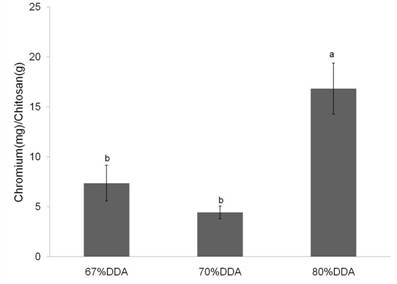
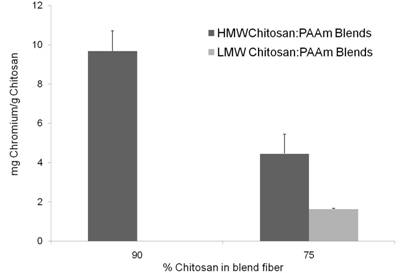
Metal Binding - Chitosan/PAAm Blends
Anti-microbial Properties of Chitosan/PEO Blends
References:
Journal Articles:
No journal articles submitted with this report: View all 13 publications for this projectSupplemental Keywords:
Sustainable Industry/Business, RFA, Scientific Discipline, Waste, Water, Remediation, Technology for Sustainable Environment, Sustainable Environment, Engineering, Chemistry, & Physics, Chemistry and Materials Science, New/Innovative technologies, Physics, Environmental Engineering, metal removal, detoxification, membrane technology, membranes, ultrafiltration, pollution prevention, aquifer remediation design, innovative technologies, industrial wastewater, environmental sustainability, nanocatalysts, contaminated aquifers, groundwater contamination, remediation technologies, antimicrobial nanostructured membranes, environmentally applicable nanoparticles, transition metal carbides, groundwater remediation, nanotechnology, in situ remediation, electrospun nanofiber chitosan membranes, membrane-based nanostructured metals, environmental chemistry, disinfection, metal bindingProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
