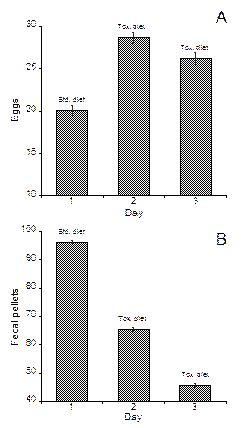Grantee Research Project Results
Final Report: Linking Food Web Structure, Grazer Toxin Resistance and Ecological Stoichiometry in Understanding Harmful Algal Blooms
EPA Grant Number: R831706Title: Linking Food Web Structure, Grazer Toxin Resistance and Ecological Stoichiometry in Understanding Harmful Algal Blooms
Investigators: Dam, Hans G. , McManus, George , Kremer, Patricia
Institution: University of Connecticut
EPA Project Officer: Packard, Benjamin H
Project Period: January 1, 2005 through December 31, 2007 (Extended to December 31, 2008)
Project Amount: $408,315
RFA: Ecology and Oceanography of Harmful Algal Blooms (2004) RFA Text | Recipients Lists
Research Category: Aquatic Ecosystems , Water , Environmental Statistics
Objective:
Planktonic trophic interactions are characterized by negative interactions, which involve a reduction in the number, or the growth rate of the prey and arise from herbivory (animals eating plants) and predation (animals eating other animals). However, when plants, herbivores and predators are all present, positive (indirect) interactions are possible. For instance, predators negatively impact herbivores, which in turn results in an increase in plants. Thus, there is an indirect and positive effect of predators on plants. Such effect when removal or addition of a trophic group affects changes at lower trophic levels constitutes a trophic cascade. The main goal of this project was to examine the effects of three feedback factors seldom considered in tandem in the expression of planktonic trophic cascades involving toxic algae: (1) The toxicity of the algae, 2) toxin resistance of grazer populations, and 3) the elemental stoichiometric (C:N:P) imbalance between algae and grazers. The original hypotheses of the study were: 1) trophic cascades are stronger in the presence of toxic algae; 2) trophic cascades are weaker in the presence of toxin-resistant grazer populations; and 3) the strength of trophic cascades depends on the interaction of the stoichiometric imbalance of the grazers, the toxicity of the algae and the complexity of the foodweb.
Summary/Accomplishments (Outputs/Outcomes):
Our work focused on one the red tide dinoflagellate, the genus Alexandrium. Some species in this genus produce saxitoxins, a potent suite of neurotoxins that block sodium channels. Our original intention was to create model food web models involving four trophic levels: Ctenophores, copepods, microzooplankton, and phytoplankton (including toxic Alexandrium). Only a few experiments were carried out involving ctenophores, as scaling constraints for the experiments are considerable. Therefore, most of the experiments were limited to three food levels. Further, before we could run full-fledged experiments to test the specific hypotheses of this study, we had to do considerable background work to test some of the implicit assumptions upon which the objectives of the research were based. Here we summarize the main findings:
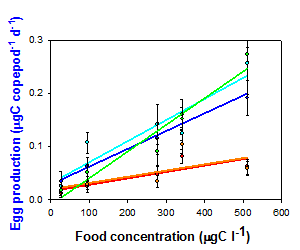
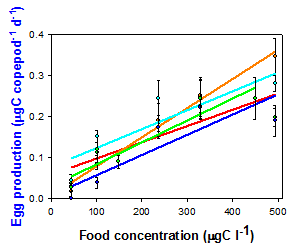
Degree of grazer population resistance: There were dramatic differences in the response of ingestion and egg production rates versus food concentration in copepod populations that were historically exposed (putatively resistant or tolerant to saxitoxin) and those with little or no exposure (less resistant or tolerant to saxitoxin). (Fig. 1). The implication is that toxin resistance has evolved in some populations. This was a necessary condition for testing hypothesis 2.
Figure 1. Differences in the egg production rates of populations of the copepod Acartia hudsonica from areas historically exposed to blooms of the toxic dinoflagellate Alexandrium (New Brunswick, Canada (NB), Maine (ME) and Massachusetts (MA)) and areas of little or no exposure (Connecticut (CT) and New Jersey (NJ)). Shown are responses when food was the toxic dinoflagellate (left) and a control, non-toxic flagellate (right). From Colin and Dam (2007).
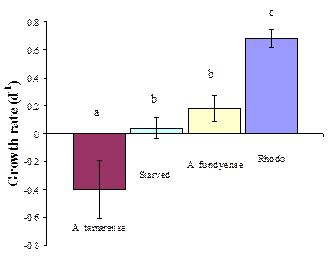
Microzooplankton: We documented how toxic Alexandrium would affect microzooplankton, the major group of grazers in the oceans. Experiments were carried out with the ciliate Strombidinopsis sp. fed two members of the Alexandrium fundyense complex, A. fundyense and A. tamarense. A. tamarense is considered to be non-toxic or less toxic than A. fundyense because it does not produce saxitoxin (STX). While both dinoflagellates make other STX-related compounds, A. tamarense produces less of these than A. fundyense. The ciliate fed on both species of Alexandrium. It survived, but did not grow on A. fundyense; however, there was significant mortality in ciliates fed A. tamarense (Fig. 2). . Behavioral assays showed unexpected differences in reactions of ciliates to A. fundyense and A. tamarense extracts, with avoidance (backwards swimming) induced by fresh A. tamarense extract but not A. fundyense and backwards swimming was induced by both extract when aged. Hence, toxicity to microzooplankton does not seem to be a function of saxitoxin content alone. This finding complicates interpretation of the designed trophic cascade experiments, but more importantly, it points to a new mechanism of toxicity in the microzooplankton. This past fall, we started a new line of research to elucidate the nature of this mechanism. We now hypothesize that Alexandrium toxicity in the microzooplankton is related to reactive oxygen compounds in Alexandrium (Skelton et al., submitted).
Figure 2. Growth rate versus food type in 45 h assays. Rhodo= Rhodomonas, a nontoxic food. From Schoenert et al. (2007).
Discovery of copepod reproductive phenotypes: Our experimental design called for the use of toxin resistant/tolerant populations in trophic cascade experiments. Our work with individuals led to an important discovery. We documented for the first time toxin-related reproductive phenotypes of copepods, and a novel procedure to identify these phenotypes (Avery and Dam 2007). During this study, we discovered five resistance-related reproductive and calculated the fitness of each phenotype on diets with and without toxic Alexandrium. From these results, we argue that resistance is conferred by a simple genetic system showing heterozygote advantage. A polymorphism for resistance will prevent the fixation of resistance alleles in natural populations. It may also confound the interpretation of typical experiments that measure average population responses, which complicates interpretation of cascade experiments.
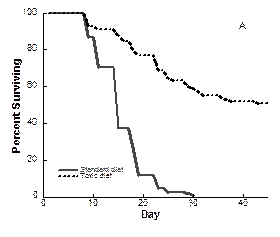
Positive effects of toxin on copepods: We also discovered a novel response that will help us to understand the role of grazing in the fate of toxic blooms. In laboratory experiments copepods responded to toxic Alexandrium sp. by increasing survivorship and egg production (Fig. 3). Neither food quality nor food quantity could account for the responses. We propose a physiological mechanism by which ingested toxins improve assimilation efficiency—by increasing the residence time of food in the copepod gut. Such positive effects, which are currently unappreciated, will affect grazing feedbacks and therefore, the fate of harmful algal blooms (HABs). Potential positive effects, which are novel responses to paralytic shellfish poisoning toxins, should be considered when evaluating the co-evolution of toxic prey and their consumers.
Figure. 3. Survivorship (left) and egg production and fecal pellet production (right) in the copepod Acartia hudsonica exposed to toxic Alexandrium and a nontoxic diet. From Avery et al. (2008).
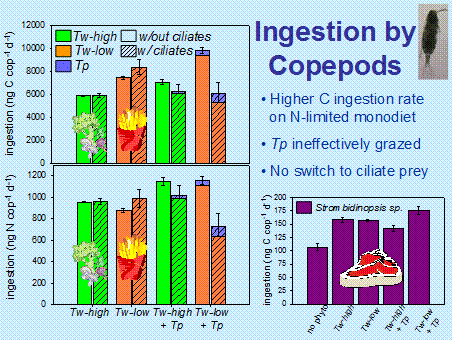
Effects of omnivory and stoichiometric imbalance on trophic interactions: One aspect of food web complexity, omnivory, is predicted to dampen trophic cascades, whereas stoichiometric imbalance between consumers and producers should strengthen them. We created experimental model food webs to examine the combined effects of complexity and stoichiometric imbalance on estuarine trophic cascades (Siuda and Dam, submitted). A copepod, Acartia tonsa, and an aloricate ciliate, Strombidinopsis sp., served as grazers on large and small centric diatoms, Thalassiosira weissflogii and Thalassiosira pseudonana, respectively. T. weissflogii cultures were grown under nitrogen-replete and nitrogen-limited conditions, which resulted in a three-fold difference in cell nitrogen content. Relative to the nitrogen-replete culture, carbon ingestion rates increased by 25% for A. tonsa and 160% for Strombidinopsis sp. fed solely on nitrogen-limited T. weissflogii (Fig. 4). However, when T. pseudonana was added to the diet of Strombidinopsis sp., the ingestion rate for T. weissflogii significantly decreased and was independent of T. weissflogii nitrogen content. A. tonsa grazed poorly on T. pseudonana, and ingestion of both nutrient replete and limited T. weissflogii increased on a mixed phytoplankton diet. For A. tonsa, the presence of the ciliate did not significantly change ingestion of T. weissflogii, regardless of phytoplankton nitrogen content. Additionally, the ingestion of ciliates by A. tonsa remained constant and proportionally small relative to phytoplankton carbon ingestion rates in all treatments. In the present study, the effects of omnivory on trophic interactions were stronger than those of stoichiometric imbalance. Similar effects were observed in a field study to address omnivory versus stoichiometric imbalance effects (Siuda and Dam, in prep.). Thus, the apparently stronger effect of omnivory implies that cascading trophic effects will not be the norm in planktonic communities, at least in coastal areas where HAB predominate.
Figure 4. Left panels: Copepod ingestion rate of phytoplankton versus phytoplankton with varying nitrogen content in the presence or absence of alternate food, ciliate. Right panel: Copepod ingestion rate of ciliate in the presence or absence of phytoplankton with varying nitrogen content. High and low nitrogen content in phytoplankton is denoted, respectively, by the vegetable and French fry icons. The higher nitrogen content of the ciliate is denoted by the steak icon. From Siuda and Dam (submitted).
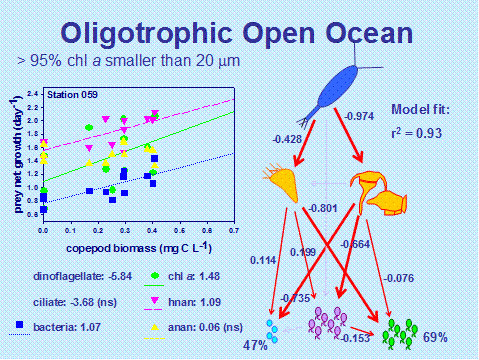
Trophic cascades in natural communities: We conducted nine experiments, along a north to south transect in the western North Atlantic (Woods Hole, Massachusetts, USA to Scarborough, Tobago), to examine planktonic trophic cascades in contrasting pelagic ecosystems (Siuda and Dam, submitted). We summarized direct and indirect food web interactions in each region with models derived from path analysis, a technique that integrates correlation and multiple regression analysis. Copepods directly consumed dinoflagellates and ciliates, which resulted in positive effects on nanoplankton and bacteria. Total phytoplankton as chlorophyll a was grazed directly by copepods in New England shelf waters, but indirect (positive) effects were observed in all other regions . Indirect effects of copepod grazing were strongest in the Amazon/Orinoco plume, where 51% of the variance in bacterial growth and 81% of the variance in heterotrophic nanoplankton growth, respectively, was explained through linkages to copepods via ciliates. In the Sargasso Sea, indirect effects on bacteria via microplankton were weaker, but still evident (Fig. 5). The main conclusion from this exercise is that while the complexity of food web structure superficially conceals cascading interactions, top-down effects on the lower food web can be detected if the appropriate statistical approach is employed.
Figure 6. Illustration of indirect cascading effects of copepods on a food web. Left panel: Net growth rate of prey type versus copepod biomass. Not all prey are plotted. Numbers under the panel for each of the prey types are regression coefficients. NS = non-significant regression coefficient.
Right panel: Best fit food web model derived from path analysis. Direction of arrow indicates consumption of prey. Numbers next to arrows are path coefficients, which indicate the strength of the interaction. Width of the arrow is proportional to path coefficient. Percentages next to prey type indicate % explained variance of the prey growth rate associated with all paths leading to that prey.
Trophic cascades and HAB: The work described so far led us to conclude that the stoichiometric imbalance effect was secondary relative to omnivory in driving possible cascading effects (Siuda and Dam, submitted (a)). In addition, work published by others after the start of this project revealed that any nitrogen limitation effects on Alexandrium, which would presumably alter predator-prey stoichiometric imbalance, would also result in changes in the saxitoxin content of the cells. That is, nitrogen limitation reduces saxitoxin per cell. In addition, the apparent lack of fixation of toxin resistance in copepod populations (Avery and Dam, 2007), the apparent lack of effect of saxitoxin on ciliates (Fig. 2) and the observed positive effects of toxin on grazers (Avery et al. 2008) were surprising results that led us to reconsider our original hypotheses. For instance, lack of fixation of resistance in a population made it impossible to properly test hypothesis 2. Accordingly, our strategy for trophic cascade experiments focused mostly on the effects of toxicity of cells.
We completed seven trophic cascade experiments similar in scope to the ones described above, but involving the toxic dinoflagellate Alexandrium and wild populations of copepods. These experiments tested for: 1) effect of addition of toxic Alexandrium to an otherwise nontoxic phytoplankton community; 2) possible effect of prolonged intoxication of grazers in the presence of toxic Alexandrium; 3) varying doses of toxic Alexandrium; 4) toxic versus nontoxic strain of Alexandrium; and 5) addition of higher trophic level (ctenophore) in expression of trophic cascade. In preparation for the ctenophore experiment, we also tested whether the ctenophores differentially fed on prey (copepods) that had ingested Alexandrium toxins or not—they did not. In all experiments, we measured growth rates of chlorophyll, microzooplankton, and phytoplankton as a function of varying consumer (copepod or ctenophore) abundance. Data have been processed and analyzed for the first four categories above. Analysis of the fifth category is still under way.
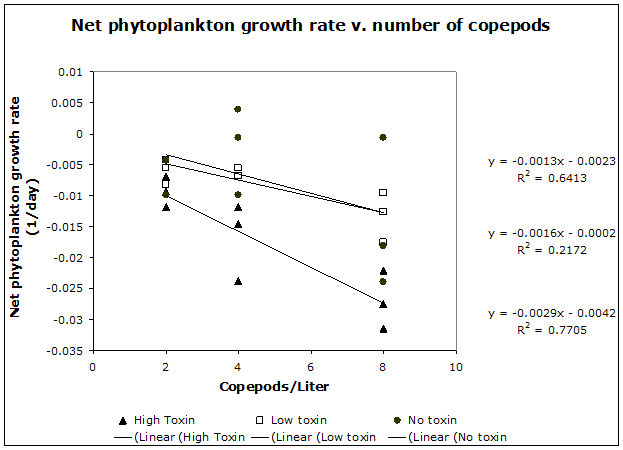
In all cases, we only observed direct effects (Fig. 7); that is, the net growth rate of phytoplankton decreased with increasing abundance of the consumer. Hence, no cascading trophic effects were obvious in any case. The implication is that the copepods behaved mostly as herbivores, or omnivorous feeding on both phytoplankton and microzooplankton obscured indirect effects. Figure 7 also illustrates a very interesting result—the direct effect was more pronounced as the toxicity of the cells increased (Fig. 7). This effect is consistent with the positive toxin effect of copepods shown in Fig. 3. A hypothesis for these results is the following: In all natural populations, there are individuals with different degrees of resistance to saxitoxin, and heterozygotes are favored (Avery and Dam, 2007). Heterozygotes have copies of mutant and wild forms of the sodium channel gene (Chen et al. 2007). The mutant form of the gene in copepods is not on the saxitoxin binding site, as is the case in bivalves, but in the inner side of the channel and results in incomplete inactivation of the channel (Chen et al. 2007), resulting in a leaky channel. Previous research has shown that leaky sodium channels have deleterious effects under normal conditions, and that sodium channel blockers are used to alleviate symptoms associated with leaky channels. Thus, we hypothesize that saxitoxin blocking of leaky channels restore normal function to nerve cells. Hence, under moderate toxin conditions (where not all sodium channels are blocked), an increase in saxitoxin improves copepod performance as long as the population is dominated by heterozygotes. If confirmed, this would represent a radically new mechanism of toxin adaptation in grazers.
Figure 7. Example of results from trophic cascade experiments in natural plankton assemblage. In this experiment, the control is the natural assemblage. Treatments of (relatively) low and high toxin are represented by addition of toxic Alexandrium cells to the natural community. The net community phytoplankton growth rate is shown versus the number of copepods (top predators in this case). An indirect effect (cascading effect) would have resulted in an increase of net phytoplankton growth rate with increasing addition of copepods. In this experiment, only direct effects were obvious.
Conclusions:
- Copepod resistance to saxitoxin is evidenced by differences in functional and numerical responses between copepod populations from areas historically exposed and not exposed to toxic Alexandrium blooms. That is, copepods from historically exposed areas perform better than those from areas unexposed to toxic Alexandrium. This means that there has been evolution of toxin resistance.
- Copepod saxitoxin resistance , however, does not appear to be fixed in populations presumably because of heterozygote advantage. The implication is that while grazer toxin resistance can lead to control of toxic blooms, this is not always a certainty.
- Microzooplankton respond in a different way than copepods to saxitoxin. That is, although some Alexandrium are toxic to ciliates, this effect is independent of saxitoxin content. This suggests a new mechanism of dinoflagellate toxicity to microzooplankton. Such mechanism is yet unknown and requires further investigation.
- There appears to be a positive response to saxitoxin in copepods. This finding, coupled with conclusion 2, above suggests a radically different mechanism for saxitoxin adaptation in copepods. This phenomenon requires further investigation.
- The two putatively new mechanism of toxin adaptation in microzooplankton and copepods require a revision of our concepts regarding saxitoxin transfer in the foodweb.
- In natural planktonic communities, the effects of omnivory appear to be stronger than those of elemental predator-prey stoichiometric imbalance. Thus, nutrient limitation of phytoplankton has a relatively minor role in trophic cascades.
- Direct effects of copepods on phytoplankton are evident in coastal regions. Trophic cascade effects are evident in more oligotrophic regions.
- The predominance of copepod omnivory or herbivory, and the relatively smaller role of stoichiometric predator-prey imbalance inhibit trophic cascades in communities exposed to toxic Alexandrium. The implication is that HAB have little effect on trophic cascades.
Journal Articles on this Report : 5 Displayed | Download in RIS Format
| Other project views: | All 29 publications | 5 publications in selected types | All 5 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Avery DE, Dam HG. Newly discovered reproductive phenotypes of a marine copepod reveal the costs and advantages of resistance to a toxic dinoflagellate. Limnology and Oceanography 2007;52(5):2099-2108. |
R831706 (2006) R831706 (2007) R831706 (Final) |
Exit Exit |
|
|
Avery DE, Altland KK, Dam HG. Sex-related differential mortality of a marine copepod exposed to a toxic dinoflagellate. Limnology and Oceanography 2008;53(6):2627-2635. |
R831706 (Final) |
Exit Exit |
|
|
Colin SP, Dam HG. Comparison of the functional and numerical responses of resistant versus non-resistant populations of the copepod Acartia hudsonica fed the toxic dinoflagellate Alexandrium tamarense. Harmful Algae 2007;6(6):875-882. |
R831706 (Final) |
Exit Exit |
|
|
Senft C, Avery D, Dam H. A novel approach to identifying PST tolerant copepods:An individual ingestion assay. HARMFUL ALGAE 2011;10(6):804-810. |
R831706 (Final) |
Exit Exit |
|
|
Siuda ANS, Dam HG. Effects of omnivory and predator-prey elemental stoichiometry on planktonic trophic interactions. Limnology and Oceanography 2010;55(5):2107-2116. |
R831706 (Final) |
Exit Exit |
Supplemental Keywords:
Marine, ecology, zoology, aquatic, toxics, RFA, Scientific Discipline, Water, Ecosystem Protection/Environmental Exposure & Risk, Oceanography, algal blooms, Ecological Risk Assessment, Ecology and Ecosystems, marine ecosystem, bloom dynamics, food web, nutrient kinetics, phytoplankton, algal bloom detection, grazing and window opportunitiesProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.