Grantee Research Project Results
Final Report: Sustainable Biodegradable Green Nanocomposites From Bacterial Bioplastic For Automotive Applications
EPA Grant Number: R830904Title: Sustainable Biodegradable Green Nanocomposites From Bacterial Bioplastic For Automotive Applications
Investigators: Drzal, Lawrence T. , Mohanty, Amar K. , Misra, Manjusri
Institution: Michigan State University
EPA Project Officer: Aja, Hayley
Project Period: January 1, 2004 through December 31, 2006 (Extended to December 31, 2007)
Project Amount: $369,613
RFA: Environmental Futures Research in Nanoscale Science Engineering and Technology (2002) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
The objective of this project is to replace/substitute existing petroleum derived polypropylene (PP)/TPO (thermoplastic olefin) based nanocomposites with eco-friendly, biobased nanocomposites produced from compatibilized clay reinforced bacterial bioplastic (polyhydroxyalkanoate, PHA) for automotive applications. The ‘green’ nanocomposites that are the subject of this project are ‘sustainable’ materials since they are: i) recyclable; are stable in use but can be ‘triggered’ to biodegrade under composting conditions; ii) are environmentally benign; and iii) are commercially viable. Compatibilization between the exfoliated clay and the bioplastic is the key to achieving success. This project has a goal of synergistically combining ‘green’ materials technology and nanotechnology to produce a new generation of sustainable materials for industrial applications that will have a positive impact upon the environment. In order to achieve ‘sustainability’ this project integrated the environmental, economic, life cycle analysis, energy and education components critical to achieving sustainability. The original aims/objectives remain unchanged throughout the project duration.
Summary/Accomplishments (Outputs/Outcomes):
A critical part of this research in the investigation of novel eco-friendly techniques for surface modification of the nano reinforcements. Currently, the use of quaternary complexes is reported using aromatic solvents that are environmentally persistent. Researchers have addressed this problem by: i) using an aliphatic solvent having similar characteristics as the aromatic solvent but significantly more environmentally benign and ii) developing a solvent-less technique using ultrasonic atomization of the modifier directly onto the surface. Researchers have also studied the blend PHAs with a low-cost biodegradable polymer in order to fabricate PHA-based materials intended for flexible packaging applications. Furthermore, researchers have addressed the issue of brittleness of poly(hydroxybutyrate), PHB from two different perspectives: i) the low nucleation density, and ii) the need for plasticization. In this context, expanded graphite nanoplatelets (xGnP) were shown to act as extremely good nucleating agents for PHB, while simultaneously being efficient reinforcements and capable of imparting electrical conductivity to the PHB matrix. Ionic liquids (ILs) were shown to plasticize PHB. The study addressed the adsorption of ILs onto xGnP surfaces and the implications on the mechanical properties of the resulted bionanocomposites. This study also aimed to compare and contrast the effect of xGnP on the crystallinity of PHB and another biopolyester from agricultural resources; polylactic acid, PLA and to determine the fundamental physical and chemical interactions responsible for the changes induced in polymers’ crystallization behavior and the mechanical properties of the nanocomposites obtained. The results are particularly important since they may be applied to similar semicrystalline polymers. Understanding how xGnP affects the nanoscale structure of the polymers and interferes with nucleation, lamellar structure, and crystallization rates will allow the optimization of the processing conditions leading nanocomposites with controllable mechanical properties.
Functionalized Bioplastic Synthesis:
Researchers successfully synthesized maleic anhydride (MA) and octadecenyl succinic anhydride (ODSA) and grafted it to the bacterial bioplastic poly (3-hydroxybutyrate)/PHB by using a solvent free method utilizing a twin-screw reactive extrusion process. The functionalized PHB was verified by spectroscopic, microscopic and thermal analyses. This synthesized maleated PHB was used as a compatibilizer in the preparation of PHB based clay nano-composites. As a result, improved engineering properties such as flexural, tensile and impact strengths and moduli were obtained when compared to the pristine material. Addition of a small amount of this maleated PHB compatibilizer leads to its reaction with the OH groups of organically modified clay and this in turn helps to increase the d001 spacing between clay galleries.
Toughened Bacterial Bioplastic Synthesis:
The bacterial bioplastic PHB is isotactic in nature and combined with its chain linearity results in a highly crystalline material with very attractive strength and modulus but very poor elongation (~3%) and low impact strength (23 J/m). Researchers successfully toughened the inherently brittle PHB by using a functionalized natural elastomer (epoxidized natural rubber, ENR) and a maleated compatibilizer. The effects of processing conditions, various types and content of the elastomers and compatibilizers on the performance of these toughened materials have been evaluated.
Surface Modification of Clay:
The clay surface has successfully been modified with alkyl-titanate complexes to make it organophilic and thus ideal for organic matrices to produce nanocomposites with enhanced properties. The hydroxyl groups of clay are targeted to be exchanged with alkyl-titanate complexes from the surface modifier in the modification reaction. i) Clay surface modification was carried out in a non-aromatic solvent (Kwik-dri) and compared with the modification that was done by the traditional aromatic solvent toluene. It was observed that, the same surface modification levels were achieved without any compromise in characteristics. ii) A solvent-less process was developed that worked by atomizing the coupling agent onto a fluidized bed of clay. The atomizing was achieved by using an ultrasonic probe that produced a fine spray of the titanate-coupling agent directly onto the clay thus eliminating the need for any solvent. The XPS spectra of the modified clays show significant reduction in the atomic concentration of oxygen thus justifying the modification mechanism. The contact angle for pristine clay is close to zero as is expected for the hydrophilic surface with ample hydroxyl groups capable of interacting with water. For the modified clay, the organic groups in the alkyl-titanate complex increase the surface energy of the clay surface. This increase in the surface energy reflects in decrease in wettability and thus the contact angle increases. The increase in contact angle was seen to be in agreement with the increase in organic modifier content on the clay surface as also determined from the XPS. Similarly XRD studies revealed the inter-gallery spacing increased from 9.8 to 12.7 A°. This new surface-modification method provides an effective technique to convert the hydrophilic surface of the montmorillonite clay into organophilic for polymer-clay nanocomposites.
Clay Nanocomposites from Toughened PHB:
Organically modified clay platelets were introduced into the toughened blend system to regain the stiffness and modulus. Nanocomposites were melt compounded in the micro-compounding-molding instrument. Initially a masterbatch of 20% loaded clay with PHB was prepared in the microcompounder and then mixed with the calculated amounts of other components to fabricate nanocomposites. After extrusion, the molten materials were transferred through a preheated cylinder to the mini injection molder (pre-set at mold temperature of 60°C) to obtain the desired specimen samples for various measurements and analysis. PHB-ENR compatibilized nanocomposites with 5 wt % modified clay loading showed ~300% improvement in impact properties and only 40% reduction in modulus in comparison with pure PHB. The mechanical properties of the nanocomposites have been correlated with theoretical model predictions. A modified Haplin-Tsai equation was used to evaluate the reinforcing effect of randomly-oriented clay platelets (Ref: Halpin, J. C., Journal of Composite Materials, 1969, 3, 732-734 and Hill, R., Journal of the Mechanics and Physics of Solids, 1964, 12, 199-212). The elastic modulus of clay is taken to be 170 GPa from literature in these calculations (Ref: Giannelis, E. P., Advanced Materials, 1996, 8, 29-35). Experimental values of modulus are 1.6 GPa for PHB and 0.0017 GPa for rubber. Applying the Halpins-Tsai equation to just the toughened PHB matrix without the nanoclay while considering the rubber particles as the reinforcements in the PHB, the effective predicted modulus is 0.874 GPa. The volume fraction of rubber (Vrubber) is 0.25 and the rubber regions are assumed to be spherical droplets with L/D=1. Van-Es has proposed the following parameter (Ref: Es, M. Van, 2002, Ph.D. Thesis, Technical University of Delft, Delft): ξ =2/3 (L/D) and η = (Ereinforcement-Ematrix)/(Ereinforcement+ξEmatrix) Comparing this with the experimental modulus value of 0.77 GPa for the same composition, it is seen that the actual modulus is 88.1% of the predicted value. This difference can be attributed to the interface between the PHB and the rubber particles and hence subsequent calculations for the nanocomposite modulus are multiplied by this factor. Also the modified rubber is in liquid form and cannot be characterized for modulus and so is considered to have similar physical properties as ENR in the model and this leads to the difference between the predicted and the actual value. Similarly considering the clay in virgin PHB system, the predicted modulus by the Halpin-Tsai equation is 2.35 GPa while the experimental value for this system is 1.9 GPa. This again can be attributed to the clay-PHB interface and hence a factor of 80.8% is also considered in the calculations of the toughened PHB nanocomposites. The fabrication method for the toughened PHB nanocomposites, as explained in the experimental methods section, is by initially making a highly concentrated master-batch of the clay in virgin PHB and subsequently adding the rest of the components. This fabrication technique leads to two boundary scenarios for the clay distribution: case I where all the clay remains in the PHB phase and case II where the clay is distributed equally into the rubber and PHB regions as denoted in Figure 1. These two conditions are numerically modeled as function of varying clay aspect ratio (L/D) from 10 to 100 in the nanocomposites. The volume fraction of rubber (Vrubber) is 0.25 for all the cases and the rubber or rubber-clay regions are assumed to be spherical droplets with L/D=1. Case I: The boundary case assumes all the clay remains in the PHB regions and hence the the Halpin-Tsai equation was initially applied to the clay-rich PHB phase and equation 10 gives the predicted modulus value for these regions as: EI = EPHB (1+ξηVclay) / (1-ηVclay) (1)
The volume fraction of clay (Vclay) is 0.0288 for case I and η = (Eclay-EPHB)/(Eclay+ξEPHB). Using the value of EI from the above equation and ERubber in a combined equation 2 gives the modulus of the final nanocomposite: Enanocomposite (I) = EI (1+ξηVRubber) / (1-ηVRubber) (2) In this case η = (ERubber-EI)/(ERubber+ξEI) and the rubber regions are again assumed to be spherical droplets with L/D=1. The final value is multiplied by 0.881 (rubber-PHB interface effect) and 0.808 (clay-PHB interface effect) to get the effective value of the modulus and this is plotted as a function of clay aspect ration in Figure 2. Case II: The lower boundary case assumes equal distribution of the clay in the PHB regions and the rubber regions. Thus the Halpin-tsai equation is first applied to the clay-rich PHB phase and equation 3 gives the predicted modulus value for these regions as: EII = EPHB (1+ξηVclay) / (1-ηVclay) (3) The volume fraction of clay (Vclay) is 0.0187 for case II for both the regions and η = (Eclay-EPHB)/(Eclay+ξEPHB). For the rubber-clay region, the modulus is given by equation 4: EIII = ERubber (1+ξηVclay) / (1-ηVclay) (4) with η = (Eclay-ERubber)/(Eclay+ξERubber). Using the value of EII and EIII from the above equations in a combined equation 5 gives the modulus of the final nanocomposite: Enanocomposite (II) = EII (1+ξηVIII) / (1-ηVIII) (5) In this case VIII= 0.25, η = (EIII-EII)/(EIII+ξEII) and the rubber-clay regions are again assumed to be spherical droplets with L/D=1. Again the final value is multiplied by 0.881 (rubber-PHB interface effect) and 0.808 (clay-PHB interface effect) to get the effective value of the modulus and this is plotted as a function of clay aspect ration in Figure 2.
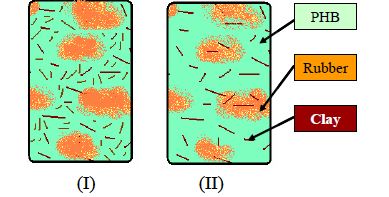
Figure 1: Schematic representation of morphology and clay distribution used for modified Halpin-Tsai equation. (I) Case I: all the clay remains in the PHB phase and (II) Case II: The clay is distributed equally into the rubber and PHB regions.
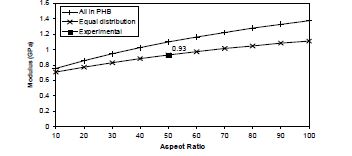
The experimental value for this system is 0.91 GPa and the subsequent TEM images show the aspect ratio for the clay to be ~50. The predicted value for this aspect ratio is 0.93 for the equal-distribution model and 1.53 for the model that considers all the clay in the PHB. Thus the experimental value is closely in agreement to the case II model and this suggests that the system is behaving in a manner as to influence the clay to migrate from the PHB to the rubber regions. There is also no empirical technique to determine the exact oncentration of the clay in the PHB and the rubber regions and hence this distribution and migration cannot be established.
Blending of PHB with a Flexible Biodegradable Polymer followed by Nanoclay Incorporation:
PHB films or sheets cannot be made by conventional processing due to their low elongation (3%). A flexible polymer additive for PHB was sought, which will give synergistic properties sufficient for film/sheet applications. PHB was blended with a flexible (760% elongation) petroleum-based biodegradable polymer e.g. Poly-(butylene adipate-co-terephthalate) (PBAT) by twin-screw extrusion. PHB-PBAT blends of varying compositions were reinforced with 5% organically modified montmorillonite (OMMT) clay. These blends and their nanocomposites were fabricated into films using a single screw extruder coupled to a blown film line. The blends and nanocomposites were characterized through thermo-physical and mechanical analysis. The above material combination exhibited attractive mechanical properties and displayed increased oxygen barrier properties over that of traditional polyolefins and nylon.
Blending of poly (3-hydroxybutyrate valerate)/PHBV with a Plasticized Starch Composition Followed by Nanoclay Incorporation:
The reactive blending of a plasticized biobased polymer with PHBV matrix to produce a novel and flexible biodegradable material was studied. A thermoplastic starch (TPS) system was developed by blending of corn starch, glycerol (byproduct of soy-based biodiesel industries) and poly-(butylene adipate-co-terephthalate) (PBAT). Following optimization by structure-property-processing correlation, this thermoplastic starch system was blended with PHBV to obtain a biodegradable flexible polymer blend and further reinforced with talc and processed into cast films. The incorporation of talc promoted disruptive mixing, homogeneity and subsequently reduced the droplet size of the components ultimately giving improvement in physical properties. The talc-filled films showed remarkable improvement in barrier properties because of a combination of the tortuosity effect and the nucleating effect of the talc and the improved mixing. The ability of PHBV-TPS system as well as the talc-filled composites to resist ageing was also established.
Figure 2: Predicted and Experimental values of the modulus of the nanocomposite materials as a function of clay aspect ratio.
Biodegradation by Respirometry:
Respirometry experiments were done in collaboration with United States Department of Agriculture’s Bioproduct Chemistry and Engineering Research, Western Regional Research Center, Agricultural Research Service at their facilities. Samples were milled to a fine consistency (average particle size is 25 μm) using a hammer mill under liquid nitrogen environment. These milled samples were analyzed for initial carbon content using an automatic Carbon/hydrogen/nitrogen analyzer. About 0.2 g of each sample of known carbon content was mixed with 20 g of compost under appropriate conditions. These sample-soil mixtures were kept in separate 250-mL sample chambers of a fully computerized, closed-circuit Micro-Oxymax Respirometer System (Columbus Instruments, Inc., Columbus, OH) equipped with an expansion interface, a condenser, and a water bath. The sample chamber was placed in a water bath controlled at 25° C and 50% RH and connected to the Micro-Oxymax Respirometer. Experiments were carried out over a period of 105 days. CO2 evolution from each sample was measured every 8 hours. The total available carbon in the starting material (0.2 g) of each sample was calculated from the predetermined percent carbon values. The values for the total accumulated CO2 volume during biodegradation for a sample in a reaction chamber were obtained through respirometry. These volume V data were used in the equation of state of the ideal gas law; PV = nRT, to determine the moles (n) of carbon converted into CO2 as a result of microbial processes in a compost, where the molar gas constant R= 0.082055 L-atm/K-mol; T = 298 Kelvin (25° C), and pressure P = 1 atm. The percent biodegradation was obtained from the sum of net moles of CO2 released in a sample chamber divided by the moles of CO2 present in the original sample and multiplied by 100. For aerobic biodegradation, samples were ground to a fine particle size using a granulator with a 2 mm screen. The compost was obtained from the Michigan State University composting facility (East Lansing, MI). The compost pile had an initial composition of cow manure, wood shaving and waste and was matured for 3 months. The initial temperature of the compost was 65 ± 5° C with relative humidity of 63 ± 5%, and pH of 8.5 ± 0.5 as given by the provider. This was altered by drying under vacuum to get 50± 1 % relative humidity. The compost as received was filtered through a wire mesh having 5 mm hole size in order to get an uniformly sized material devoid of large chips. 15 g of each polymeric material that is to be tested was mixed with 180 g of compost having 50% moisture in order to get a testing material to compost dry solids ratio of 1:6. These mixed samples were introduced into the testing chambers set at 60°C for testing. The experimental setup was designed as per ASTM D 5338. The external air is scrubbed of CO2 by bubbling it through sodium hydroxide solution (10 N strength). This carbon dioxide-free air is then fed through distilled water to appropriately moisten the air stream and this is then divided to each biodegradation chamber. Fifteen chambers were prepared having experimental compositions. The above air stream passes through the biodegradation chamber and is then fed to individual bottles containing sodium hydroxide (0.25 N). This sodium hydroxide reacts with the carbon dioxide released from the biodegradation reaction present in the air and forms sodium carbonate. This solution of sodium carbonate and unreacted sodium hydroxide was titrated over specific time intervals against hydrochloric acid (0.5 N) to determine the carbon dioxide released by aerobic biodegradation. The entire biodegradation setup was installed in a controlled environmental chamber maintained at 60 °C and 50% relative humidity for the duration of the experiment. The system was allowed to tabilize overnight and then the air stream was introduced into the chambers. The air was controlled by the individual flow meters on the chambers so as to achieve a bubble rate of approximately one per second. All tested samples degraded in compost, albeit at different rates and extents (Figure 3).
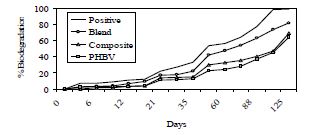
Figure 3: Aerobic Biodegradation of the PHBV-TPS and PHBVTPS-talc composite with 5 wt. % talc.
From the iodegradation view point, PHBV-TPS showed the maximum rate of biodegradation. The PHBV component is the slowest to degrade amongst the constituents and this agrees with literature values. The PHBV-TPS-talc composite samples appeared to be quite retarded in biodegradation possibly because of the homogeneous mixing and presence of talc that is limiting diffusion of reactants and products to the polymer by the compost bacteria. The positive sample (pure starch) showed 60% biodegradation in 60 days indicating the validity of the test. Testing standards mandate 60% biodegradation in 190 days for a system to be considered biodegradable. The biodegradation values for the PHBV-TPS system as well as the PHBV-TPS-talc composite indicate that this value is being approached well within the required time period; both these systems were 60 % biodegraded in 125 days.
Plasticizing effect of environmentally benign ionic liquids on PHB:
Attempts were made to reduce the brittleness of PHB by plasticizing it using ionic liquid. A literature review of toxicity of ionic liquids indicated that imizazolium acetates were non-toxic, good candidates for plasticizing PHB, but the experiments and analyses (thermal, morphological and molecular weight evaluation) of PHB/1-butyl-4-methylimidazolium acetate systems revealed that the polymer was actually degraded in the presence of the ionic liquid.
Expanded graphite nanoplatelets (xGnP) as nucleating agent for PHB:
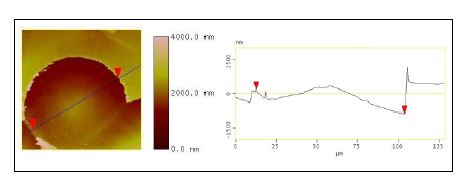
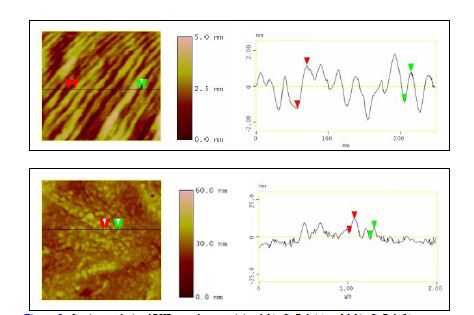
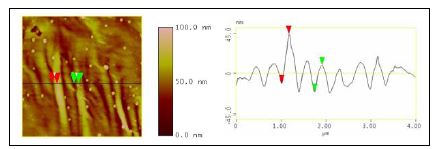
A new nanoreinforcement developed at MSU, expanded graphite nanoplatelets, xGnP acted as nucleating agents and reinforcements for poly(hydroxybutyrate). A detailed kinetic study of the non isothermal crystallization, important in the view of optimizing processing conditions, was performed. The morphology of the resulted nanocomposites was studied by optical microscopy and atomic force microscopy. The percolation threshold for PHB/xGnP-1 systems seems to be above 3 % (wt) xGnP-1. This assumption is based on the results of preliminary experiments done to measure the electrical conductivity of PHB and PHB/xGnP-1 samples. So far, the results show that the samples were not electrically conductive, which means that the amount of xGnP-1 in the PHB matrix needs to be higher than 3 % in order to reach the percolation threshold. At this point, the PHB/xGnP-1 systems have the potential for developing into new novel bio-nanocomposites with enhanced mechanical, thermal, and barrier properties. This study continues with a goal to produce electrically conductive bio-nanocomposites based on xGnP-1 and poly(hydroxyalkanoates) that would increase the potential applications of these green materials. AFM images were collected for the pure PHB, PHB containing 1 % xGnP-1, and PHB containing 3 % xGnP-1 (Figures 4 – 6). The spherulites of pure PHB were very large (approximately 90 μm, as indicated in the section analysis in Figure 4) in comparison to the ones formed in the presence of xGnP-1. The spherulites of pure PHB were also more ordered and well-defined, so it was easy to measure their sizes. In case of PHB/xGnP-1 systems, the spherulites seemed to grow into each other so it was difficult to determine where one ended and another one begun. Smaller scans show that the lamellar structure is still present in the PHB/1 % xGnP-1 system (Figure 5, a), and also what appears to be a distribution of expanded graphite nanoplatelets through the crystalline structure, especially at the higher (3 %) concentration of xGnP-1 (Figure 5,b). The crosssectional analysis of PHB samples containing 1 % xGnP-1 showed that the thickness of the lamellae is between 1-3 nm. Previous reports indicated that PHB lamellae have a thickness of 5 nm. Our results indicated that the lamellar thickness decreases as the amount of xGnP-1 increases, and is proportional to the size of the spherulites. PHB samples containing 0.01% xGnP-1 are shown in Figure 6.
Figure 4. Section analysis of a PHB spherulite
Figure 5. Section analysis of PHB samples containing 1 % xGnP-1 (a) and 3 % xGnP-1 (b)
Figure 6. Section analysis of PHB samples containing 0.01 % xGnP-1
Effects of expanded graphite nanoplatelets (xGnP™) on poly(hydroxybutyrate) and polylactic acid and nanocomposite properties:
The crystallization of xGnP/PHB and xGnP/PLA nanocomposites was compared in the view of understanding the nanoscale interaction between xGnP and these semicrystalline biopolyesters. Two aspects of PHB/xGnP and PLA/xGnP nanocomposites are analyzed in the view of the nucleating effect of xGnP: − the rate of overall crystallization as a function of the amount of xGnP, and − the influence of xGnP on the morphology and properties of the nanocomposites. The preliminary results obtained PHB/xGnP and PLA/xGnP systems showed different extents of the nucleating effect of xGnP, depending on each polymer’s crystallization behavior. Attempts were made to relate the kinetics of the crystallization processes and the morphologies of the resulted bionanocomposites such that xGnP can be used to tune the properties according to the desired properties. The results for nanocomposites prepared by extrusion followed by injection molding indicate that relying solely on the mixing during the extrusion process might not be sufficient for achieving good dispersion of the graphite platelets. Alternative mixing methods need to be investigated in relation to dispersion of nanoparticles, with the ultimate goal of obtaining better understanding of the distribution of expanded graphite platelets in semicrystalline polymer matrices.
Scanning Probe Thermal Analysis of Polylactic Acid/ Exfoliated Graphite Nanoplatelet (xGnP™) Nanocomposites:
Scanning probe thermal analysis, or nano-thermal analysis (nano-TA), is a technique utilizing a novel micromachined scanning probe microscopy tip with an integrated heater to measure thermal properties of polymeric materials. The primary measurement mode is to position the tip at a fixed location, increase the tip temperature, and monitor the deflection, to detect transitions such as melting temperature (Tm) and glass transition temperature (Tg). The spatial resolution for imaging with this system is reported to be one hundred times better than its predecessor, μTA, primarily due to advances in probe technology. The new probes have increased tip height and sharpness, enhanced cantilever geometry for laser alignment and relatively low spring constants, which are important features for imaging soft materials such as polymers in the contact mode without causing significant damage to the surface of the specimen. Experiments were conducted to determine the effect of heating rates on the Tm of a variety of polymers and to establish suitable calibration standards over a wide temperature range. Poly(ξ-caprolactone) (PCL) and poly(ethylene terephthalate) (PET) are suitable materials for use as calibration standards at 60 oC (low) and 240 oC (high) melting ranges. In this work, polymers such as polylactic acid (PLA), polyethylene (PE), a polypropylene (PP) and polyhydroxybutyrate (PHB) were investigated. In particular, the results of the nano-TA are compared with traditional differential scanning calorimetry (DSC) methods, and the implications of localized heating and melting versus complete melting are discussed. The extrapolated onset and the endothermic peak melting and crystallization temperatures, as measured by DSC, are correlated here with the nano-TA analysis. While DSC gives quantitative information regarding the dependence of the degree of crystallinity on cooling rates, nano-TA offers the unique advantages of in-situ observation of the polymer’s morphology under the same conditions, providing valuable information for practical applications. Scanning probe thermal analysis of two different molecular weight poly(L-lactic acid) samples and their composites with expanded graphite nanoplatelets revealed essential differences in cold rystallization of the two polymer samples in the presence of the nanoplatelets as well as the dispersion of xGnP in the polymers. The difference in melting and crystallization behavior of the two polymers was not detectable by differential scanning calorimetry, which by itself could not explain the differences in the mechanical properties of the composites. Nanoscale investigations of the thermal transitions occurring in polymer nanocomposites were proven to be important for the characterization of the dispersion of nanosized reinforcements, as well as for understanding the extent of their nucleating effect on semicrystalline matrices.
Thermal conductivity of polylactic acid/ expanded graphite nanoplatelets composites:
Composites with high thermal and electrical conductivities were obtained by incorporating expanded graphite nanoplatelets in polylactic acid matrices. Electricity and heat follow different transport mechanisms, but the platelet geometry of xGnP appears to lead to better transport when percolated networks care obtained. The thermal conductivity of PLA composites containing 25 % xGnP-15 was 560 % higher than that of neat PLA. Using polymer blends to obtain better localization of the xGnP in the polymer matrices appears to be a promising strategy that can lead to improvements in both thermal and electrical conductivities, as shown by our current results. The polymer blends approach is currently considered and will continue to be considered by future projects in our research group.
Assessing life cycle environmental performance of bioplastic- montmorillonite clay nanocomposites:
Biopolymer layered silicate nanocomposites are emerging as attractive alternatives to conventional filled/nanofilled polymers or polymer blends in automotive applications due to their comparatively similar mechanical properties and weight saving potential. Biodegradability of the components at the end of life and potentially lower greenhouse gas emissions are additional attractive features of these nanocomposites employing biobased polymers such as poly(lactic acid) (PLA) and polyhydroxybutyrate (PHB). However, taking a comprehensive life cycle perspective is essential while assessing the relative environmental performance of biobased nanocomposites. The study on conceptual and empirical issues in carrying out life cycle environmental assessments of biobased nanocomposites using a PHB-organoclay nanocomposite are under progress.
Conclusions:
Environmentally friendly green nanocomposites were successfully developed having the potential to serve as eco-friendly materials for automotive packaging applications. A new surface treated nanoclay was synthesized which acted as a reinforcement for the toughened bioplastic in presence of a compatibilizer. Expanded graphite nanoplatelets were identified as an efficient alternative nucleating agent and reinforcing agent for bioplastics.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 30 publications | 3 publications in selected types | All 3 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Joshi S. Can nanotechnology improve the sustainability of biobased products? The case of layered silicate biopolymer nanocomposites. JOURNAL OF INDUSTRIAL ECOLOGY 2008;12(3):474-489 |
R830904 (Final) |
Exit |
Supplemental Keywords:
RFA, Scientific Discipline, INTERNATIONAL COOPERATION, TREATMENT/CONTROL, Sustainable Industry/Business, POLLUTION PREVENTION, Sustainable Environment, Energy, Environmental Chemistry, Technology, Technology for Sustainable Environment, Chemistry and Materials Science, Chemicals Management, Environmental Engineering, biopolymers, energy conservation, biodegradable plastics, clean technologies, cleaner production, environmentally conscious manufacturing, green design, nanocomposite, air pollution control, automotive industry, environmental conscious construction, environmental sustainability, nanotechnology, biodegradeable nanocomposites, alternative materials, clean manufacturing, environmentally applicable nanoparticles, environmentally friendly green products, nanomaterials, environmentally benign alternative, nanoparticles, Design for Environment, polypropylene substitute, automotive interior parts, environmentally conscious designProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.