Grantee Research Project Results
Final Report: Lumped Chemical Approach for Fate and Transport Modeling of Organic Pollutant Mixtures
EPA Grant Number: R829355Title: Lumped Chemical Approach for Fate and Transport Modeling of Organic Pollutant Mixtures
Investigators: Reardon, Kenneth F. , Shackelford, Charles D. , Pruden, Amy
Institution: Colorado State University
EPA Project Officer: Hahn, Intaek
Project Period: October 1, 2001 through September 30, 2004 (Extended to January 31, 2006)
Project Amount: $721,695
RFA: Complex Chemical Mixtures (2000) RFA Text | Recipients Lists
Research Category: Hazardous Waste/Remediation , Safer Chemicals , Land and Waste Management
Objective:
Contamination of soil and water is caused more often by chemical mixtures than by single pollutants. Common examples are gasoline, landfill leachates, and fuel hydrocarbon/chlorinated solvent mixtures. The presence of these contaminant mixtures poses challenges for risk assessment and risk management. With regard to the former, direct or indirect interactions among mixture components lead to different exposures and effects. Remediation methods are similarly affected (e.g., the presence of one chemical may inhibit the degradation of another), and conflicting or incompatible conditions may be required to remediate all of the contaminants in the soil or water.
Clearly, knowledge and description of the movement of contaminant mixtures is critical for both risk assessment and risk management. For individual chemicals, transport and fate models have been developed that incorporate advection, dispersion, sorption (and/or other partitioning phenomena), and reactions. But how should this be done for a mixture of chemicals? The primary goal of this research project was to evaluate the use of pseudocompounds (grouped chemicals) for modeling the transport and fate of organic chemical mixtures. A secondary goal was to evaluate microbial community changes and changes in biodegradation rates along the flow path of a model aquifer system. The four objectives of the project were: (1) measure and model mixture biodegradation kinetics; (2) measure and model mixture transport properties; (3) evaluate mixture transport and fate in soil columns (including microbial population analysis); and (4) develop lumped chemical modeling for the transport and fate of the organic chemical mixture.
The pollutant mixture used in this study was chosen with the following criteria: prevalence at Superfund sites, high rankings on the Agency for Toxic Substances and Disease Registry (ATSDR) list, at least moderate solubility (to facilitate experimentation), documented biodegradability, and groups of three from the same structural class (at least one of which would not be aromatic). The selected mixture chemicals were benzene (BZ), toluene (TOL), m-Xylene (m-XYL), chlorobenzene (CB), 1,4-dichlorobenzene (1,4-DCB), 1,2,4-trichlorobenzene (1,2,4-TCB), acetone (ACE), 2-butanone (2-BUT), 2-hexanone (2-HEX), phenol (PHE), p-cresol (p-CRE), and 2,4-dimethyl phenol (2,4-DMP). Chemical concentrations were determined by a two-stage gas chromatographic method: nonpolar chemicals were extracted into hexane and analyzed by gas chromatography-mass spectrometry (GC-MS), while non-polar chemicals were analyzed by gas chromatography-flame ionization detection (GC-FID) with direct injection of aqueous solutions.
Approach:
To carefully establish the utility of lumped chemical modeling, the research outlined in this proposal uses a model system of a 13-chemical mixture (benzene, toluene, ethylbenzene, chlorobenzene, o- and m-xylene, o- and m-cresol, phenol, naphthalene, diethyl and dibutyl phthalate and phenanthrene), a stable microbial community, and a set of model soils. The work consists of two kinds of experiments (suspended cell batch cultivations and soil column tests) and mathematical modeling/simulation. The experimental results will guide the development of the lumped chemical model, and some of the Objective 3 data will be reserved for validation of the model. The research will be performed by a multidisciplinary team of engineers and scientists.
Summary/Accomplishments (Outputs/Outcomes):
Mixture Biodegradation
Microbial Community. Biodegradation of the 12-chemical mixture was studied with a microbial community enriched from soil samples from several sites in Fort Collins, CO. The community was maintained in a mixed continuous flow bioreactor on a mineral salts medium. Characterization of the microbial community has employed three approaches: denaturing gradient gel electrophoresis (DGGE), capillary electrophoresis single-strand conformational polymorphism (CE-SSCP), and pure culture isolation. DGGE and CE-SSCP were the primary tools for monitoring changes in the composition of the community with time (continuous bioreactor), with location (column studies, below), and with exposure to different substrates in the batch tests. Also, cutting the bands and reamplifying and sequencing the DNA present enabled the determination of the phylogenetic identity of the organisms represented by the bands. Pure culture isolation was used to determine which species were able to grow on which chemicals. The full-length 16S rRNA gene sequences were obtained for the isolates, and they were grown in liquid culture and stored as freezer stocks.
Biodegradation Kinetics. Batch biodegradation testing demonstrated that all 12 of the chemicals were degraded when present in the full mixture. Also observed were mixture effects, both inhibition and enhancement. Notably, acetone and toluene degradation was inhibited in the mixture, while butanone degradation appeared to be enhanced. None of the three chlorinated benzenes degraded significantly alone, but all three were degraded in the full mixture, indicating that these chemicals are only degraded through cometabolism (Figure 1).
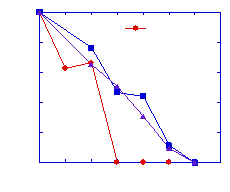
Figure 1. Observed Mixture Effects on Biodegradation. Left: inhibition of toluene biodegradation in mixture. Right: stimulation of chlorobenzene biodegradation (cometabolism).
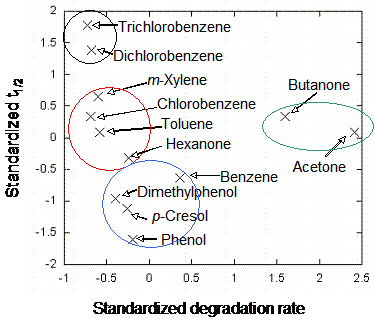
Lumping Analysis for Mixture Biodegradation. “Lumping” is the process of grouping similar chemicals into pseudocompounds, which are then assigned a characteristic property and used to represent the group. This grouping procedure can greatly simplify the complexity of the mathematical model needed to predict the chemical process. Since the chemicals within a pseudocompound are not perfectly represented by the pseudocompound’s properties, lumping leads to loss of information. The optimal point in the tradeoff between simplification and accuracy is situation specific. On the basis of the time to biodegrade half of the initial chemical concentration (t1/2) and a standardized biodegradation rate, four pseudocompounds for biodegradation were identified (Figure 2).
Figure 2. Identification of Pseudocompounds for Biodegradation
Sorption of Chemicals and Mixtures To Model Aquifer Materials
Sorption of Individual Mixture Components To Model Aquifer Materials. The sorption of each chemical was first studied individually in order to assess whether mixture effects were significant. Since sorbent properties were expected to have an impact on the extent of mixture effects, seven model sorbents were prepared: silica sand coated with humic acid to achieve fractions of organic carbon (foc) of 0.006%, 0.06%, 0.15%, 0.22%, and 0.54%; silica sand coated with iron hydroxide and humic acid (foc = 0.042%); and silica sand coated with aluminum hydroxide and humic acid (foc = 0.031%). The sorption behavior of individual chemicals was influenced primarily by their hydrophilic/hydrophobic nature. Regardless of the model sorbent, sorption of the more polar chemicals (ACE, 2-BUT, 2-HEX, PHE, p-CRE, 2,4-DMP) could be described by the nonlinear Freundlich isotherm, while sorption of the nonpolar chemicals (BZ, TOL, m-XYL, CB, 1,4-DCB, 1,2,4-TCB) was described by the linear isotherm (Table 1). Sorption of nonpolar chemicals increased significantly with foc, while sorption of polar chemicals was only slightly higher to sorbents with higher foc.
Table 1. Summary of Sorption Results for Individual Chemicals and Mixtures (HA = Humic Acid)
Sorbents |
Organic Carbon Fraction, foc (%) |
Sorption Isotherm |
Mixture Effect Based on Statistical Analysesa |
|
Heated at 700°C |
< 0.005 |
Linear for nonpolar |
No |
|
Untreated |
0.006 |
Linear for nonpolar |
No |
|
HA-coated |
0.150 |
Linear for nonpolar |
No |
|
FeO(OH)-coated |
0.006 |
Linear for nonpolar |
No |
|
FeO(OH)-coated+HA |
0.042 |
Linear for nonpolar |
No |
|
Al2O3-coated |
< 0.005 |
Linear for nonpolar |
No |
|
Al2O3-coated+HA |
0.031 |
Linear for nonpolar |
No |
Sorption of Mixtures To Model Aquifer Materials. To ascertain whether the sorption of a chemical was different when it was present in a mixture, mixture sorption experiments were performed and the parameters (Kf and n) of the Freundlich isotherm determined. The 95% confidence limits of least squares means for these parameters were determined, and Dunnett’s procedure was used to compare parameter values between control (i.e., individual-sorbate systems) and treatments (i.e., mixture). Significant (α = 0.05) mixture effects were identified for sorption of polar chemicals to the model sorbents, but not for nonpolar chemicals (Table 1). In addition, the observed mixture effects occurred in a highly selective manner. For example, a large suppression in sorption of chemicals with intermediate polarity (i.e., PHE and p-CRE) to several sorbents by chemicals with greater polarity (i.e., ACE and 2-BUT) was found. The absence of significant mixture effects for nonpolar chemicals is attributed to the enrichment of nonpolar chemicals in the vicinal water regions, without competing for the specific sites among nonpolar chemicals. In addition, the nonpolar chemicals cannot compete with polar chemicals as well as with water for the specific sites on the hydrophilic mineral surfaces and for functional groups of humic acid. However, polar chemicals can be adsorbed closer to the surface due to hydrogen bonding with surface hydroxyls or with strongly adsorbed water molecules, and organic carbon may have influence via specific interactions in addition to hydrophobic partitioning. Thus, other polar chemicals compete for specific sites on mineral surfaces or functional groups with humic acid. These results suggest that different mechanistic processes are operative for the sorption of polar and nonpolar sorbates in a mixture.
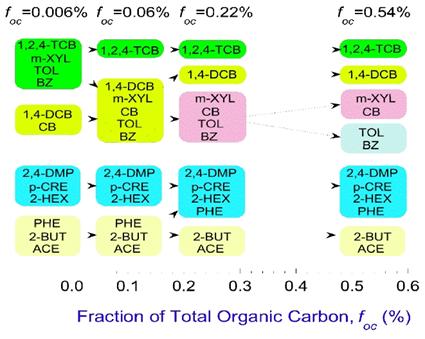
Lumping Analysis and Sorption Modeling with Pseudocompounds. Lumping analysis was also performed for sorption. There are no established methods for assembling pseudocompounds, and thus four methods of cluster analysis were tested (i.e., average linkage, centroid, median, and Ward). In all cases, clustering was performed using parameters of the Freundlich isotherm (Kf and n). The best method was determined to be cluster analysis with Ward’s method, with the criterion for which data were set to fuse chosen to be minimization of the increment of the total error within the group error sum of squares caused by fusion. Separate cluster analyses were performed for six different sorbents—one set of humic acid-coated quartz sand with four foc levels, and one set with different mineral coatings. The pseudocompounds formed for each sorbent were slightly different (Figures 3 and 4).
Figure 3. Pseudocompounds for Sorption as a Function of Sorbent foc
Figure 4. Pseudocompounds for Sorption as a Function of Mineral Hydroxide Surface
The pseudocompounds identified for each sorbent were used in a model derived from the ideal adsorbed solution theory (IAST), and the results were compared to those produced with individual chemicals. The two approaches yielded similar results, supporting the use of lumping analysis and suggesting that a minimum amount of information was lost in the lumping process.
Spatial Heterogeneity of Microbial Populations
The composition of an organic pollutant mixture has a strong influence on the makeup of the microbial population growing on it, which in turn affects the rates at which various components of the mixture will be consumed (thereby changing the composition of the pollutant mixture). Thus, there is an important connection between the composition of the pollutant mixture and that of the microbial “mixture.” As a chemical mixture moves along the flow path in an aquifer, the more rapidly biodegraded components will be depleted, and this can be expected to impact the composition of the microbial community. To test this hypothesis, soil column tests were performed using untreated and humic acid-coated sands. Column tests consisted of feeding the synthetic groundwater containing individual or mixtures of organic chemicals into a column [60 mm (ID) x 400 mm (L)] until solute breakthrough occurred. The concentrations of individual or mixtures of chemicals also were determined as a function of time along the length of the column. Notably, a novel solid-phase microextraction (SPME) method was developed for use in column experiments, because the available sample volumes were small. (An invention disclosure has been filed with Colorado State University for this method.)
The CE-SSCP method was used to provide a fingerprint of the microbial community at four locations along the column (Figure 5). The results of this analysis provided clear evidence that the community near the inlet, where most biodegradation occurred, was significantly different than the communities further along the flow path.
Figure 5. CE-SSCP Analyses of the Microbial Communities at Four Locations Along the Length of a Soil Column. The chemical mixture entered the column at the 0 cm point.
Combined Lumped Chemical Modeling
Since sorption and biodegradation led to different lumping schemes, and thus pseudocompounds of different compositions, their combination in a fate and transport model required the development of a cyclical algorithm for solution of the concentration profiles as a function of distance and time (Figure 6). A one-dimensional abiotic transport equation with sorption of the pseudocompounds described by the Freundlich isotherm was solved using a modified Crank-Nicholson algorithm. The kinetics were modeled using the sum kinetics with interaction parameters model, which was solved using the Runge-Kutta fourth order algorithm. The chemical composition at a particular point in time was determined first by solution of the biodegradation equation (with biodegradation pseudocompounds) and then a re-lump was performed before the transport equation (with sorption pseudocompounds) was solved. This algorithm was implemented using Matlab. Inputs to the model consist of the variable parameters for both transport and kinetics and the information on which chemicals mapped together as psuedocompounds in each portion of the model.
Figure 6. Schematic of the Algorithm for Solution of the Combined Lumped Biodegradation-Transport Model. Inputs and outputs are chemical concentrations at adjacent locations.
Chemical Clustering for Fate and Transport Modeling of Mixtures
The results of this project demonstrate that chemical clustering can simplify the modeling effort without a significant loss of accuracy. Since model parameters are typically obtained through experimental observation (or field measurements) and fitting of the model to those data, clustering leads to a large savings in the number of observations required to calibrate the model. For example, if one considers the case of 50 chemicals and a fate and transport model containing two parameters for sorption, two for biodegradation, and two interaction parameters, then the non-lumped model would include 4,500 parameters that would require 2,552 experiments to determine (without considering replicate experiments). In contrast, if the 50 chemicals could be grouped into 10 pseudocompounds, there would be a total of only 220 parameters and a minimum of 112 experiments. If the system were simplified to 5 pseudocompounds, the model would contain only 60 parameters requiring a minimum of 32 experiments to determine.
Conclusions:
The significant findings of this research are:
- Mixture effects occur for biodegradation of organic chemicals.
- Mixture effects occur for sorption of polar organic molecules.
- Chemical clustering to form pseudocompounds can be accomplished in a rigorous statistical manner, leading to simpler models that retain accuracy.
Expected Results:
This project will demonstrate the effectiveness and utility of the lumped chemical approach for contaminant transport and fate modeling, a potentially powerful tool that would improve risk assessment and facilitate risk management. In addition, our work will suggest useful strategies for forming pseudocompounds and for model development in an imperfectly lumpable system. This project will produce knowledge on the changes in a microbial community with time during batch biodegradation and distance in soil column tests, as well as knowledge about mixture effects on sorption and transport of organics and the impact of soil composition on those effects.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 3 publications | 3 publications in selected types | All 3 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Jon J, Schackelford C, Reardon K. Lumping Analysis for Sorption of Neutral Organic Compounds in Mixtures to Simulated Aquifer Sorbents. JOURNAL OF ENVIRONMENTAL ENGINEERING-ASCE 2012;138(5):552-561. |
R829355 (Final) |
Exit |
|
|
Jon J, Song M, Kim J. Sorption of nonpolar neutral organic compounds to low-surface-area metal hydroxide-and humic acid-coated model aquifer sands. JOURNAL OF ENVIRONMENTAL SCIENCE AND HEALTH PART A-TOXIC/hazardous SUBSTANCES & ENVIRONMENTAL ENGINEERING 2012;47(6):909-918. |
R829355 (Final) |
Exit Exit |
|
|
Jon J, Shackelford C, Readdon K, Lee W. Electromagnetically-vibrated solid-phase microextraction for analysis of aqueous-miscible organic compound transport in soil columns. CHEMOSPHERE 2021;263(127941). |
R829355 (Final) |
Exit Exit |
Supplemental Keywords:
aquifer, biodegradation, bioremediation, chemical lumping analysis, chemical mixtures, chemical pollutants, environmental microbiology, humic acid, microbial ecology, molecular biology, soil organic matter, sorption,, RFA, Scientific Discipline, Ecosystem Protection/Environmental Exposure & Risk, Waste, Hazardous, Ecological Risk Assessment, Fate & Transport, chemical mixtures, Environmental Chemistry, Hazardous Waste, Ecology and Ecosystems, environmental transport and fate, marine mammals, fate and transport , fate and transport modeling, biodegradation, complex mixtures, fate and transport, lumped chemical approach, organic pollutant mixtures, contaminated soils, environmental chemicals, exposure assessmentProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.