Grantee Research Project Results
Final Report: An Investigation Into HONO Formation Mechanisms in the Troposphere
EPA Grant Number: R826182Title: An Investigation Into HONO Formation Mechanisms in the Troposphere
Investigators: Zhou, Xianliang
Institution: New York State Department of Health
EPA Project Officer: Hahn, Intaek
Project Period: December 1, 1997 through November 30, 1999 (Extended to November 30, 2000)
Project Amount: $162,728
RFA: Exploratory Research - Environmental Chemistry (1997) RFA Text | Recipients Lists
Research Category: Water , Air , Safer Chemicals , Land and Waste Management
Objective:
The objective of this research was to investigate reactions leading to nitrous acid ("HONO" in gas phase and "HNO2" in liquid phase) formation, and thus to provide insights into the mechanisms for HONO formation in the troposphere under both NOx-rich and NOx-limiting conditions. Two types of reactions are to be investigated in the project as potential HONO sources in the troposphere, i.e., nitric acid reduction by various reducing agents in liquid phase and heterogeneous NO/NO2-H2O reactions on various surfaces.
Summary/Accomplishments (Outputs/Outcomes):
HNO3-HCHO System
The nitrous acid formation via HNO3 reduction by HCHO, reaction (1), was studied at various acidities:
NO3- + HCHO → HNO2 + HCOOH
At the acidity encountered in cloud water or fog water, i.e., in reaction media containing ~mM levels of H2SO4, the reaction was found too slow for the HNO2 production to be detected in laboratory using our technique. The HNO2 production rate was estimated to be #10-8 M/hour in the reaction medium containing 10 mM of NO3-, HCHO, and H2SO4, and ~10 mM initial concentration of HNO2, and will be several orders of magnitude lower than 10-8 M/hour via this reaction in cloud/fog waters. Therefore, the HNO3 reduction by HCHO in cloud/fog water is not a significant HONO source in the atmosphere.
In 1.5-11.5 M H2SO4 reaction media, the acidity encountered in atmospheric liquid aerosols, the reaction of HNO3-HCHO leads to significant production of HNO2. The production rate of nitrous acid was found to be first order towards HNO2 and HCHO. The dependence of the reaction on HNO2 is a characteristic of autocatalysis. The dependence of this reaction on acidity and HNO3 was more complex. An averaged reaction order of 1.3 was found towards Hammett functions (H0) of sulfuric acid in the concentration range of 1.5-8 M. The reaction slowed down with sulfuric acid concentration higher than 9.0 M (see Table 1). This may be due to the change of the existing form of nitrous acid in concentrated H2SO4 media. The reaction order towards HNO3 varied with H2SO4 concentration. In lower concentration of H2SO4, the reaction rate increases with increase of HNO3. In high H2SO4 concentration media, however, zero order reaction is observed towards HNO3. An apparent activation energy of this reaction was calculated to be 60 kJ mol-1 from the temperature dependence experiments.
| [H2SO4]/M | -log(H0) | k (M-1 s-1) | log(k) |
| 1.5 | 0.56 | 0.000222 | -3.6536 |
| 2.5 | 1.12 | 0.00125 | -2.9031 |
| 3.5 | 1.62 | 0.00624 | -2.2048 |
| 4.5 | 2.06 | 0.0224 | -1.6498 |
| 6 | 2.76 | 0.34 | -0.46852 |
| 6.5 | 3.03 | 0.43 | -0.36653 |
| 8 | 3.87 | 5.6 | 0.74819 |
| 9 | 4.4 | 8.5 | 0.92942 |
| 11.5 | 5.65 | 3.4 | 0.53148 |
That is, NO2 and/or N2O4 were assumed to be a reaction intermediate. We carried out experiments to examine the validity of this mechanism using a gas-liquid reactor, where NO2 (and N2O4, due to their fast equilibration) was effectively purged out by bubbling N2 through the reaction media. It was found that the reaction proceeded at the same rate in the gas-liquid reactor as in the static liquid reactors. This result casts a doubt on NO2 and/or N2O4 as an intermediate during the reduction of HNO3 by HCHO, as described by reaction equations (2) and (3). However, we are yet to find a reaction mechanism for the observed reaction orders.
We estimated the conversion rate of HNO3 to HNO2 up to 10 mM hour-1 in the liquid aerosols containing 60 percent H2SO4, assuming a steady state concentration of [HCHO] = 10 mM and [HNO2] = 5 mM. From the above results, it could be concluded that in the troposphere, the reduction of nitric acid by formaldehyde may proceed too slowly to be a significant mechanism for the formation of nitrous acid. In the stratosphere where the concentration of sulfuric acid in the aerosol can be as high as 11.5 M (70 percent weight), this reaction could be important for the formation of nitrous acid from nitric acid.
HNO3-Br- System
Similar to the reaction with HCHO, the reduction of HNO3 by Br- was found too slow for the HNO2 production to be detected in the laboratory using our technique, at the acidity encountered in cloud water or fog water, i.e., in reaction media containing ~mM levels of H2SO4.
In more concentrated H2SO4 media, HNO3 is readily reduced by bromide to produce HNO2:
HCO3 + 2H+ + 2 BΓ → HNO2 + H2O + Br2
The reaction was again found to be auto-catalytic towards HONO. The production of HONO was found to be 1.5-order towards HONO, first order towards bromide, and 0.5-order towards [HNO3]. That is, the production rate of HONO can be expressed as:
d[HONO]/dt = k(H+)[HNO3]1/2[BΓ][HONO]3/2 = kr[HONO]3/2[BΓ]
where k(H+) is a function of acidity, i.e., second order towards Hammett functions (H0) of H2SO4. The results were summarized in Table 2. In the 8 M H2SO4 reaction medium, an apparent activation energy of 70 kJ mol-1 was determined.
| [H2SO4]/M | -log(H0) | k in M-2s-1 | log(k) |
| 4.0 | 1.85 | 2.3 | 0.36173 |
| 5.0 | 2.28 | 30.5 | 1.4843 |
| 6.0 | 2.76 | 378 | 2.5775 |
| 7.0 | 3.32 | 5500 | 3.7404 |
Experiments using a gas-liquid reactor also have been carried out. Similar to the reaction with HCHO, the reduction of HNO3 by Br- proceeded at the same rate in the gas-liquid reactor as in the static liquid reactor, suggesting that NO2 is not likely to be the intermediate of the reaction. We are yet to find a reaction mechanism to explain the complex reaction orders of this reaction..
HNO3-SO2 System
SO2 was found to be an effective reducing agent for HNO2 as well as HNO3. The reaction rate between HNO2 and SO2 was found to increase rapidly with acidity in the reaction medium. In acidic reaction media containing mM - M H2SO4, the consumption rate of HNO2 via its reaction with mM levels of SO2 was much higher than its production rate via HNO3 reduction, with a HNO2 lifetime of < 60 minutes. Therefore, the presence of SO2 in atmospheric waters and liquid aerosols may be a sink rather than a source for HONO.
Reaction of NO2 with Liquid Water
Experiments have been carried out to study the reaction of NO2 with liquid water (1 mM H2SO4) using a gas-liquid reactor:
2NO2 + H2O(1) + NHO2 + HNO3
The surfaces include glass, NaCl coating on glass, an oil-based enamel, and high-density polyethylene (HDPE). We also have investigated the role of NO in HONO formations on glass surface via:
d[HNO2]/dt = k p(no2)
k = 2.8 x 10-4 M atm-1 s-1
This result is not in agreement with that of Lee and Schwartz (Journal of Physical Chemistry 1981;85:840-848) that the reaction (7) was second order towards NO2 at higher NO2 concentrations in their experiments.
Heterogeneous Reaction of NO2 with H2O(g)
We have studied the heterogeneous reaction between NO2 and H2O vapor on surfaces:
2NO2 + H2O + surface → HNO2 + HNO3 + surface
The surfaces include glass, NaCl coating on glass, an oil-based enamel, and high-density polyethylene (HDPE). We also have investigated the role of NO in HONO formations on glass surface via:
NO + NO2 + H2O + surface → 2HNO2 + surface
We found that NO addition to the reactor containing NO2 and H2O did not significantly affect the HONO production rate, within our experimental precision of ~10 percent.
Figure 1 is a typical time series of HONO concentration in the effluent gas from a 12-L glass reactor. The experiment began with the measurement of blank through the reactor when no NO2 and water vapor were introduced into the glass reactor. Water vapor and NO2 were then introduced and the production of HONO was measured. The formation of HONO was almost instantaneous upon introducing NO2 and water vapor into the reactor. HONO concentration in the effluent gas from the glass reactor increased rapidly to a maximum followed by decay to a relatively stable state. The decay was caused by the poisoning of the bare glass surface by the produced HNO3 adsorbed on the surface. We have tried to measure the rate constants using the initial HONO concentration right after NO2 and H2O addition, but the data obtained were not very reproducible, highly dependent on the way the reactor surface was pretreated and conditioned. Therefore, we avoided using the beginning portion of the data for rate constant calculation, and used the only data within relatively short and stable periods after long-period conditioning of the glass surface.
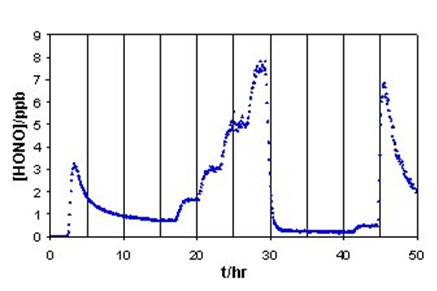 Figure 1. The change of [HONO] in effluent gas from a 12-L glass reactor. Temperature: 24±1 oC; Flow rate: 1 L min-1 |
|
On other surfaces studied, including NaCl coating on glass, an oil-based enamel coating on glass, and high-density polyethylene, we did not see the sharp initial increase followed by decay of HONO production (Figure 2 as an example), as on the bare glass surface (Figure 1), when NO2 and water vapor were introduced into the reactor.
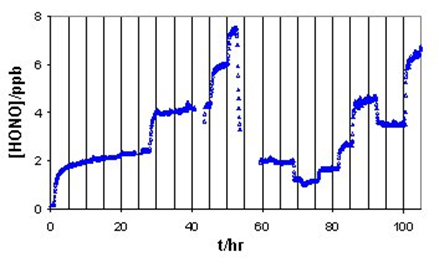 Figure 2. The change of [HONO] in effluent gas from a 12-L enamel-coated glass reactor. Temperature: 24±1 oC. |
|
Figure 2
Results are summarized in Table 3. The reaction order of reaction (10) is 1 towards NO2. However, its reaction order towards H2O is difficult to determine, especially on bare glass surface, as H2O concentration changes in the reaction would cause big disturbances in the HONO production rate (Figure 1). The HONO production rates obtained are within the range of values reported in the literature. The rate constant of reaction (10), k or k', is defined by (12):
P = k[NO2][H2O]m S = k'[NO2]S
where P is the HONO production rate from the reactor (moles/s), [NO2] and [H2O] the mixing ratios of NO2and H2O, m is the reaction order towards H2O in the range of 0-2, S is the surface area of the reactor, and k' = k[H2O]m.
| Surface | k' moles m-2 s-1 | Reaction order NO2 | Reaction order H2O
| Production rate ppb (ppb NO2)-1 hr-1 |
| Glass | 5 x 10-6 | 1-1.4 a | 0.2-1.5 a | 4 x 10-4 |
| Enamel Coating | 1.2 x 10-5 | 1.0 | ~1 | 1 x 10-3 |
| HDPE | 4 x 10-5 | 1.0 | ~1 | 3 x 10-3 |
| NaCl coating | 1.2 x 10-6 | 1.0 | ~1 | 1 x 10-4 |
a A large increase in HONO production followed by steady decrease were observed when H2O concentration was changed in the reactor. The determined reaction rate dependence and thus the reaction order were therefore highly variable, depending on where the data were used for the calculation.
It is obvious that HONO production rate on different surfaces varied significantly, in the following order: HDPE > enamel coating > glass > NaCl coating. It should be pointed out that the rate constant calculation has not taken the roughness of the reactor surfaces into consideration. Glass surface may be the smoothest and the NaCl the roughest. The reaction rate on the NaCl coating thus may be significantly reduced if a larger surface area of the reactor is used in the calculation, suggesting that the presence of salt on surface may have a poisoning effect on reaction (10).
Uncertainties of the Data Reported. The uncertainty associated with measurement method itself was relatively small, better than ?15 percent for the concentrations encountered in the experiments. Major uncertainties were associated with: (1) sample transfer/delivery during the experiments studying HNO3 reduction in concentrated H2SO4 media at low temperature, and (2) reactor surface poisoning during the experiments studying heterogeneous NO2-H2O reaction. For the HNO3 reduction rate constants in concentrated H2SO4 media, a relative standard deviation (RSD) of ?30 percent was estimated at experimental temperature higher than 0oC and of about ?50 percent at experimental temperature lower than 0oC. For the heterogeneous NO2-H2O reaction rate constants, the reproducibility was better than a factor four for glass surface and was better than ?70 percent for other surfaces.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 3 publications | 2 publications in selected types | All 2 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Dai H, Zhou X. HONO formation via heterogeneous reaction of NO2 and H2O on various surfaces. Journal of Physical Chemistry. |
R826182 (Final) |
not available |
|
|
Dai H, Zhou X. Reduction of nitric acid by bromide in sulfuric acid. Journal of Physical Chemistry. |
R826182 (Final) |
not available |
Supplemental Keywords:
air, ambient air, atmosphere, tropospheric, NO2, NOx, H2O, heterogeneous reaction, surfaces, HNO3 reduction, formaldehyde, bromide, HONO source., Air, Scientific Discipline, Chemistry, Environmental Chemistry, Engineering, Chemistry, & Physics, gas-liquid reactor, photooxidants, HONO, chemical composition, troposphere, reaction kinetics, chemical kinetics, ozone, nitrous acid formationProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
