Grantee Research Project Results
Final Report: Hydrothermal/Thermal Decomposition of Perchlorate
EPA Contract Number: 68D99032Title: Hydrothermal/Thermal Decomposition of Perchlorate
Investigators: Li, Lixiong , Coppola, Edward N.
Small Business: Applied Research Associates Inc.
EPA Contact:
Phase: I
Project Period: September 1, 1999 through March 1, 2000
Project Amount: $69,978
RFA: Small Business Innovation Research (SBIR) - Phase I (1999) RFA Text | Recipients Lists
Research Category: Watersheds , SBIR - Water and Wastewater , Small Business Innovation Research (SBIR)
Description:
The purpose of this SBIR Phase I project was to demonstrate near complete decomposition of perchlorate ions in a 7% sodium chloride (NaCl) brine solution via non-catalytic hydrothermal (high pressure) and thermal (low pressure) treatment approaches. A secondary purpose was to demonstrate a process concept to reject sulfate and permit multiple reuses of the brine solution.
The ion exchange process has shown great potential for removing perchlorate from contaminated drinking water. A 7% NaCl brine (~1.25 molar) is used to regenerate ion exchange resin from the treatment of perchlorate contaminated groundwater. A major operating cost for the ion exchange process is the disposal of spent brine regenerating solution. Modern ion exchange processes, such as Calgon Carbon Corporation's ISEP process, have minimized brine generation to 1% or less of the treated groundwater for typical groundwater contamination. Brine disposal cost of $80 per 1000 gallon and salt cost of $30 per 1000 gallon equates to a distributed cost of over $350 per acre foot of water treated (based on a 1% rejection rate). Therefore, the thermal and hydrothermal processes were conceived to recover and reuse brine to make ion exchange more efficient and cost-competitive with typical drinking water production costs.
A surrogate ion exchange brine was used in this study. The brine was prepared using water softener salt, and contained anticipated concentrations of sulfate, nitrate, bicarbonate, and perchlorate, shown in the table below, that could result from the ion exchange treatment of perchlorate-contaminated groundwater. Decomposition testing was conducted in a continuous-flow reactor system and in a batch apparatus. Key variables included temperature, pressure, and reaction time. Reducing agents and promoters were used to enhance perchlorate decomposition. In addition, exploratory tests were conducted to evaluate the feasibility of removing perchlorate from the spent salt brine via an adsorptive/desorptive approach.
| Components | Concentration |
| Water softener salt | (99.5% NaCl + impurities) 7 wt % |
| Sodium Nitrate (NaNO3) | 800 mg/l (as NO3-) |
| Sodium Sulfate (Na2SO4) | 3000 mg/l (as SO4=) |
| Sodium Bicarbonate (NaHCO3) | 200 mg/l (as CO3=) |
| Sodium Perchlorate | 50 mg/l (as ClO4-) |
Summary/Accomplishments (Outputs/Outcomes):
Complete destruction of perchlorate ions was achieved by thermal decomposition. The presence of ferric or ferrous salts reduced the decomposition temperature to 170oC. Without the addition of promoting agents, complete thermal decomposition of perchlorate was obtained at 600oC. Sulfate ions were effectively precipitated and removed from the salt brine by the addition of barium chloride or calcium chloride.
Complete destruction of perchlorate could be obtained in the aqueous phase (hydrothermal) at 340oC when 7% ferric chloride was substituted for sodium chloride. Only partial destruction of perchlorate could be obtained by the additions of ferric salts to sodium chloride brine. Essentially no perchlorate reduction was obtained with or without other promoting agents up to the temperature limitation of the hydrothermal process (380oC).
Carbon, metal oxide, clay, and polymer sorption agents were also evaluated as a means to concentrate perchlorate and enhance the performance of thermal decomposition approaches. These tests did not result in an agent that could remove perchlorate to the extent necessary for practical application.
Based on these findings, ARA developed an Integrated Thermal Treatment Process that can meet all requirements for regenerating the salt brine from the ion exchange process. The figure shown on page 3 is a simplified block diagram of the overall treatment scheme in which contaminated groundwater is first treated by ion exchange. The spent brine used to regenerate the ion exchange resin can be less than 1% of the volume of the treated groundwater. This brine is then concentrated by reverse osmosis (RO) by a factor of 2-3 to further reduce the volume requiring thermal treatment and improve process economics. The concentrated brine then enters the Integrated Thermal Treatment Process, in which nitrate and sulfate salts are rejected from the brine in effluent streams, while the perchlorate is thermally destroyed, effectively regenerating the brine. The regenerated brine can then be recycled for reuse in the ion exchange process, greatly reducing the costs associated with spent brine disposal and salt replacement. While some NaCl loss in the effluent streams is inevitable, the effluent volume will only be 1-5% of the original brine. Therefore, brine disposal and replacement costs can be reduced 95-99%.
Figure 1. Block Diagram of the Overall Groundwater Treatment Scheme
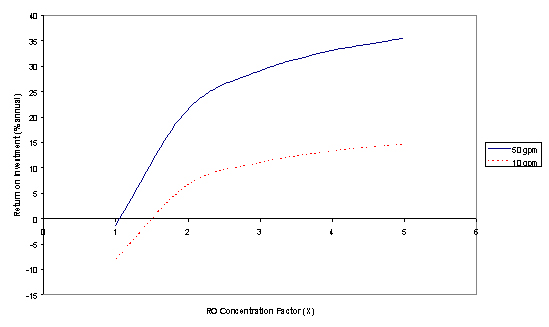
Preliminary heat and mass balance calculations were conducted as a function of the RO concentration factor, X. A concentration factor of X = 1 corresponds to no pre-concentration, while X = 5 corresponds to five-fold reduction of the incoming water volume.
In these calculations, a basis of 50 gal/min (GPM) of 6.5 wt% brine was chosen (5000 GPM groundwater treatment plant). The heat recovery in the evaporator effects was assumed to be 50% and the boiler efficiency was assumed to be 50%. Boiling point elevation in both evaporators were neglected. Using these constraints, the capacities and heat duties of the unit operations were obtained. Literature values (M.S. Peters and K.D. Timmerhaus, "Plant Design and Economics for Chemical Engineers", McGraw-Hill Inc., 4th ed., 1991) were then used to calculate installed costs of equipment based on unit operation capacities. Energy costs were calculated from equipment heat duties based on energy cost of $3.0/MBTU. Capital costs were calculated based on annual interest rate of 12%, maintenance cost based on 11% of fixed-capital annually, and straight-line depreciation based on 15-year equipment service life. The bases for these calculations are very conservative and, therefore, much lower costs and higher return on investment are possible. All of these costs were then distributed as cost per 1000 gallons of groundwater treated based on an ion-exchange brine rejection rate of 1.0%. Depending on the perchlorate concentration in the groundwater, brine rejection rates can actually be lower than 1% in the Calgon Carbon ISEP process.
The figure below shows the estimated annual return on investment obtained from the Integrated Thermal Treatment Process as compared to brine disposal and salt replacement at a combined cost of $100/Kgal. Conventional seawater desalination RO processes operate by concentrating the brine by a factor of about 1.7. Minor modifications to an off-the-shelf RO unit could provide a concentration factor of 2 to 3, corresponding to an annual return on investment of 20-30%. The heat and mass balances and return-on-investment calculations were repeated using a basis of 10 GPM 6.5 wt% brine (1000 GPM groundwater treatment process). As shown in the figure, the return on investment for the smaller-scale process is greater than 10% for a concentration factor of 2 to 3.
Figure 2. Double Effect Evaporator Plant for Treatment of CI O4-Contaminated Brine Return
on Investment for 10gpm versus 50 gpm plant capacity.
Conclusions:
Results derived from these tests show that the spent salt brine can be regenerated for reuse in the ion exchange process. Based on these findings, ARA has developed an Integrated Thermal Treatment Process that can meet or exceed end-user needs in terms of performance and cost.
Supplemental Keywords:
Drinking water treatment, perchlorate decomposition, brine regeneration., RFA, Scientific Discipline, Toxics, Water, Geographic Area, Waste, Chemistry, Contaminant Candidate List, Engineering, Drinking Water, Engineering, Chemistry, & Physics, West Coast, Groundwater remediation, hydrothermal decomposition, industrial wastewater, nitrates, perchlorate, hydrothermal decompostion, treatment, drinking water treatment, water treatment, other - risk management, ion exchangeThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.