Grantee Research Project Results
Final Report: Microwave-Catalytic Membrane for PFAS Degradation
EPA Grant Number: SU840150Title: Microwave-Catalytic Membrane for PFAS Degradation
Investigators: Zhang, Wen , Liu, Fangzhou , Sai, Krishna Venkata , Potts, Courtney , Luk, Jeffrey , Solomon, Samuel , Kokkula, Ashish , Abdali, Humza
Institution: New Jersey Institute of Technology
EPA Project Officer: Page, Angela
Phase: I
Project Period: December 1, 2020 through November 30, 2021
Project Amount: $25,000
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2020) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
Traditional membrane filtration technology suffers from membrane fouling due to cake layer accumulation, concentration polarization or pores clogging by particles, colloids, macromolecules, or organic matters. Moreover, removal of trace-level, small-molecular sized or recalcitrant organic pollutants via membrane filtration is also a long-unresolved challenge. Membrane filtration processes that exhibit robust and stable filtration with antifouling and reaction-enabled functions are urgently needed for efficient water treatment or chemical separation. This project embarks on a novel microwave-assisted membrane filtration process that is designed to improve filtration performance, enhance pollutant degradation and mitigate membrane fouling. Under microwave irradiation, the filtration process may also involve the production of nanobubbles and hydroxyl radicals on membrane surface, where engineered microwave-responsible catalysts are coated. Functionalized membrane fabrication, stability, reactivity, and fundamental mechanisms such as how microwave irradiation promotes formation of nanobubbles and radicals, pollutant degradation efficiency, and antifouling performances will be rigorously investigated. The ultimate goals are to transform passive membrane filtration to the next-generation reactive membranes that can proactively degrade water contaminants and prevent surface fouling, and to enable novel membrane processes in chemical conversion and separation.
This project aims to develop reactive, durable, and sustainable membrane systems that proactively degrade refractory water contaminants and prevent surface fouling with focuses on fundamental studies of interfacial engineering of functional catalyst coating on ceramic membranes. The scientific merit from this project include: (1) providing optimized fabrication processes for catalyst-coated ceramic membranes with tunable structures such as catalyst coating density, layer thickness and porosity to maximize filtration flux and stability (2) quantifying efficacy of antifouling and degradation performances of microwave-assisted filtration under optimized design of catalyst layer thickness, membrane porosity and microwave irradiation. (3) unraveling mechanisms and roles of microwave assisted Fenton-like reactions and nanobubbles/radicals formation in pollutant degradation and resistance toward fouling; For example, kinetics of radical formation under microwave irradiation will be thoroughly assessed by novel approaches (e.g., electrochemical activity measurement coupled with scanning probe microscope) to understand molecular level mechanisms of microwave catalysis and impacts on pollutant degradation and foulant removal on membrane interfaces. (4) supporting rational designs of microwave-enabled reactive membrane systems to synergize catalytical reactions and membrane filtration.
Summary/Accomplishments (Outputs/Outcomes):
1. Preparation and characterization of BiFeO3 (BFO) coated membrane
The morphologies of pristine membrane, low BFO coated membrane (1.6 µg∙cm-2 ) and heavy BFO coated membrane (2.7 µg∙cm-2) are prepared and characterized by SEM, EDX and AFM. The SEM showed the structure of BFO coated membrane in cross-sectional images, consisting of α-Al2O3 supporter, Zr/TiO2 layer and BFO coated layer. EDX analysis proved that the ceramic membranes have been coated with BFO particles on the membrane surface. Comparing the AFM imaging and the root mean square (RMS) roughness (Rq) of low coated membrane, the heavy coated membrane presents a denser catalysts distribution. Furthermore, Rq only report z-direction roughness, which does not reveal the lateral heterogeneity of the roughness distribution. The one-dimensional Power Spectral Density (PSD) was employed to determine the fractal dimensions. The results showed that the pristine membrane’s surface structure is close to 2D, whereas the two catalyst-coated membrane have a 3D surface structure.
2. Filtration flux and membrane stability assessment
The water permeability of pristine and coated membrane indicated that membrane pore blocking by catalyst particles was not detrimental (reduced by 14%-22% compared to the pristine membrane). Then, the impacts of catalyst coating and microwave irradiation (solution temperature) on membrane permeability were carefully examined and interpreted by the Carman-Kozeny, Hagen-Posieulle models, which assume that the flux pathway is similar with capillary pores, slit-like pores and cylindrical pores, respectively. The result showed that the Carman-Kozeny model prediction well matched the experimental measurement of the permeability of feed solution under different temperatures, suggesting the enhanced permeation under microwave irradiation resulted from the water viscosity changes. It is reported that Carman-Kozeny and Hagen-Posieulle model can both explain the experimental data when the membrane pore sizes are 500 and 800 nm 1. However, the variation of water flux on ceramic membrane with a pore size of 100 nm was better predicted by the Carman-Kozeny model.
3. Removal efficiency of persistent organic foulants
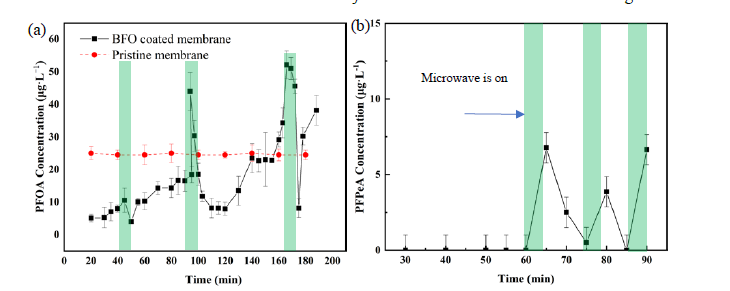
Two ceramic membranes with different BFO coating densities (1.6 and 2.7 µg∙cm-2) were used in a dead-end filtration mode with the feed solution passing through the membrane. the PFOA (50 µg∙L-1) and H2O2 (30 mM) solutions were mixed in a volume ratio of 1:1 at a tee, then pumped to the membrane filtration system with modified syringe pump at different flow rates (1.25-7 mL·min). After the microwave irradiation for 5 min as indicated by the green bar, the PFOA concentration was rapidly reduced, as the microwave-assisted Fenton-like reaction on BFO-coated membrane may enable the PFOA degradation (Fig.1a). The microwave-assisted Fenton-like reaction contributed to approximately 65.9% of PFOA that was removed this reactive membrane filtration. Furthermore, the PFPeA have been detected in the outlet samples using established PFPeA calibration curves with LC-QQQ/MS. The increased concentration of PFPeA under the microwave irradiation indicated that PFPeA may be the main intermediate of PFOA degradation a) PFOA concentration changes in three consecutive round of filtration tests by using pristine and BFO coated membrane. (b) the PFPeA concentration change with filtration time The green bars indicate the operation of microwave irradiation during filtration, whereas other areas were operated without microwave irradiation and under filtration only. The initial spiked PFOA concentration: 25 µg∙L-1; the microwave intensity: 125 watts (7.2 watt·cm-2); the catalyst coating density: 2.7 µg∙cm-2; and the permeate flux: 43 LMH.
Fig. 1 (a) PFOA concentration changes in three consecutive round of filtration tests by using
pristine and BFO coated membrane. (b) The PFPeA concentration change with filtration time. The
green bars indicate the operation of microwave irradiation during filtration, whereas other areas
were operated without microwave irradiation and under filtration only. The initial spiked PFOA
concentration: 25 μg∙L-1; the microwave intensity: 125 watts (7.2 watt·cm-2); the catalyst coating
density: 2.7 μg∙cm-2; and the permeate flux: 43 LMH.
4. Detection of nanobubbles and radicals on catalyst-coated membranes under microwave irradiation
The sample was measured with Dynamic Light Scattering (DLS) to get the size distribution and zeta potential of nanobubbles with/without the microwave irradiation. The result shows that without the microwave irradiation, no positive signals were detected as there was only one peak at 10 nm or smaller with a very low zeta potential value of -3.7 mV, which might be attributed to the instrumental noise. After the microwave irradiation, bubbles of different sizes were detected as indicated by the multiple peaks that randomly involved. Some bubbles were in greater sizes like 1 µm or larger, which were clearly micro-bubbles, whereas some others were of tens to hundreds of nanometers.
The formation of hydroxyl radicals (•OH) on the surface of BFO-coated membranes was detected by a photoluminescence (PL) technique with terephthalic acid as a probe molecule. The result shows PL peak present in BFO+H2O2 and H2O2 samples under microwave irradiation, while no obvious PL increase was observed in the absence of H2O2 samples. This suggests that the fluorescence is caused by chemical reactions of terephthalic acid with •OH formed by H2O2.
Conclusions:
This EPA P3 phase I research has fabricated the BiFeO3 catalyst and successfully coated the ceramic membrane surface, which was characterized to reveal the changes of physiochemical and hydrodynamic properties, such as roughness and fractal dimension of different coating densities and permeability under different temperature. High catalyst coating densities on membrane surface likely reduce permeability and adsorb PFOA, which will leach out without sufficient degradation. By contrast, moderate coating (1.6 µg∙cm-2) and low hydraulic retention time (2 min) enabled effective degradation of refractory PFOA through the presented microwave-assisted membrane filtration. The degradation by-product analysis indicated that the mineralization of PFOA was not complete as significant levels of PFPeA were detected. The nanobubble and radicals are detected for the analysis of PFOA degradation mechanism. However, the current research on wastewater treatment using MW-assisted catalysis is still in the development stage, and the technology and equipment are not completely mature. In especial, there has always been a dispute between the thermal effect and the nonthermal effect. The thermal effect of MW has been recognized by most researchers. However, some researchers believe the existence of the nonthermal effect. Therefore, future research should focus on a complete understanding of the interactions between MW and catalysts, the exploitation of high efficiency catalysts and the optimization of technology and equipment, which can favour the further development of MW-assisted catalytic technology for application to wastewater treatment.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 1 publications | 1 publications in selected types | All 1 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Liu F, Rittmann B, Kuthari S, Zhang W. Viral inactivation using microwave-enhanced membrane filtration. Journal of Hazardous Materials 2023;458: 131966. |
SU840150 (Final) SV840419 (Final) |
Exit |
Supplemental Keywords:
Microwave responsive catalyst, Functional membrane reaction, PFAS, Microwave penetration, Microwave catalysis, Sustainable water treatment, Water disinfectionRelevant Websites:
Microwave Catalytic Membrane for PFAS Degradation Exit
P3 Phase II:
Microwave-Catalytic Membrane for PFAS Degradation and Antiviral Applications | 2023 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.