Grantee Research Project Results
Final Report: Biofilters to Effectively Remove Toxins
EPA Grant Number: SU839962Title: Biofilters to Effectively Remove Toxins
Investigators: Barua, Sutapa , Wang, Jee-Ching , Razdan, Sidharth , Adler, Justin , Barua, Dibbya
Institution: Missouri University of Science and Technology
EPA Project Officer: Page, Angela
Phase: I
Project Period: October 1, 2019 through September 30, 2020 (Extended to June 30, 2021)
Project Amount: $24,997
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2019) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
This EPA P3 phase I project addresses a fundamental problem of adequate access to clean water by developing a portable, low-cost and high performance biofilter to remove freshwater harmful algal bloom (FHAB) derived cyanotoxins and heavy metal ions from water. The goal is to deliver clean and safe water by flowingcontaminated waterthrough biofilters that yield enhanced toxin removal performance and lead to sustainable access of safe water. Thegoalwas supported bythree specific objectives as detailed below.
Objective 1:Measure cyanotoxin and heavy metal ion (toxin) concentrations, and understand the need for cyanotoxin and metal ion treatmentsin Missouri FHAB events
Objective 2: Develop an environment friendly natural soy protein derived cellulose filter embedded with biocompatible polymeric NPs for the removal of toxins from water
Objective 3: Investigate the effects of relevant factors as design parameters to optimize the cyanotoxin and metal ion removal efficiency using filters impregnated with NPs
Objective 4: Education -Form and advise a multidisciplinary engineering student team to assess the biofilter need and develop appropriate engineering specifications. Guide thestudent team through the entire design process
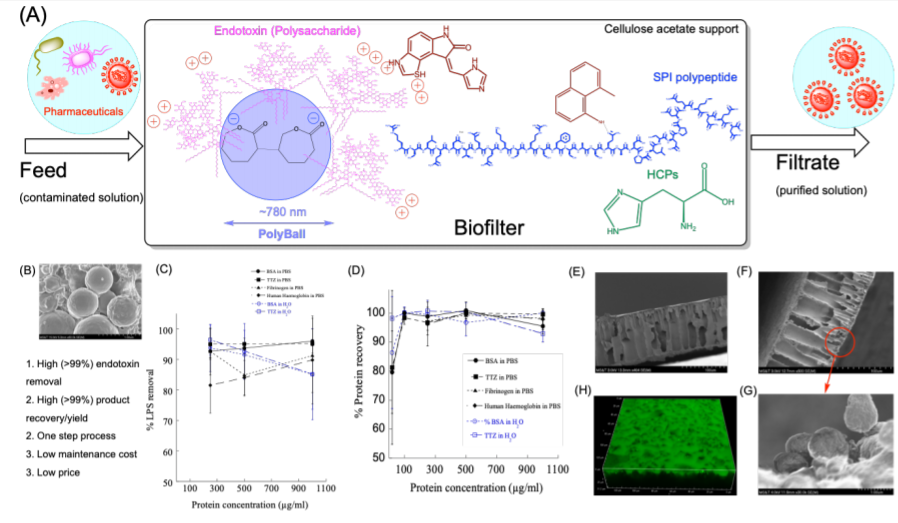
Figure 1: (A) Schematic showing biopharmaceutical solution purification using PolyBall particles in cellulose acetate and SPI Biofilters. PolyBalls selectively bind endotoxins via hydrophobic, van der Waals, and charge-charge interactions. The cellulose acetate acts as a support. Proteins and solutes smaller than filter pores permeate through the Biofilter, while larger solutes (contaminants) are retained. SPI contains polypeptides that bind other contaminants. (B) Scanning electron microscopy (SEM) image of the nanostructure of PolyBalls. (C) Endotoxin removal from protein solutions. Increasing protein concentrations have no significant effect on the % of endotoxin removal from protein solutions prepared in phosphate buffered saline (PBS) and water. Symbols • , o, and indicate endotoxin containing BSA solutions in PBS and water, and trastuzumab (TTZ) solutions in PBS and water, respectively. (D) % of protein recovery as a function of protein concentrations. SEM of cellulose acetate filters (E) without and (F) – (H) with PolyBall
Summary/Accomplishments (Outputs/Outcomes):
Removal of endotoxins using PolyBall nanoparticles:
We have synthesized a novel composite biofilter modified with cellulose acetate(CA), soy protein isolates (SPI), and PolyBall nanoparticles to remove endotoxins for the purification of therapeutic protein solutions and water(Fig. 1(A)). The process began with PolyBall particle synthesis and characterization of the particles using dynamic light scattering (DLS), SEM, and TEM.37,45,46PolyBalls were manufactured in the PI’s laboratory using a solvent/emulsion method which were spherical in shape and monodisperse (Fig. 1(B)). Particle size measurements showed that the NPs have an average diameter of 760±345nm(Fig. 1(B)).The surface charge (ζ−potential)of PolyBall alone was (−17±4)mV,suggesting that the particles were reasonably stable in suspension form and would resist aggregation. In general, the endotoxin removal efficiency using PolyBalls was >98%using1 mg/ml of PolyBallsin presence of salts. PolyBallsare incorporated in CA and SPI biofilters’ matrix to bind and remove endotoxin contaminants from water samples.PolyBalls were added into cellulose acetate and SPI mixture which interacted through the coordination of hydrogen bonding and created uniform biofilter pores. The permeability, porosity, selectivity, and toxin removal were studied. Fig.s 1(E), and1(F) –1 (G) represent the SEM images of a biofilter without PolyBalls, and with PolyBalls incorporated in it, respectively. The biofilters have a thickness of (117 ± 4) μmand (120 ± 3) μm in the absence and presence of PolyBalls, respectively. The ability of endotoxin removal using PolyBall nanoparticle powder and biofilters were investigated for protein and water purification. Protein solutions and water were spiked with E. coliO111:B4 endotoxins and passed through the biofilter. The amount of endotoxins in filtrate and retentate were measured using EndoDye assay.37,45,46Increasing protein concentrations from 250 to 1000 μg/ml did not alter the ~90% endotoxin removal efficacy by PCL NPs in PBS (Fig. 1(C)). Product (protein) recovery is as important as endotoxin removal to reflect an interaction of the protein with endotoxin binding sites. Fig. 1(D) shows the results of protein recovery at varying protein concentrations. As it is seen that protein recoveries were close to 100% for a wide range of protein concentrations(0 –1000 μg/ml). These results further confirm the selectivity of PCL NPs for endotoxinswhile showing 100% protein recovery.
Removal of endotoxins using biofilters:
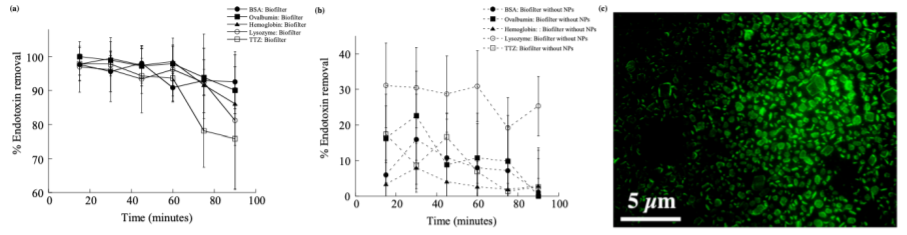
To confirm the adsorption capability of PCL NPs in a portable filter form for potential application in larger scale operations, the endotoxin removal efficiencies by the CA membranes with or without PCL NPs were measured and compared.The % of endotoxin removal kinetics were tested for the purification of 1000 μg/ml of each five individual protein solutions (BSA, hemoglobin, lysozyme, ovalbumin, and TTZ) from endotoxin contamination (5.74×10!EU/ml) using biofilters with and without PolyBalls (Fig. 2). The average % endotoxin removal efficiency using biofilters with PolyBalls varied between 95% (5.5×10!EU/ml)to 99% (5.7×10!EU/ml)within the first 60 min (Fig. 2(a)). After 90 min of incubation, the endotoxin removal efficiency decreased to ~90% for BSA, hemoglobin, and ovalbumin, to ~80% for lysozyme, and up to 75% for TTZ using biofilters with PolyBalls. Biofilters without PolyBalls did not remove more than 30% of endotoxins from protein solutions (Fig. 2(b)). Fluorescence microscopy was used to detect green layers of Alexa Fluor 488 labeled endotoxins on the surface of PolyBalls (Fig. 2(c)). The distribution of fluorescence indicated endotoxin binding around the PolyBall surface. These results reveal that PolyBall plays an important role in the binding of endotoxins onto the rigid NP surface. There is a time-dependent equilibrium of protein adsorption thatdo not exhibit significant variations of desorption from the surface of PolyBalls. After 60 min, the % endotoxin removal decreases slightly presumably due to the desorption of proteins by repulsive interactions after reaching equilibrium. The desorption was higher for an order of magnitude larger molecular weight TTZ than smaller size proteins due to higher repulsive interactions between proteins andthe PolyBall surface.
Figure 2: The endotoxin removal efficiency using biofilters (a) with and (b) without PolyBalls from BSA (●), ovalbumin (■), hemoglobin (▲), lysozyme (○), and TTZ (□) spiked endotoxin solutions in water. (c) Detection of Alexa Fluor 488 conjugated endotoxin binding on the surface of PolyBalls using fluorescence microscopy.
Table 1: Summary of the general conditions for each of the three water samples collected at the time of this report. Measurements for pH were made in the lab using a Fisher Scientific Accumet AE150 pH meter. Measurements were taken immediately after returning to the lab after sample collection. The pH was recorded to the nearest 0.01. The temperature was recorded at the listed location where the sample was originally obtained and was measured using a standard thermometer. Measurements were recorded to the nearest 0.5°C.
| Location | pH | Temperature (℃) |
| Ber-Juan | 6.80 ± 0.01 | 18.5 ± 0.12 |
| Lion’s Park | 7.16 ± 0.11 | 19.75 ± 0.25 |
| Schuman | 7.39 ± 0.20 | 28.25 ± 0.80 |
Table 2: The overall concentrations of contaminants determined to be in each evaluated water sample
| Toxin | Ber-Juan Park | Lion’s Park | Schuman Park | ||||||
| µg/mL | unfiltered | filtered | % removal | unfiltered | filtered | % removal | unfiltered | filtered | % removal |
| Anatoxin-α | 4.48 ± 0.08 | 3.98 ± 0.09 | 11.2 | 3.57 ± 0.08 | 3.14 ± 0.06 | 8.8 | 8.50 ± 0.24 | 7.30 ± 0.46 | 14.1 |
| Microcystin | 7.30 ± 0.05 | 5.74 ± 0.15 | 21.4 | 6.39 ± 0.18 | 5.83 ± 0.15 | 12.0 | 15.90 ± 0.40 | 13.04 ± 0.13 | 18 |
| Lead | 2.60 ± 1.0 | 0.82 ± 0.56 | 90.88 | 0.74 ± 0.40 | 0.31 ± 0.054 | 43.78 | 0.75 ± 0.36 0 | 0.39 ± 0.13 | 52.15 |
Lake water purification using biofilters:
We collected 12 local water samples from three lakes from October 2019 to February 2020. The water pH and temperature were measured at the time of sample collection (Table 1). Table 2 shows the overall concentrations of contaminants determined in each evaluated water sample. The highest anatoxin-aand microcystin concentrations were observed in the water collected from the Schuman park lake. Analysis of the surface water has shown concentrations of anatoxin-a ranging from (3.57 ± 0.08) to (8.50 ± 0.24) μg/ml that are above the WHO acceptable level of 0.1 –1.6 μg/ml.37. Microcystin was detected from (6.39 ± 0.18) to (15.90 ± 0.40) μg/ml. Lead concentration varied from (0.74 ± 0.40) to (2.60 ± 0.10) μg/ml. The biofilter removed ~10-20% of anatoxin-a, ~12-20% of microcystin, and ~40-90% of lead from water samples. The Pb2+ions were most likely removed by the presence of soy protein’s polypeptides containing carboxyl, hydroxyl, phenyl, and sulfhydryl functional groups that are effective metal chelators.58-61. The multiple binding sites of metal ions from the polypeptide backbone enhanced the metal adsorption capacity in a high surface area to volume ratio of the biofilter. The lead adsorption capacity was 115 ±20.8 mg lead per g of soy protein in the biofilter.
Conclusions:
In summary, SPI based CA biofilter is developed impregnating PolyBalls that remove >99% endotoxins from protein solutions in water and PBS with >99% protein recovery. The size of PolyBalls is ~760𝑛𝑚 in diameter by analyzing SEM images. The effectiveness of PolyBalls on removing endotoxinswas studied using five protein solutions of various molecular weights in water and PBS. It was clear and important to note that PolyBalls were effective in adsorbing and removing endotoxin from solutions regardless of the presence or absence of salts (PBS). Protein recovery is as important as endotoxin removal to reflect an interaction of the protein with endotoxin binding sites. As it is seen that protein recoveries using biofilters were close to 100% for a wide range of concentrations and pHs. These results further confirm the selectivity of PolyBalls for endotoxins while showing >99% endotoxin removal and >99% protein recovery.The selective high endotoxin removal efficiency with a large scale protein yield was due to hydrophobic-hydrophobic interactions of PolyBalls as well as partial charge-charge interactions and distributions of negatively charged phosphate groups and those in the inner core on endotoxins with solvent molecules, e.g., hydrogen ions (H+) of water and sodium ions (Na+) of PBS.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 3 publications | 2 publications in selected types | All 2 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Schneier M, Razdan S, Miller AM, Briceno ME, Barua S. Current technologies to endotoxin detection and removal for biopharmaceutical purification. Biotechnology and bioengineering 2020;117(8):2588-609. |
SU839962 (Final) |
Exit |
|
|
Razdan S, Adler J, Barua D, Barua S. Multifunctional Biofilter to Effectively Remove Toxins. ACS Applied Bio Materials 2020. |
SU839962 (Final) |
Exit |
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.