Grantee Research Project Results
Final Report: Simple and Inexpensive Paper-Based Assay for Lead
EPA Grant Number: SU839890Title: Simple and Inexpensive Paper-Based Assay for Lead
Investigators: Toote, Lauren E , Read-Daily, Brenda
Institution: Elizabethtown College
EPA Project Officer: Page, Angela
Phase: I
Project Period: October 1, 2019 through September 30, 2020 (Extended to September 30, 2021)
Project Amount: $23,811
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2019) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
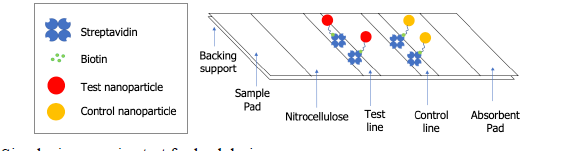
There are 6.1 million lead water service lines in the United States. This fact leaves many Americans in a precarious situation, as slight changes in the properties of their water, such as pH, could result in the leaching of lead into their tap water. Unfortunately, the average person does not have the ability to test for lead in their tap water. This problem exists because current technologies for lead detection are expensive and require trained personnel. As a result, these tests are performed in laboratory settings, not in homes. To address this issue, we propose a simple and inexpensive paper test for lead(II) in tap water. In this test, polymer nanoparticles, which encapsulate a molecular probe that has a colorimetric response to lead(II), will be anchored at the test line of a lateral flow assay. As a tap water sample flows down the strip, if lead(II) is present, a signal will result from the interaction of the lead(II) ions with the molecular probe. This signal can be visually observed by the user, giving communities which fall victim to contaminated water the opportunity to test their water and have control over their health.
Figure 1. Simple, inexpensive test for lead design.
Summary/Accomplishments (Outputs/Outcomes):
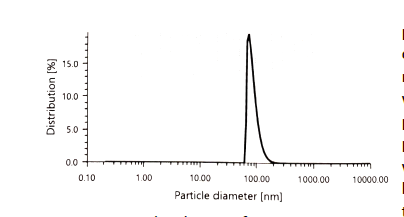
The chemistry team has optimized the synthesis and purification of the phenanthroline-based probe for detection of lead(II), with a final yield of 37%. This probe was tested for response to lead(II) in 9:1 acetonitrile:water, using lead(II) acetate, and produced a visual response at lead(II) concentrations below 500 µM. The nanoparticles for encapsulation of the probe were synthesized through optimization of the polymer, the solvent, the solvent to water ratio, polymer concentration and type of stabilizer. It was found that the phenanthroline probe could be incorporated in poly(styrene-co-maleic acid) to form stable nanoparticles that could be used for detection of lead(II) in aqueous solutions. These particles also allowed for attachment of the probe to the lateral flow membrane. They were found to have a size of 90 nm by dynamic light scattering (DLS) (Figure 1). These particles were found to be selective in their response to lead(II) when tested against other metal ions commonly found in tap water. Control nanoparticles that are incorporated into the test to confirm it is working properly were also synthesized. For these particles, bromocresol green was encapsulated in poly(lactic-co-glycolic acid) nanoparticles, with a size of approximately 120 nm. Their visual, colorimetric response to solutions of differing pH is shown in Figure 2. The control nanoparticles were attached to the lateral flow strip and a yellow line was observed. When a neutral water sample flows down the test, the line turns blue.
The engineering team completed life cycle analyses (LCA) for several different environmentally friendly materials to be used for housing the lateral flow strip. The scope of their LCA included raw material sourcing, manufacturing, transportation, retail and use, and disposal. The best option was selected based on carbon footprint and biodegradability. The plastic housing design went through multiple iterations to determine the optimal settings for 3D printing and design features allowing the two parts of the housing to snap together and ensure the lateral flow assay paper stays in place (Figure 3). The final prototype produces 0.405 lbs of CO2. Additionally, the engineering team determined the optimal amount of water sample to add so that there was sufficient volume
saturating the system. It was found that for a sample size between approximately 80 and 200 microliters, the ideal strip width was 5 mm. The final design was made for a 10 mm strip so that a greater sample size could be used.
Figure 1. Size distribution of PSMA nanoparticles that encapsulate the phenanthroline probe.
Finally, the nanoparticles were put onto the lateral flow membrane and the ability of the completed test to produce a visual signal for detection of lead(II) was evaluated. First, it was found that the lead(II) detecting nanoparticles could be attached to the nitrocellulose lateral flow membrane through hydrophilic/hydrophobic interactions. The amount of nanoparticles for maximum signal was then evaluated and the LOD of the test determined. As can be seen in Figure 4, a visual signal change can be seen down to 0.5 mM lead(II). Evaluation of these results through pixel analysis of the images gave a similar LOD. These results were observed within 10 minutes of adding the spiked water sample. These results show the usefulness of colorimetric nanoparticles for detection of heavy metals in a lateral flow assay format. One of the most significant advantages to our technique is that along with its simple use and quick time to result, it is very cost-effective. Currently, the cost of material per test is approximately $4.50, making this an accessible option for all homeowners. As a result, we have shown the feasibility of a lateral flow assay for detection of lead(II) in drinking water using a lateral flow assay with colorimetric nanoparticles.
Figure 2. Colorimetric response of control nanoparticles to pH.
Figure 3. Prototype lead (II) test housing
Supplemental Keywords:
sustainable water management, drinking water, heavy metal detection, nanotechnologyProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.