Grantee Research Project Results
2020 Progress Report: Simple and Inexpensive Paper-Based Assay for Lead
EPA Grant Number: SU839890Title: Simple and Inexpensive Paper-Based Assay for Lead
Investigators: Toote, Lauren E , Read-Daily, Brenda
Institution: Elizabethtown College
EPA Project Officer: Page, Angela
Phase: I
Project Period: October 1, 2019 through September 30, 2020 (Extended to September 30, 2021)
Project Period Covered by this Report: October 1, 2019 through September 30,2020
Project Amount: $23,811
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2019) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
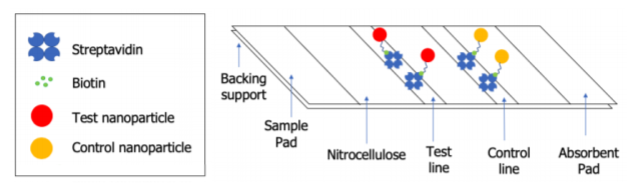
The P3 Grant Team at Elizabethtown College has been working development of a Simple and Inexpensive Paper-Based Assay for Lead. The test is a paper-based, lateral flow assay that produces a visual signal in response to lead(II) in water. The signal is a result of a color change in polymer nanoparticles that incorporate a molecular probe that responds to lead(II). The design for this test is shown in Figure 1, where the test nanoparticles responds to lead(II) and the control nanoparticle confirms that water is flowing down strip and the test is working properly. Work on this project progressed according to plan until our labs were closed on March 13, 2020, due to the COVID-19 pandemic. As a result, the work of the chemistry team was completely halted and the work of the engineering team was reduced, though they were able to take a 3D printer home with them to continue that aspect of the project. Furthermore, both the Chemistry and Engineering students planned to complete summer research on campus through the College's Summer Creative Arts and Research Program (SCARP). However, the college switched to offering only remote research opportunities, preventing the chemistry team from participating and forcing the engineering team to adapt the scope of the work accordingly. To do so, Dr. Read-Daily shipped students required materials, and they completed 3D printing of the housing prototypes, a life cycle analysis of the Algix 3D AlgaTM and PLA filaments, and lateral flow testing, details of which are provided below. The engineering team shared their research virtually on July 27, 2020, as part of the Elizabethtown College SCARP Presentations. Both teams were able to return to research on campus in September 2020.
Figure 1. Simple, inexpensive test for lead design
The original timeline for this project is shown in the Figure 2. At this point the chemistry team has been able to completetwo stages of the project and the engineering team has completed three stages. The data and results from this work is described in detail below. A one-year, no cost extension on this award was requested and approved, to allow for completion of the project now that research labs have been reopened.
Figure 2. Original project timeline
Progress Summary:
Synthesis of Phenanthroline Probe
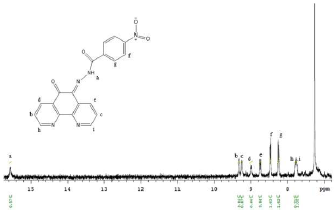
The chemistry team optimized the synthesis and purification of the phenanthroline-based probe for detection of lead(II), getting a final yield of 37% [1]. This was achieved through the reaction of 1,10-phenanthroline-5,6-dione and 4-nitrobenzhydrazide in dry methanol and then purification with a silica column. The proton NMR spectra and corresponding structure of the phenanthroline probe are shown in Figure 3. This probe was tested for response to lead(II) in 9:1 acetonitrile:water, using lead(II) acetate, and produced a visual response even at lead(II) concentrations below 500 M. This work was presented at the UMBC Undergraduate Research Symposium.
Figure 3. 1H NMR spectrum ofphenanthroline
Synthesis of Lead(II) Detecting Nanoparticles
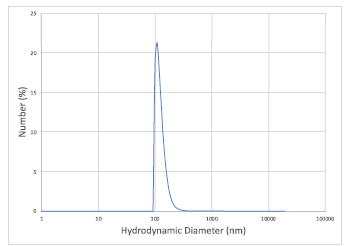
The phenanthroline probe was incorporated into polymer nanoparticles to allow for lead(II) detection in aqueous solutions and attachment to the lateral flow membrane. Nanoparticles were synthesized from several different polymers and it was found that the most stable particles were formed using polystyrene. These nanoparticles had a size of 140 nm as shown in the size distribution plot in Figure 4, that was collected by dynamic light scattering (DLS). Unfortunately, a consistent colorimetric response to lead(II) was not observed with these particles, possibly because the high hydrophobicity of polystyrene prevents the lead(II) from interacting with the probe. Thus, other polymers are being explored that retain the hydrophobicity needed to successfully encapsulate the probe, while still having hydrophilic character to allow interaction with lead(II). Polymers that will be explored include poly(4- vinylphenol) and poly(styrene-co-methacrylic acid). If these attempts do not give the detection limits needed, a rhodamine-based probe that has been synthesized in our lab that also shows sensitivity to lead(II) will be incorporated into this design.
Figure 4. Size distrubution of polystyrene nanoparticles that encapsulate the phenanthroline probe
Development of Control Nanoparticles
Much work has also been completed on the control nanoparticles that will be incorporated into the test to confirm it is working properly. It was found that bromocresol green was the best pH sensitive molecule to incorporate into the control nanoparticles. This molecule turns from yellow to blue at neutral pH, thus giving a signal when a water sample flows down the test. Poly(D,L-lactideco-glycolide) nanoparticles, encapsulating bromocresol green, were synthesized. The resulting nanoparticles were stable in solution and in environments of varying pH, with a size of approximately 120 nm as confirmed by DLS. Their visual, colorimetric response to solutions of differing pH was confirmed, as shown in Figure 5.
Figure 5. Colorimetric response of control nanoparticles to pH.
Incorporation of Nanoparticles into Lateral Flow Assay
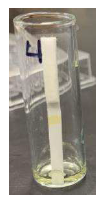
Once the control nanoparticles were made, they were attached to the lateral flow strip using the well-known interaction between biotin and streptavidin. To achieve this goal, biotin was conjugated to the control nanoparticles and streptavidin was adsorbed on the test strip. The strip was then placed in a solution of the control nanoparticles and flow up the test and attachment of the yellow particles at the line of streptavidin was observed (Figure 6). The strips were then allowed to dry and DI water was added to the strip. When the water flowed up the strip, the control nanoparticles turned green, showing their ability to be incorporated into the assay and to function as a control for the test.
Figure 6. Lateral flow strip with yello control line
Life Cycle Analysis on Algix 3D AlgaTM and PLA Filament for LFA Housing
The engineering students completed life cycle analyses (LCA) for Algix 3D Alga TM and PLA filament, environmentally friendly materials to be used for housing the lateral flow strip developed by the chemistry team. The scope of the LCA included raw material sourcing, manufacturing, transportation, retail and use, and disposal. They determined that the Algix 3D AlgaTM and PLA filaments require 7.9 and 11.8 kWh, respectively. In terms of overall carbon footprint, the Algix 3D AlgaTM and PLA generate 10.3, and 16.6 pounds of CO2, respectively. Table 1 provides a complete breakdown of the energy requirements and CO2 emissions resulting from Algix 3D AlgaTM and PLA. The Algix 3D AlgaTM biodegrades in three years whereas the PLA takes 15 years [2]. Another sustainable benefit of the Algix 3D AlgaTM filament is that it provides a market for nuisance algae [3], [4]. As are result, this material was used in the house design shown below.
Table 1. Comparison of Energy Requirements and CO2 Emissions for Algix 3D Alga(TM) and PLA
| Energy Required (kWh/lb) | CO2 Emissions Produced (lb) | |||
| Algix 3D Alga(TM) | PLA | Algix 3D(TM) | PLA | |
| Raw Materials | 3.35 [4], [5] | 5.37 [5] | 5.2 | 8.4 |
| Manufacturing & Processing | 4.412 [4] | 6.3 [5] | 6.9 | 9.8 |
| Transportation | 0.057 [2] | 0.057 [9] | 0.089 | 0.089 |
| Retail & Use | 0.04 | 0.04 | 0.062 | 0.062 |
| Waste | - | - | -2 [6], [3] | -2 [2] [6] [8] |
| Total Energy | 7.859 | 11.767 | 10.251 | 16.351 |
Housing Design and 3D Printing
To ensure accurate and safe use of the lateral flow assay, a plastic housing was designed and eight prototypes were tested. With each prototype students established the optimal settings for 3D printing, design features allowing the two parts of the housing to snap together and features to ensure the lateral flow assay paper stays in place (Figure 7). With the final prototype, they found that their housing produces 0.405 lbs of CO2.
Figure 7. Images of Prototype Lead Test Housing Using Algix 3D Alga(TM) filament
Lateral Flow Assay Tests
Using food dye and a set of pipettes for precise measurement, the engineering team determined the optimal amount of water sample to add so that there was sufficient volume to travel through the lateral flow assay without over-saturating the system (Figure 8). It was found that for a sample size between approximately 80 and 200 microliters, the ideal strip width was 5 mm. For larger volumes, a 7.5 or 10 mm strip may be better suited though, as of now, there are no specific measures of volume for those sizes. The final sample volume will be determined once the lead(II) detecting nanoparticles are incorporated into the test strip.
Figure 8. Lateral flow tests showing red dye traveling down strip material
Educational Work and Presentations
Beyond the development of a simple test for lead(II), several educational objectives of this grant have been completed. Both Dr. Toote and Dr. Read-Daily presented case studies in their respective courses that explored the need for lead(II) testing and the P3 approach. Additionally, students gave virtual presentations on their results during a campus-wide Scholarship and Creative Arts Day. Dr. Toote also gave seminars at Bucknell University and Gettysburg College. Furthermore, our P3 grant received much publicity in the local news, allowing us to share the P3 approach to lead testing with an even broader audience. One article in the Lancaster Newspaper (LNP) resulted in a connection with Dr. Alan Peterson from the Lancaster Lead Coalition, who offered to help us find lead(II) testing sites in the future [9]. Unfortunately, the chemistry team was unable to present posters for their abstracts that had been accepted for the National American Chemical Society Meeting, due to COVID-19, and other summer conferences were cancelled. Furthermore, planned visits to AP high school classrooms were postponed. So, though times are uncertain, in the next reporting period we plan to seek out venues for presentation of our results and also share the P3 approach in high school classrooms.
Quality Assurance and Research Misconduct
Quality assurance of the results obtained during this project are ensured through the training students received in lab procedures from the PI and co-PI. Furthermore, methodical notes were taken throughout the project as every student was required to keep a lab notebook. Additionally, the PIs ensured research misconduct did not occur, and this was done in part by holding regular meetings between team members where results are presented and discussed.
Budget
We have spent $2,400 of the ~$15,000 allocated in our original budget. These funds were used to buy supplies and reagents for the project. In this next reporting period the remaining funds will be used for travel to the P3 Expo if it is held and supplies, as greater amount will be needed once a complete prototype is developed and we begin to test water samples.
Subaward Monitoring Activities
There are no subawards associated with this grant.
Future Activities:
The only change to the goals of this project is the one-year extension. This extension allows us to make up for lost time during lab closures due to the COVID-19 pandemic. Also, there have been no key changes in personnel, as the principle investigator and co-investigator have stayed the same and any new student members have been trained by previous students or the PI. Unless policies relating to COVID-19 restrict our access to research labs again, future work will focus on the continued optimization of the polymer nanoparticles for lead(II) detection. This work will include optimization of the loading density of the phenanthroline probe, pH required for optimal signal and testing of the selectivity for lead(II). These results will be analyzed visually, as well as with UV-Vis spectroscopy. The lead(II) detecting nanoparticles will then be attached to the lateral flow membrane as has already been done with the control nanoparticles. Next, the sensitivity of the test will be determined through measuring the test signal by scanning an image of the test and measuring the pixel density at the test line. Finally, the chemistry and engineering teams will work together to test the final design with prepared water samples spiked with lead and with tap water samples.
References:
- Goswami, S., Chakrabarty, R. "Highly Selective Colorimetric Fluorescent Sensor for Pb2+ " in European Journal of Organic Chemistry, 2010, pp. 3791-3795.
- Representative from 3DPrintLife. [Interview]. July 2020.
- Algix, "About Algix," [Online]. Available: https: //algix.com/#abou t. [Accessed 22 12 2020].
- S. Hartter and M. Dave, "Algix," 17 12 2014. [Online]. Available: https://ltsexperts.com/wp- content/uploads /2017/03/Algix-LCA-of-Algae-Blended-Plastics_12.17.14.pdf . [Accessed 22 12 2020].
- Agriculture XPRT, "Algent - Model MR - Algae Harvester, Xprt Agriculture," [Online]. Available: https://www.agriculture-xprt.com/products/algent-model-mr-algae-hravester-567255 . [Accessed 22 12 2020].
- World Centric, "Eco-Profile of Compostable Plastic," 3 July 2018. [Online]. Available: https://www.worldcentric.com /joumal/compostable-plastic#:~:text=11.67%20kWh%20to%20produce%201.0,kWh%20for%20product%20production. [Accessed 22 12 2020].
- A. Fulke, K. Kannan, T. Chakrabarti and S. d. Sivanesan, "CO2 Sequestration by Microalgae: Advances and Perspectives," in Recent Advances in Microalgal Biotechnology, Foster City, California: OMICS Group eBooks, 2014, pp. 1-7.
- K. Thelen, "Com Fields Help Clean Up and Protect the Environment," 7 June 2007. [Accessed 22 12 2020].
- S. Sauro, "There's no in-home test for lead in your water; Elizabethtown College is working on one," LNP, 28 February 2020.
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.