Grantee Research Project Results
2021 Progress Report: Fundamentals of Adsorption, Desorption, and Biodegradation of PFAS and Precursor Compounds in the Soil and Landfill Leachate System and An Innovative Treatment Strategy for Their Removal in Landfill Leachate and Groundwater
EPA Grant Number: R839660Title: Fundamentals of Adsorption, Desorption, and Biodegradation of PFAS and Precursor Compounds in the Soil and Landfill Leachate System and An Innovative Treatment Strategy for Their Removal in Landfill Leachate and Groundwater
Investigators: Xiao, Feng
Institution: University of North Dakota
EPA Project Officer: Hahn, Intaek
Project Period: August 1, 2019 through July 31, 2022 (Extended to July 31, 2023)
Project Period Covered by this Report: August 1, 2020 through July 31,2021
Project Amount: $498,907
RFA: Practical Methods to Analyze and Treat Emerging Contaminants (PFAS) in Solid Waste, Landfills, Wastewater/Leachates, Soils, and Groundwater to Protect Human Health and the Environment (2018) RFA Text | Recipients Lists
Research Category: Drinking Water , Water Quality , Human Health , Water , PFAS Treatment
Objective:
- Identify the governing soil sorption and desorption mechanisms (thermodynamics and hysteresis) of per- and polyfluoroalkyl substances (PFAS) in landfill leachate.
- Investigate the biodegradation of sorbed precursor compounds of perfluoroalkyl acids (PFAAs) and examine the effect of sorption hysteresis.
- Innovatively integrate and enhance commonly used water treatment technologies to effectively and practically remove PFAS from the test water.
Progress Summary:
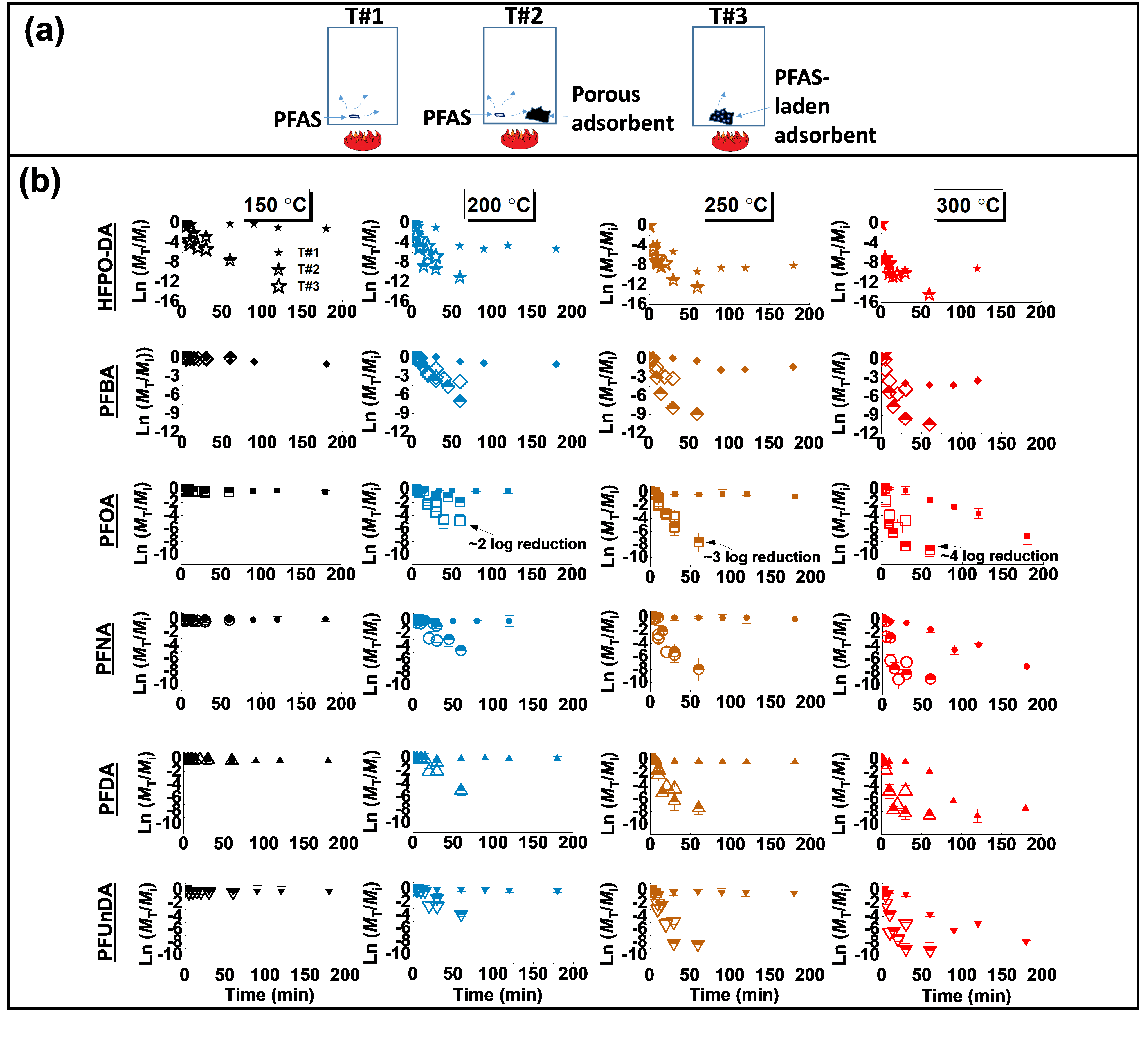
We performed a detailed investigation of the fate of per- and polyfluoroalkyl substances (PFAS) and one PFAS alternative (GenX) in thermal processes, focusing on the effect of GAC. We demonstrate that the thermolysis of perfluoroalkyl carboxylic acids (PFCAs), including perfluorooctanoic acid (PFOA), and GenX can occur at temperatures of 150‒200 oC. We found that the thermal decomposition of PFAS mostly followed first-order kinetics at temperatures of 150‒250 °C, in which the residual PFAS mass (MPFAS,T) decreased in an exponential manner over heating time (Fig. 1). At a higher temperature (300 °C), the decomposition of these chemicals can also be described by second-order kinetics, in which a plot of 1/MPFAS,T versus heating time is approximately linear. For comparison purposes, we used the first-order decomposition rate constant (k1st, min-1) in the following discussion.
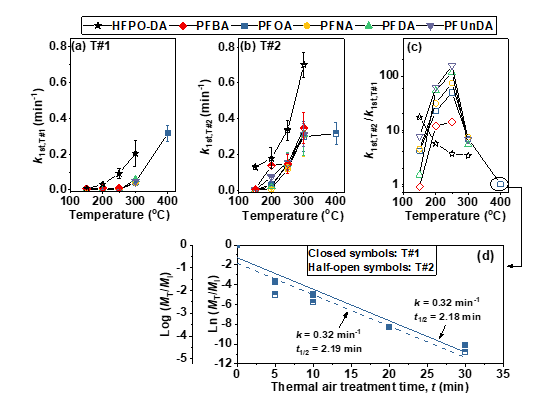
The value of k1st was below 0.005 min-1 for PFCAs at 150‒250 °C in T#1 (Fig. 2). PFECA (HFPO-DA) appears to be more easily degraded than the PFCA with the same number of perfluorinated carbons (i.e., PFBA) (Figs. 1 and 2). This is consistent with our previous observation 1 that the perfluorinated chain becomes less thermally stable with the inclusion of a foreign group such as the ether group (see Figs. 1 and 2).
(1) database of soil sorption and desorption of cationic and zwitterionic PFAS compounds; (2) publications documenting the effects of PFAS and soil properties on the sorption and desorption hysteresis; (3) preliminary data for the innovative treatment strategy to remove PFAS from water and decompose the compounds on granular activated carbon; (4) database and publications documenting PFAS thermal stability; (5) database and publications documenting PFAS decomposition during GAC thermal reactivation; and (6) preliminary results on the biodegradation of precursor compounds of PFOA and PFOS.
The progress outcomes include an increase in the ability to predict the fate of PFAS in the soil environment and an increase in the ability to remove and decompose PFAS from the drinking-water treatment system.
Fig. 1. (a) A schematic of PFAS thermal decomposition experiments in a closed system in three conditions (T#1, T#2, T#3). (b) Degradation of studied PFAS and a PFAS alternative (HFPO-DA) at various temperatures. Thermal treatments were triplicated for PFOA, PFNA, PFDA, and PFUnDA. The error bars represent the standard deviation (1-sigma) of three trials. Some points without visible error bars have errors that are smaller than the symbol. T#1 (closed symbols): PFAS only (initial PFAS mass: 1.2 × 10-5 mol). T#2 (half-open symbols): PFAS (1.2 × 10-5 mol) with the presence of 0.1 g GAC. T#3 (open symbols): 0.1 g of GAC laden with 3.0‒4.5 × 10-7 mol PFAS. MT and Mi are the residual mass of PFAS in heated samples and the initial mass in non-heated controls, respectively. The degradation of studied PFAS at 150 oC occurred at a slow rate (see Fig. 2 for decomposition rate constants).
Fig. 2. (a‒c) First-order decomposition rate constants (k1st, min-1) of PFCAs (1.2 × 10-5 mol) treated at different temperatures in conditions of T#1 (thermolysis; closed symbols) and T#2 (with the presence of 0.1 g of GAC; half-open symbols). (d) Thermal decomposition of PFOA (1.2 × 10-5 mol) in T#1 and T#2 (with the presence of 0.1 g GAC) in a closed container at 400 °C. Lines in (d) represent the best-fit to the decomposition data using the first-order kinetic model.
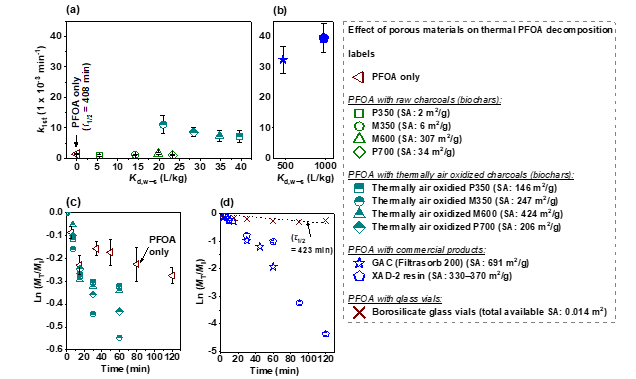
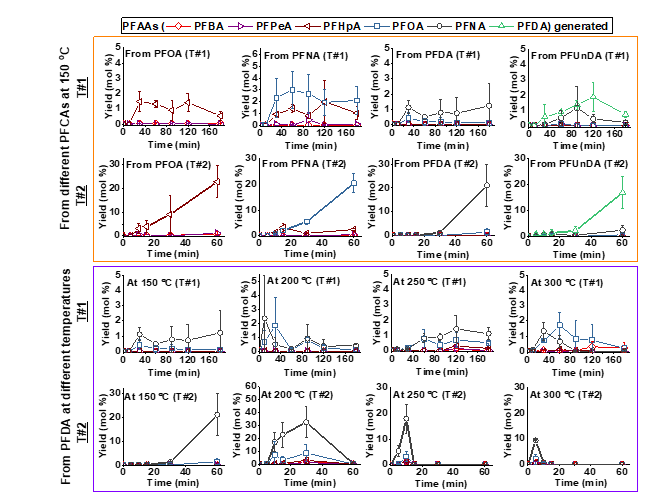
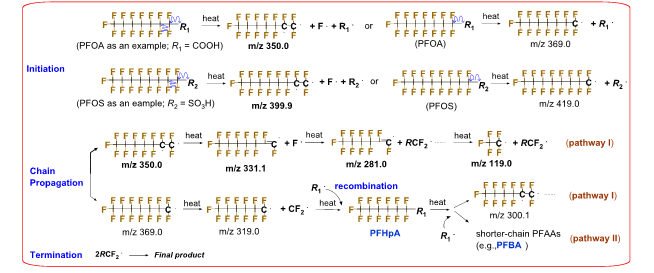
We also found that the addition of a highly porous adsorbent, such as GAC or a copolymer resin, compressed the intermediate sublimation zone of PFCAs, changed their thermal decomposition pathways, and increased the decomposition rate constant by up to 150-fold at 250 oC (Fig. 3). The results indicate that the observed thermal decomposition acceleration was linked to the adsorption of gas-phase PFCA molecules on GAC. The presence of non-activated charcoals/biochars with a low affinity for PFOA did not accelerate its thermal decomposition, suggesting that the π electron-rich, polyaromatic surface of charcoal/GAC played an insignificant role compared to the adsorbent’s porosity (Fig. 3). The thermal decomposition began with the homolysis of a C‒C bond next to the carboxyl group of PFCAs, which formed unstable perfluoroalkyl radicals. Dual decomposition pathways seem to exist (Figs. 4 and 5).
Fig. 3. (a) and (b): First-order decomposition rate constants (k1st, 1 × 10-3 min-1) of PFOA (1.2 × 10-5 mol) treated at 200 °C in T#1 (thermolysis) or in T#2 with the presence of a raw charcoal sample (M350, M600, P350, or P700) (0.5 g), a thermally air oxidized charcoal (0.1 g), GAC (Filtrasorb 200) (0.1 g), a non-carbonaceous porous material (XAD-2 resin) (0.1 g), or five 2-mL borosilicate vials (11.1 ± 0.01 g). (c) and (d): Thermal decomposition of PFOA in different conditions at 200 °C. The dashed line in (d) represents the best-fit to the decomposition data using the first-order kinetic model. The N2 B.E.T. SA of XAD-2 resin was obtained from the literature 2,3. The N2 B.E.T. SAs of nine PCM adsorbents were measured in this study.
Fig. 4. Generation (and subsequent decomposition) of intermediate short-chain PFCAs from parent compounds treated in different conditions (T#1 and T#2; Fig. 1). The intermediates were detected by the UPLC‒QToF-MS/MS method as detailed in the Supplementary data. Note the difference in y-axis scales.
Fig. 5. Possible “unzip” decomposition pathways of perfluoroalkyl substances during thermal treatment. Species with m/z 399.9, 350.0, 331.1, 281.0, and 119.0 were shown in TD‒Pyr‒GC‒MS spectra of PFOA 1 and PFOS (Fig. S17). We found no MS spectral evidence for ׃C7F14 (m/z 300.1), ·C6F13 (m/z 319.0), ·C7F15 (m/z 369.0), and ·C8F17 (m/z 419.0), but this does not preclude their formation in small amounts.
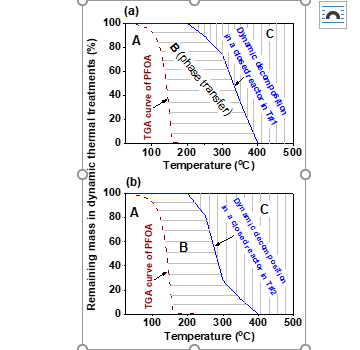
Three temperature zones were discovered for PFOA, including a stable and nonvolatile zone (≤90 oC), a phase-transfer and thermal decomposition zone (90‒400 oC), and a fast decomposition zone (≥400 oC) (Fig. 7). Overall, the results indicate that (1) decomposition of PFCAs and GenX in thermal water/wastewater/sludge/waste treatments is very likely provided sufficiently high temperatures are used, and (2) the presence or addition of GAC or other highly porous materials can accelerate thermal PFAS decomposition.
Fig. 7. Brown dashed lines: dynamic (10 oC/min) TGA curves of PFOA determined in an open system 1. Blue solid curves: mass loss of PFOA in dynamic (10 oC/min) thermal treatments performed in a closed container in two conditions: (a), T#1 (1.2 × 10-5 mol PFOA); (b), T#2 (1.2 × 10-5 mol PFOA with 0.1 g GAC).
Future Activities:
Research in progress: (1) the adsorption of PFAS in the soil‒leachate system; and (2) an innovative treatment system to remove and destroy PFAS of various types in landfill leachate samples.
Research planned (or just started) (1) the biodegradation of precursor compounds of certain perfluoroalkyl substances in the soil environment; and (2) the effect of sorption and sorption hysteresis on the biodegradation of precursor compounds of a few commonly detected perfluoroalkyl substances.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 15 publications | 13 publications in selected types | All 13 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Charbonnet J, McDonough C, Xiao F, Swichtenberg T, Cao D, Kaserzon S, Thomas K, Dewapriya P, Place B, Schymanski E. Communicating Confidence of Per-and Polyfluoroalkyl Substance Identification via High-Resolution Mass Spectrometry. ENVIRONMENTAL SCIENCE & TECHNOLOGY LETTERS 2022;9(6):473-481. |
R839660 (2021) R839670 (2021) R839670 (2022) |
Exit Exit |
Supplemental Keywords:
Adsorption; desorption; degradation; practical removal; enhanced water treatment
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.