Grantee Research Project Results
2021 Progress Report: The BOHP/UV Process for Destruction of PFAS in Leachate and Groundwater: Tandem mechanistic advancement and pilot demonstration
EPA Grant Number: R839630Title: The BOHP/UV Process for Destruction of PFAS in Leachate and Groundwater: Tandem mechanistic advancement and pilot demonstration
Investigators: Cates, Ezra L
Institution: Clemson University
EPA Project Officer: Hahn, Intaek
Project Period: August 1, 2019 through July 31, 2022 (Extended to July 31, 2023)
Project Period Covered by this Report: August 1, 2020 through July 31,2021
Project Amount: $458,469
RFA: Practical Methods to Analyze and Treat Emerging Contaminants (PFAS) in Solid Waste, Landfills, Wastewater/Leachates, Soils, and Groundwater to Protect Human Health and the Environment (2018) RFA Text | Recipients Lists
Research Category: Drinking Water , Water Quality , Human Health , Water , PFAS Treatment
Objective:
This works seeks to apply a semiconductor photocatalytic treatment method and reactor system to the destruction of poly-/perfluoroalkyl substances (PFAS) present in landfill leachate. The original objectives of the 3-year project are to (1) assess degradation kinetics of leachate-relevant poly/perfluorocarboxylic acids (PFCAs and FTCAs) by Bi3O(OH)(PO4)2/ultraviolet process (BOHP/UV) using catalyst particles prepared in the PI’s lab; (2) test hypothesized process modification for photocatalytic mineralization of perfluorosulfonates; and (3) conduct trials of leachate treatment in an integrated BOHP/UV reactor with ceramic ultrafiltration pretreatment (Purifics Photo-cat). During the first year of work, the proposed Fe3+coordination-assisted oxidative photocatalytic degradation of the recalcitrant perfluorooctane sulfonate (PFOS) pertaining to Objective 2 was found to be ineffective; as a result, an alternative approach was proposed for testing in Year 2, involving a BiPO4/UV photocatalytic reduction method. The commercial Photo-cat system was also found to be inefficient and a stirred tanks-in-series approach was proposed instead. These changes were outlined in the 2020 project report. Task 1 has proceeded largely as originally planned. The schedule of tasks for Year 2 as planned following previous year results is shown below for reference:
| Year 2 Tasks | 2020 | 2021 | |
|---|---|---|---|
| 1. Degradation assessment of Leachate-Relevant FTCAs by BOHP/UV | |||
| 2. Assessment of leachate matrix co-constituent effects on BOHP/UV degradation of PFAS | |||
| 3. Comparison of DOC reduction rates by BOHP/UV and TO2/UV | |||
| 4. PFOS treatment studies via BIP)/UV reductive process | |||
| 5. Construction and preliminary testing of pilot-scale stirred-tank Photocatalutic reactors | |||
Progress Summary:
Research activities in the second year focused more specifically on applying the BOHP/UV process to landfill leachate, as opposed to more generalized water treatment capabilities. 2 Project tasks and outcomes during this period are described below, with resultant modified project tasks following thereafter.
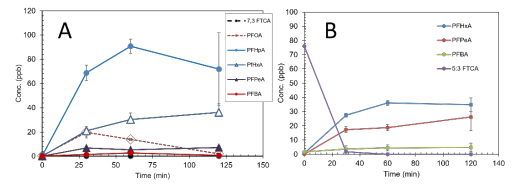
Task 1. Degradation Assessment of Leachate-Relevant FTCAs by BOHP/UV. Literature studies on PFAS in landfill leachate have indicated that in addition to the more commonly studied longand short-chain PFCAs and PFSAs, several fluorotelomer carboxylic acids (FTCAs) can be the dominant PFAS contaminant in these matrices. We have assessed the degradation of the two most commonly reported leachate FTCAs by BOHP/UV, and found that they are readily degraded by photocatalysis. At low pH, these compounds strongly adsorbed to the positivelycharged BOHP particle surfaces; in fact, following FTCA spikes and stirring in dark conditions to achieve adsorption equilibria, the bulk-phase concentration of the FCTAs was in some cases below the detection limits of our analysis method, indicating highly favorable adsorption. Subsequent photocatalytic degradation of the adsorbed compounds during UV irradiation was thus confirmed via detection of intermediate product formation. Figure 1A below shows the time profiles of bulk phase parent compound and intermediate product concentrations during degradation of 7:3 FTCA (C0 = 100 ppb) by BOHP/UV, indicating this compound is fully adsorbed at the onset; then, two of the three nonfluorinated carbons are cleaved off via oxidation to form the 8-carbon PFOA product, which was in turn rapidly converted to perfluoroheptanoic acid via stepwise chain shortening. Based on the intermediate product concentrations, the parent compound was completely degraded. Similarly, 5:3 FTCA degradation was also examined (Figure 1B), showing a lesser degree of adsorption and degradation but an identical mechanism, which lead primarily to a perfluorohexanoic acid byproduct. These results show that the FTCAs commonly found in leachate are readily converted to PFCAs by the BOHP/UV process. While earlier studies have found that short-chain PFCAs (6-carbon and shorter), such as these intermediates, are poorly adsorbable to BOHP and not significantly degraded by photocatalysis, they are in fact degraded via photolysis from the 185 nm emission component of low-pressure mercury lamps used in the pilot-scale BOHP/UV process.
Figure 1. Parent compound and intermediate PFAS degradation products during BOHP/UV photocatalytic degradation of leachate-relevant fluorotelomer carboxylic acids, including (A) 7:3 FTCA and (B) 5:3 FTCA. Conditions: 254 nm, pH 4, C0,parent = 100 ppb.
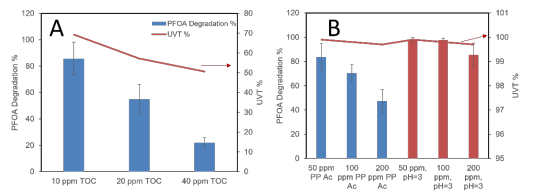
Task 2. Assessment of leachate matrix co-constituent effects on BOHP/UV degradation of PFAS. Previous study of PFAS degradation by BOHP/UV as part of a different project revealed that Cl– and SO4 2– anions interfere significantly with BOHP/UV photocatalysis at groundwaterlevel concentrations (100 ppm), whereas humic substances did not, if pH was lowered to 4. Herein we have further studied the effect of humic substances, as well as volatile fatty acids 3 (VFAs), at leachate-relevant concentrations in order to gauge the applicability of BOHP/UV to such matrices. As shown in Figure 2A, humic substances did have a detrimental effect on PFOA degradation at concentrations on the order of 101 ppm; however since these tests were conducted at acidic pH wherein humic acids are protonated and interact minimally with the catalyst surface, this detraction from performance was attributed solely to a decrease in UV transmittance (UVT254) of the water. Nevertheless, this result indicated that the presence of humic substances in leachate is detrimental to treatment by BOHP/UV, and pretreatment steps designed to improve UV transmittance are likely required for photocatalytic treatment of PFAS. Propionic acid (PrA) was used as a representative VFA found in leachate and its effects on PFOA degradation are shown in Figure 2B. At pH 4, increasing PrA concentration led to lower PFOA degradation ratios, likely due to competitive adsorption by propionate anion on the catalyst surface. The effect was eliminated, however, by lowering the solution pH to 3 in order to fully protonate the VFAs and eliminate the anionic portion. Furthermore, VFAs did not result in a significant decrease in UVT254; thus assuming pH is lowered to 3, VFAs are not expected to impede PFCA degradation by BOHP/UV.
Figure 2. Effects of organic leachate constituents on PFOA degradation by BOHP/UV, including (A) humic substances at pH 4 and (B) propionic acid at pH 4 (blue) and 3 (red). UVT254 of the test solutions prior to irradiation are depicted by red lines. Irradiation time (254 nm) = 2 h; C0,PFOA = ~100 ppb.
Task 3. Comparison of DOC reduction rates by BOHP/UV and TiO2/UV (Revised: Coagulation and ultrafiltration pretreatment assessment). The assessment of photocatalytic DOC reduction has been moved to year 3, following a decision to implement additional pretreatment prior to any photocatalytic process in order to remove humic substances and improve UVT254 such that photocatalytic/photolytic methods will show greater efficiency. Coagulation followed by ceramic membrane ultrafiltration was chosen as the first pretreatment approach. This was initially pursued via jar testing of differing coagulant (FeCl3) doses using a synthetic leachate; however, this surrogate proved to be too easy to flocculate compared to real leachate and ultimately real leachate samples were obtained instead. A ceramic membrane test cell was set up and is now operational. Jar testing of coagulation performance for humics removal, followed by ultrafiltration trials are in progress, and changes in UVT254, as well as resultant BOHP/UV PFOA degradation efficiency will be used as primary metrics for process optimization. It remains to be determined if the use of Fe or Al coagulants can lead to a net improvement in UVT, and whether or not they will interfere with photocatalysis as a result of the associated anion addition (Cl– or SO4 2–). These aspects will be scrutinized and compared to ultrafiltration-only (no coagulation). Following development of these physical 4 methods for pretreatment of humic substances, additional pretreatment via TiO2/UV or simply extended BOHP/UV will be assessed using bench experiments.
Task 4. PFOS treatment studies via BiPO4/UV reductive process. In the 2020 annual report, data was included from experiments attempting photocatalytic treatment of PFOS by BiPO4/UV under reducing conditions (N2 bubbling, neutral pH, and methanol addition). The data included both PFOS parent compound disappearance coupled with release of F–. Following difficulties in acceptable reproducibility, further experiments were performed which ultimately determined that PFOS was only minimally degraded by this approach. Instead, the disappearance of PFOS originally observed was attributed to acidification due to photocatalytic conversion of methanol to formic acid, followed by low pH-induced adsorption of PFOS to the catalyst surface (data not shown). Due to the nature of this phenomenon, it was not initially obvious from our control experiments. We further analyzed F– production using the selective electrode method and found that only trace fluoride was liberated, indicating poor process performance. We conclude that photocatalysis is not a viable approach for degradation of perfluorosulfonates.
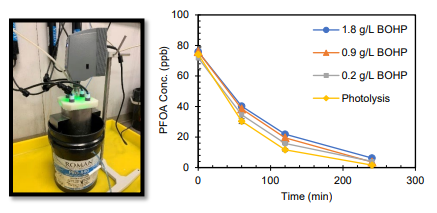
Task 5. Construction and preliminary testing of pilot-scale stirred-tank Photocatalytic reactors. Construction of 7.5, 20, and 45 L, pilot-scale stirred-tank photocatalytic reactors was completed, with a 7.5 L reactor with water bath shown in Figure 3. Unlike the smaller bench-scale reactors, wherein addition of the BOHP catalyst markedly improved PFOA degradation rates compared to UVC/VUV photolysis alone, no such photocatalytic enhancement was observed. Figure 3 shows that PFOA degradation is faster in the absence of BOHP and attributed solely on photolytic degradation. Several causes for this discrepancy have been ruled out, and further investigation is underway so that the more efficient photocatalytic degradation mechanism can be properly leveraged for treatment in the larger reactors.
Figure 3. Left: Pilot-scale stirred-tank photocatalytic reactor with external water bath, 7.5 L irradiation volume, vertical impeller mixer, and 4x 57 W ozone-generating low pressure mercury lamps (254/185 nm). Right: Comparison of PFOA degradation rates in pilot reactor via photocatalysis/photolysis at various catalyst concentrations and photolysis-only (no catalyst).
Quality Assurance. Results discussed herein comprise experiments for which reproducibility was assured by instrument calibration and full experimental repetition in triplicate. Regular meetings between the PI and lab personnel were conducted to assess experimental designs 0 20 40 60 80 100 0 100 200 300 PFOA Conc. (ppb) Time (min) 1.8 g/L BOHP 0.9 g/L BOHP 0.2 g/L BOHP Photolysis 5 and analytical outcomes. The use of ion chromatography for F– analysis was replaced with use of a F– selective electrode in order to eliminate false positive detection due to short-chain organic acids. It is hereby assured that no misconduct in research was observed during this reporting period.
Future Activities:
Based on results from Year 2, the projective objectives have been revised to include the following:
- Optimization of Coagulation/ultrafiltration Pretreatment for Increasing UVT of Leachate. Jar testing followed by membrane filtration and photocatalysis experiments is currently underway to determine if the overall cost effectiveness of PFAS photochemical destruction can be enhanced by pretreatment using physicochemical separation of humic substances. Leachate samples from Twin Chimneys landfill in Honea Path, SC, are being utilized.
- Comparison of DOC reduction rates by BOHP/UV and TiO2/UV. Residual DOC (including UV attenuating substances and photocatalysis quenching compounds) will be addressed through additional photocatalytic pretreatment, to further improve PFCA removal rates.
- Comparison Between Photocatalytic/photolytic and Photolytic-only Treatment. Given the potential for inorganic anions in leachate to disable efficient photocatalysis, optimization of photoreactors that rely only on vacuum UV (185 nm) photolysis of PFAS will be explored, which is less sensitive to salt content. Amalgam low pressure mercury lamps, which have a greater 185 nm output, will be purchased and utilized.
- Leachate Treatment Pilot Testing in Optimized Stirred-Tank Photocatalytic Reactors. As per the original project plan, leachate will be used for pilot testing within the new pilot-scale photocatalytic reactors developed jointly for this project and DoD-funded work. Selected pretreatment processes will be applied and degradation of a wide range of leachate-relevant PFAS analytes will be monitored.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 9 publications | 2 publications in selected types | All 2 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Qanbarzadeh M, Wang D, Ateia M, Sahu SP, Cates EL. Impacts of reactor configuration, degradation mechanisms, and water matrices on perfluorocarboxylic acid treatment efficiency by the UV/Bi3O(OH)(PO4)2 photocatalytic process. ACS ES&T Engineering 2020;1(2):239-248. |
R839630 (2021) R839630 (Final) |
Exit Exit |
Supplemental Keywords:
hazardous waste remediation; water purification technologies; photocatalytic reduction, groundwater remediation; photocatalyst water disinfectionRelevant Websites:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.