Grantee Research Project Results
Final Report: Separation and Recovery of Individual Components from theEnd-of-Life Lithium-ion Batteries
EPA Grant Number: SV839485Title: Separation and Recovery of Individual Components from theEnd-of-Life Lithium-ion Batteries
Investigators: Pan, Lei , Zhan, Ruiting , Folayan, Tinu , Payne, Trevyn , Oldenburg, Zack , Schienke, Matthew , Nunneley, Lucille , Billman, Tim , Olson, Zackary
Institution: Michigan Technological University
EPA Project Officer: Page, Angela
Phase: II
Project Period: April 1, 2019 through March 31, 2021 (Extended to March 31, 2022)
Project Amount: $75,000
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet - Phase 2 (2019) Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Chemical Safety
Objective:
The objective of this Phase II project is to develop a prototype of a laboratory-scale Li-ion battery recycling system. The system can separate individual components from spent Li-ion batteries and producing high-purity cathode materials for new batteries. The Phase II project is built upon the success of the Phase I project, in which the team successfully demonstrated that froth flotation and gravity concentrators perform effectively in separating fine and coarse battery materials, respectively. Three specific tasks include 1) developing a PID-controlled teeter-bed separator and PID-controlled 2-inch diameter flotation column, and testing the performance of individual units; 2) designing a recycling system that integrates teeter-bed separation, column flotation and other supporting systems; and 3) building a prototype system on a mobile skid. Another objective of this project is to provide resources and opportunities to promote STEM education in the United States.
Summary/Accomplishments (Outputs/Outcomes):
Figure 1 shows photos of recycled products from spent Li-ion batteries using our bench-scale recycling system. The recycled products include Cu, Al, separator, anode materials, and cathode active materials. The overall goal is to achieve zero waste through the proposed recycling system and produces value-added products for different markets.
Figure 1. Recycled products from spent Li-ion battery using the proposed recycling system.
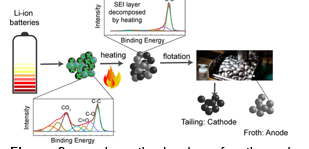
We have published three journal articles. We have demonstrated that electrode active materials from Li-ion batteries can be separated from each other using the froth flotation method [Zhan et al., 2018, Sustainable Materials and Technologies, e00062]. For end-of-life Li-ion batteries, aged electrode active materials suffered from a structural and compositional change on its surface. PI’s student team has shown that a solid electrolyte interphase (SEI) layer is formed on the aged anode surfaces, resulting in a decrease in their surface hydrophobicity [Zhan et al., 2021, ACS Sustainable Chem. Eng., 9, 531]. Figure 2 shows a schematic drawing of a pre-treatment process that has been developed to resolve this issue. The thermal process decomposes the SEI layers on the anode surfaces. As a result, separation of the electrode active materials from the end-of-life Li-ion batteries can be achieved.
Figure 2. a schematic drawing of a thermal process that improves the separation of two electrode active materials from Li-ion batteries.
Additionally, the team has developed a new method to increase both the purity and yield of the recycled cathode active materials from spent Li-ion batteries, which has been disclosed in our year 1 report. Froth flotation process produces 90%-94% pure of cathode composites from spent Li-ion batteries, and the remaining 6-10% of impurities are carbon black and PVDF binders. De-agglomeration of individual components from the cathode composite materials was carried out using a mechanical process [Zhan et al., 2020, Waste Management, 105, 39]. The separation performance has been evaluated by separating mixed materials by the degree in surface hydrophobicity using the froth flotation method. It was shown that the size of the cathode composite was effectively reduced upon a de-agglomeration process due to a liberation of PVDF binders from the cathode active materials. The performance improves with end-of-life (EOL) LIBs compared to new LIBs, benefiting from a degradation of PVDF binders after numerous charging-discharging cycles. The X-ray photoelectron spectra suggested that the de-agglomeration was achieved by breaking intermolecular bond between PVDF and cathode active materials as well as covalent bond within PVDF binders. The result demonstrated a non-chemical method for liberating individual components from cathode composites for the direct recycling of LIBs. Figure 3 shows SEM and EDS mapping images of the separated products after a 16-minute de-agglomeration process. As shown, the tailing products are free of PVDF binders and carbon additives. The purity of cathode active materials in recycled products was above 98%. This work has been published on Waste Management, one of top journals in the area of circular economy.
Figure 3. SEM/EDS images of the first froth concentrate and tailing products after a 16-minute de-agglomeration process.
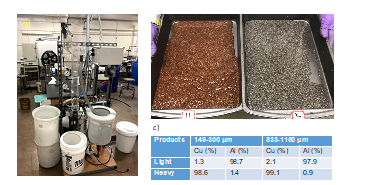
Two demonstration units were designed and constructed. They are crossflow teeter-bed separator and flotation columns. A manuscript is under the preparation that describes an overall flowsheet for the recycling of spent Li-ion batteries. Figure 4 shows a photo of a PID-controlled laboratory-scale crossflow teeter-bed separator developed by Michigan Tech battery recycling student team. The teeter-bed gravity separator fluidizes particle bed. The dense particles sink to the bottom of a fluidized bed, while light particles float with the upward fluid flow. A separation is achieved when lighter particles flow over into the overflow stream while heavy particles are collected at the underflow stream. This system was built and constructed with an assistance of Mr. Jerry Norkel, our lab machinist. There have been a number of modifications to the current version of the TBS system during the development phases. The main body of this system is made of plexiglass, which allows students observe motion of particles within the fluidized bed. The bed level is monitored by the pressure gauge. The feed materials are continuously fed to the TBS from the top, and the bed level is maintained by controlling the discharge using a pneumatic-controlled diagraph valve. Figure 4b shows photos of the separated products in the underflow and overflow streams. As shown, majority of copper pieces are concentrated in the underflow stream, while aluminum pieces are collected at the overflow stream. Figure 4c shows the separation performance of our laboratory-scale teeter-bed separator. An excellent separation has been achieved.
Figure 4. a) A photo of a crossflow-teeter bed separator built by Michigan Tech battery recycling student team for outreach demonstration; b) photos of separated products from spent Li-ion batteries; 3) separation performance of shredded copper and aluminum foils at two different size fractions (149-300 um and 833-1160 um).
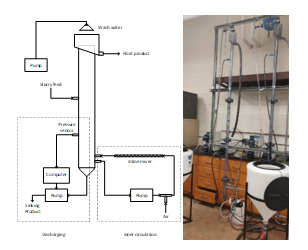
Figure 5. a) a schematic drawing and b) a photo of 2”-ID flotation columns that are design and built by Michigan Tech’s students’ team to separate electrode active materials from spent Li-ion batteries.
Figure 5 shows a photo and a schematic drawing of two laboratory-scale 1.5”-diameter flotation columns developed during this period. The system uses an in-line mixer for bubble generation. The froth height was controlled by maintaining a constant bed pressure using a PID loop control system. Washing water is used to remove entrainments in the froth layer. Air flow rate, feed rate, washing water flowrate were optimized for battery material separation experiments. The result showed that a better separation between the two electrode active materials was achieved using flotation columns compared to conventional mechanical flotation cells. Both the design and final construction of our battery recycling system will be completed in 6 months.
Conclusions:
The team has developed several battery recycling technologies for end-of-life (EOL) Li-ion batteries from both consumer electronics and electric vehicles (EVs). 80-90% separation efficiency between anode active materials and cathode active materials from the black mass has been achieved under this project. In addition, 99% purity of cathode active materials can be produced from Li-ion batteries without changing the bulk chemistry and structure of the cathode active materials. The impact of the battery recycling technologies developed under this project is to enable the development of “direct recycling” technologies as well as pre-treatment technologies that enable a significant reduction in the chemical cost with downstream hydrometallurgical refining processes. Other benefits to people, prosperity, and the planet include 1) 90%+ recovery of battery materials from secondary recycle stream into the domestic supply chain of battery materials, 2) a job creation in the United States, 3) an elimination of hazardous materials (such as fluorine and metals) entering the landfill. Future technological development in Li-ion battery recycling should focus on 1) process development of commercial-scale anode/cathode separation processes, 2) fire and explosion prevention during the shredding process, and 3) management of air, solid, and liquid wastes during the shredding process. The PI and his team are commercializing battery recycling technologies derived from this project.
Summary of Educational Accomplishments Resulting from P3 Project
This project is to provide resources and opportunities to promote STEM education in the United States. This project has recruited over six undergraduate students of diverse backgrounds at Michigan Technological University. These students gain hands-on experience in the laboratory and interact with industrial partners. In addition, undergraduate students are given opportunities to attend national and local conferences to present their research. The team has been participating Summer Youth Program (SYP) and develop a mini mobile lab. This mini lab is being used for multiple educational demonstration purposes.
Journal Articles:
No journal articles submitted with this report: View all 5 publications for this projectSupplemental Keywords:
Li-ion Battery Recycling; Direct Recycling; Resource Recovery; Critical MineralsProgress and Final Reports:
Original AbstractP3 Phase I:
Separation and Recovery of Individual Components from the End-of-Life Lithium-ion Batteries | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.