Grantee Research Project Results
Final Report: PFASs Removal by Photocatalysis for Water Reuse
EPA Grant Number: SU839460Title: PFASs Removal by Photocatalysis for Water Reuse
Investigators: Liu, Jia , Lydy, Michael J. , Goodson, Boyd , Geisler-Lee, Jane , Xia, Chunjie , Hadiuzzaman, Md , Derby, Andrew , Gemeinhardt, Max E. , Eckert, Tristin , Lipe, Kierstin , Gu, Fengtian
Institution: Southern Illinois University - Carbondale
EPA Project Officer: Page, Angela
Phase: I
Project Period: December 1, 2018 through November 30, 2019
Project Amount: $14,959
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2018) RFA Text | Recipients Lists
Research Category: P3 Challenge Area - Safe and Sustainable Water Resources , P3 Awards
Objective:
Wastewater effluent (WE) reclamation for reuse in agriculture has been practiced in many regions in the U.S. that lack sufficient freshwater supplies, including California and Florida, as a sustainable strategy to meet the water demand. However, WE reuse in agriculture is challenged by the presence of emerging organic contaminants, such as per- and polyfluoroalkyl substances (PFAS) in the reclaimed water, which bring potential adverse impacts on crops, soil and aqueous ecosystems, and human health. PFAS are ubiquitous in the environment in surface water, sediment, air, soil, and sludgeand have received global attention because of their persistence, bioaccumulation potential, and possible adverse effects on living organisms. Hence, due to potential risks to human health, perfluorooctanoic acid (PFOA) and perfluorooctane sulfonate (PFOS) have been included in U.S. EPA’s Third and Fourth Contaminant Candidate Lists (CCL3 & CCL4) for further investigation and potential regulation. PFAS comprise a large group of chemicals. Besides PFOA and PFOS, trace amounts of perfluorononanoic acid (PFNA), mainly resulting in the use of aqueous film forming foam, also present in contaminated soil, groundwater, and higher levels have been detected in human blood. In WEs from wastewater treatment plants (WWTPs) that employ secondary or tertiary treatment, up to 1.1 µg/L of PFOA, 0.2 µg/L of PFOS , and 0.4 µg/L of PFNAhave been detected. Thus it is important to remove/reduce PFAS in WEs before reclaiming WEs as irrigation water. As the current treatment technologies (mainly membrane filtration, and granular-activated carbon adsorption) are either ineffective, costly, or generate ancillary waste streams that need to be further treated, in this study a novel, cost-effective, and environment-friendly photocatalytic solution is fully developed and used to remove and degrade PFAS in reclaimed WEs.
Zero-valent iron (Fe0) nanoparticles (NPs) are used for the first time in photoreactors under ultraviolet (UV) light to induce PFAS photocatalytic removal from WEs, both with and without the presence of oxygen, and compared to under visible light and in the dark conditions. PFAS focused in this study include PFOA, PFOS, and PFNA. The specific objectives are: 1) to investigate PFAS degradation for the first time with and without oxygen in deionized water, by Fe0 NPs under visible light, UVA (315 nm ~ 400 nm), or UVC (100 nm ~ 280 nm) illumination versus dark conditions; 2) to discern the degradation mechanisms of PFOA, PFOS, and PFNA; 3) to investigate and demonstrate PFAS degradation in WEs at the bench-scale with the optimal method developed; and 4) to separate the used iron particles by precipitation and a magnetic field, and to test the concentrations of Fe ions in finished water. The overall scope is to establish a scalable method for effective PFAS removal and degradation from WEs for water reuse. Regions in the U.S. that lack freshwater supplies, have practiced and are planning for WE reuse, and have detected PFAS in WEs would benefit from the project. In addition, the successfully treatment method could also be applied to PFAS contaminated groundwater and surface water remediation.
Summary/Accomplishments (Outputs/Outcomes):
Optimal method
PFAS (1 mg/L) were most effectively removed under UVC (254 nm) light by 100 mg/L of Fe0 NPs after 72 h without presence of O2. The removal rate was in general PFNA>PFOA>PFOS, and the removal efficiency was >99.9, 98±2, and 99±1%, respectively, for PFNA, PFOA, and PFOS (Table 1). Without Fe0 NPs (only under UVC illumination), PFAS removal was less effective, with values of 86±2, 45±1, and 82±1%, respectively, for PFNA, PFOA, and PFOS for the same reaction time period.
| C?Co | Without O2 | With O2 | ||||
| PFNA | PFOA | PFOS | PFNA | PFOA | PFOS | |
| UVC | 0.00+-0.00 | 0.02+-0.03 | 0.01+-0.00 | 0.00+-0.00 | 0.13+-0.07 | 0.22+-0.06 |
| UVA | 0.85+-0.17 | 0.80+-0.01 | 0.54+-0.13 | 0.95+-0.08 | 0.87+-0.18 | 1.17+-0.12 |
| Visible Light | 0.99+-0.18 | 0.85+-0.04 | 0.48+-0.16 | 0.90+-0.10 | 1.15+-0.14 | 0.73+-0.01 |
| Darkness | 0.96+-0.17 | 1.04+-0.09 | 0.85+-0.07 | 0.86+-0.10 | 0.82+-0.04 | 0.85+-0.01 |
Table 1. C/Co of PFAS under different illumination conditions after 72h (Co= 1mg/L).
With and without O2
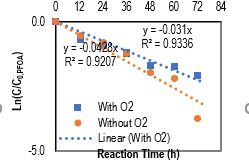
With presence of O2, Fe0 NPs also accelerated PFAS degradation under UVC light; however, PFAS was removed slower compared to the case without O2 presence (Fig. 1), exhibiting removal percentages of >99.9, 87±7, and 78±6% after 72 h by 100 mg/L of Fe0 NPs for 1 mg/L of PFNA, PFOA, and PFOS, respectively (Table 1).
Figure 1. PFOA (1mg/L) degradation under UVC with 100 mg/L FE NPs without or with presence of O2.
3) Light conditions
PFOA has a strong spectral absorption from the deep UV-region to 220 nm and a weak, broad absorption from 220 to 270 nm [21]. Our study showed PFNA has a similar absorption range; however, no apparent absorption of PFOS was shown in this range. Under darkness and under visible light, no F- ions were detected, though very little of the PFAS was removed (Table 1), especially for PFOS under visible light, which may be attributed to absorption of PFAS onto iron particles. Under UVA (350 nm), low amount of F- ions were detected, and maximum of 38.5±3.1% defluorination efficiency for PFOS was reached without O2 by 100 mg/L of Fe0 NPs.
Reaction time and dosage of Fe0 NPs
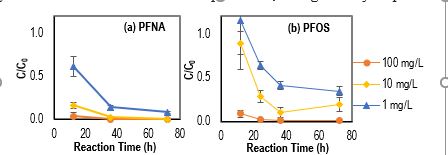
PFAS removal generally increased with reaction time up to 72 h, and generally improved with increasing dosage of Fe0 NPs up to the maximum loading of 100 mg/L under UVC (Fig. 2).
Figure 2. PFAS (1mg/L) removal under UVC by FeO NPs (1-100 mg/L) without presence of O2.
5) Degradation products and mechanism
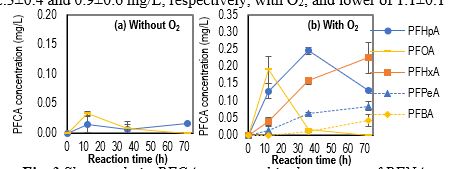
PFOA and PFNA possess similar degradation byproducts of PFHpA, PFHxA, PFPeA, PFBA (Fig. 3), and F- ions. The generation and reduction of shorter-chain PFCAs with presence of O2, e.g., PFOA and PFHpA for PFNA degradation indicated the shorter-chain PFCAs were also degraded with time after their generation (Fig. 3b). Without O2, the lack of shorter-chain PFCA products may indicate fast degradation of these species under UVC by Fe0 NPs (Fig. 3a). F- ions were also detected for PFOS, however, shorter-chain PFCAs were not detected, which is consistent with the presence of other degradation mechanisms of PFOS in comparison to PFNA and PFOA. The percentages of dissociated F- ions were 98.7±2.7, 62.3±2.3, and >99.9%, respectively, for PFNA, PFOA, and PFOS under UVC by 100 mg/L Fe0 NPs after 72 h. Both Fe2+ and Fe3+ were detected in finished water (with or without the presence of oxygen) from Fe0 NPs under UVC light illumination. For example, the concentrations of Fe2+ and Fe3+ after PFNA degradation for 72 h under UVC in the presence of 100 mg/L of Fe0 NPs were 2.3±0.4 and 0.9±0.6 mg/L, respectively, with O2; and lower of 1.1±0.1 and 0.4±0.3 mg/L, respectively, without O2. With presence of O2, rapid consumption of Fe3+ indicated possible formation of a [C7F15-COO-Fe]2+ complex that initiated PFOA and PFNA degradation.
Figure 3. Shorter-chain PFCAs generated in the process of PFNA degradtion under UVC by 100 mg/L of Fe0 NPs.
6) PFAS degradation in WEs
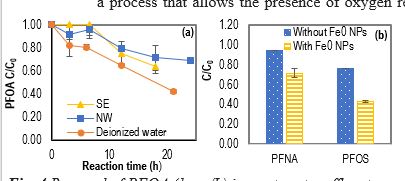
Degradation efficiencies of PFAS (1 mg/L) in WEs were lower than that in deionized water (e.g., <58±2% for PFOA after 21 h), likely reflecting the complex compositions in those environmental media (Fig. 4a). F- was also detected as a PFAS degradation byproduct in WEs, and shorter-chain PFCAs were detected as byproducts for PFNA and PFOA. PFAS of 1 µg/L were partially removed under UVC by 100 mg/L of Fe0 NPs within 30 min with presence of O2 (e.g., 28±4% for PFNA, and 56±2% for PFOS), and the removal efficiency was higher than using UVC alone (Fig. 4b); better removal was achieved with presence of O2 than without presence of O2. In addition, following bench-scale application of our approach, used iron NPs were effectively separated from the treated water by sedimentation followed by application of magnetic field. The concentrations of Fe2+ and Fe3+ in some representative samples of the finished water have been quantified to be less than 2.5 mg/L each. By using Fe0 NPs, no extra anions were brought into WEsfrom the low-toxicity Fe0 NPs, and easy of removal of the magnetic iron particles from the finished water for reuse made the technology environment-friendly and cost-effective.
ii) Technical Effectiveness, Economic Feasibility, Solution for Environmental Problems
For PFAS concentrations up to 1 mg/L, which is more than a thousand times higher than the normal concentrations of PFAS in WEs [11, 22-25], the proposed method effectively removed >98% of PFAS in 72 h without oxygen. In WEs, PFAS were better removed in our study under the presence of oxygen than without oxygen. As in most cases WEs contain oxygen after treatment, a process that allows the presence of oxygen reduces the cost of the technology. In addition to removal of PFAS, the photoreactor is also efficient for bacteria inactivation: e.g., >99.9% of Escherichia coli was inactivated after 30 min reaction time for WEs from both WWTPs. Moreover, the price of Fe0 NPs is low, ~$50-110/kg [58]. PFAS translocation to crops, pollution to soil, surface water and groundwater, and impact on human health from PFAS contaminated WE reuse can be diminished with successful PFAS removal and degradation by this technology. In addition, safe WE reuse could be promoted with the success of the technology, and millions of tons of water could be saved from potentially increased wastewater reuse practices.
Figure 4. Removal of PFOA (1mg/L) in wastewater effluents (WEs) from Southeast (SE) and Northwest (NW) WWTPs with O2 (a), and removal of PFNA and PFOS (1 ug/L each) in NW WEs after 30 min with O2 (b).
Conclusions:
PFAS removal and degradation were investigated for the first time by using Fe0 NPs under UVC, UVA, visible light, and compared to conditions of darkness. It was found both without and with presence of O2, PFAS removal was accelerated by introducing Fe0 NPs under UVC light compared to UVC light alone. Higher removal of PFAS was obtained without presence of O2 under UVC light by Fe0 NPs. PFHpA, PFHxA, PFPeA, PFBA, and F- ions were detected as main byproducts for PFOA and PFNA degradation. However, for PFOS, no shorter-chain PFCAs were detected, whereas only fluoride ions were detected, which indicated other degradation pathways for PFOS under such conditions exist. For 1 mg/L of PFAS, almost all PFAS were removed in 72 h without O2 under UVC by Fe0 NPs; the defluorination efficiency was >96.0% for both PFNA and PFOS, and >60.0% for PFOA. Fe2+ and Fe3+ ions were detected during the degradation process using Fe0 NPs under UVC both with and without O2. With presence of O2, rapid consumption of Fe3+ indicated possible formation of complexes that initiated PFOA and PFNA degradation.
Overall, Fe0 NPs under UVC light is an innovative, cost-effective, and environment-friendly solution for PFAS decomposition in environmental media for water reuse. Without the need for addition of extra anions given the presence of the low-toxicity Fe0 NPs, along with easy removal of the used iron particles from the treated water by exploiting their magnetic property, made the technology of PFAS removal by Fe0 NPs under UVC light even attractive.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 7 publications | 3 publications in selected types | All 3 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Chunjie Xia, Jia Liu (2020). Degradation of perfluorooctanoic acid by zero-valent iron nanoparticles under ultraviolet light. Journal of Nanoparticle Research. 22:188 |
SU839460 (Final) SU840174 (Final) SV840022 (2021) |
Exit Exit |
|
|
Aryal R, Xia C, Liu J. 1, 4‐Dioxane‐contaminated groundwater remediation in the anode chamber of a microbial fuel cell. Water Environment Research 2019;91(11):1537-45. |
SU839460 (Final) |
not available |
|
|
Liu J, Williams PC, Goodson BM, Geisler-Lee J, Fakharifar M, Gemeinhardt ME. TiO2 nanoparticles in irrigation water mitigate impacts of aged Ag nanoparticles on soil microorganisms, Arabidopsis thaliana plants, and Eisenia fetida earthworms.Environmental research 2019;1;172:202-15. |
SU839460 (Final) |
not available |
Supplemental Keywords:
nZVI, nanomaterial, photolysis, water resource, water recyclingRelevant Websites:
SIU researchers test method to remove contaminants from water Exit
P3 Phase II:
PFASs Removal by Photocatalysis for Water Reuse | 2021 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.