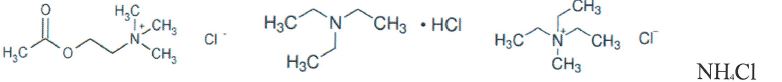Grantee Research Project Results
2017 Progress Report: Reusable Biodegradable Solvents from Biodiesel
EPA Grant Number: SU836761Title: Reusable Biodegradable Solvents from Biodiesel
Investigators: Ott, Lisa
Current Investigators: Ott, Lisa , Stone, Janine M , Bayham, Jude
Institution: California State University - Chico
EPA Project Officer: Page, Angela
Phase: I
Project Period: November 1, 2016 through October 31, 2017 (Extended to January 31, 2018)
Project Period Covered by this Report: November 1, 2016 through January 31,2018
Project Amount: $15,000
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet (2016) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Chemical Safety , Sustainable and Healthy Communities
Objective:
The purpose of Phase I was to improve biodiesel manufacturing by designing a sustainable, profitable solution to the industry's waste glycerol problem. The Phase I objectives were:
- Prepare a series of DESs using different HBAs in pure glycerol and glycerol from AC biodiesel synthesis. Since these DESs are novel, the most important goal is to measure key physical properties such as density and viscosity.
- Evaluate DESs for their utility to replace traditional solvents for organic reactions.
- Determine which of the processes used to create DES from waste glycerol would be financially beneficial for the university, both immediately and over the projected 10-semester "life" of their use.
- Create a working cost-benefit analysis spreadsheet that would show costs of producing DES under a range of assumptions (inputs used, time frame for use, discount rate, costs of traditional solvents).
The overall scope of Phase I was green chemistry and green business. The scope of work involved using a waste product to develop a "green" chemical, developing a chemical solvent that has potential for production beyond an experimental prototype, and working with universities within the California State University (CSU) system to strive to develop a waste-free, cost-effective process. Additionally, the results of the economic analysis could be adapted for use by a small-scale biodiesel producer for use in determining potential profits from producing and selling DES as opposed to paying for disposal of cogenerated glycerol.
Progress Summary:
From Phase I, our research objectives were to:
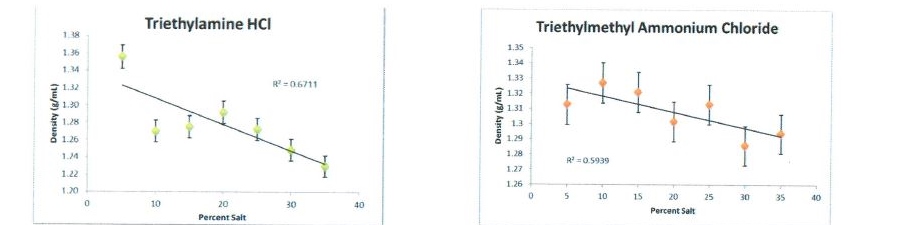
- Examine a variety of HBAs: Of the four HBAs the literature suggested would be favorable for this study (see Figure 1), our team prepared DESs with the three of them that we had not yet studied. Triethylamine-HCl, triethylmethyl ammonium chloride, and acetylcholine chloride formed liquid mixtures with waste glycerol in all ratios from 5-35 weight percent. Ammonium chloride, however, formed a solid in all quantities above 10%; consequently, we abandoned evaluating it. Density measurements were made on DESs prepared with the three remaining HBAs in both pure glycerol and waste glycerol (see Figure 2). While there is a deviation from linearity , all of the measured densities were lower than that of the initial glycerol density. Therefore, we have strong evidence for the formation of DESs using these simple, inexpensive HBAs.
Figure `1. The four HBAs studied in hase I of this project.
Figure 2. Density measurements for DESs prepared in glycerol from AC biodiesel synthesis.
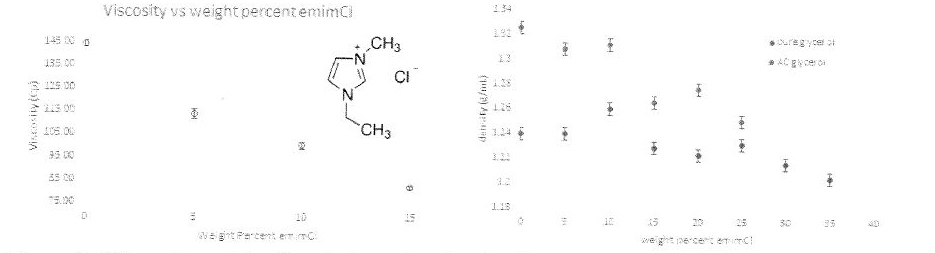
2. Evaluate a suite of DESs: The Chem 483 students involved in Phase I research selected their own candidates for HBAs. They chose to examine DESs prepared with formamidinium iodide and 1-ethyl-3-methylimidazolium chloride (emimCl; structure shown as an inset in Figure 3 below) as potential HBAs. Importantly, these students discovered that the attempted DES prepared with pure glycerol and FAI did not form a DES; instead, the density went up as the concentration of the salt increased. This mixture, then, is just the dissolution of an ionic compound in a covalent solvent and requires no further investigation. Conversely, these students determined that the emimCl followed the predicted trend of decreasing density and viscosity with increasing mass percent HBA.
Figure 3. Viscosity and density of glycerol and emimCI DESs
Progress on this research objective was slowed by the malfunctioning of the ViscoLab 3000 viscometers that had recently been acquired by PI Ott. We had to package the viscometer up and send it back for testing in Massachusetts, which significantly hampered our progress. It was not until mid-February that the viscometers were back in California, up, running, and measuring accurately again. Therefore, most the measurements reported herein were collected by the Chem 483 students instead of the students supported by the EPA P3 grant. As of this writing, more measurements are taking place on the viscometer to test the physical properties of the three HBAs used to create DESs in the Phase I proposal.
- Transfer the DESs process from AC to BC biodiesel glycerol: The undergraduate students in Chem 483 made excellent progress on a method for cleaning up BC glycerol. Purification of the BC glycerol layer was accomplished using a six step method: acidification with aPO4, phase separation, neutralization with KOH, removal of water, methanol extraction, and filtration through a short column of activated carbon.8 While this method was an effective method for purifying BC glycerol, it is a far cry from the established tenets of green chemistry.7 Josh Crane, a student supported by the EPA P3 grant, also determined that glycerol produced from biodiesel synthesis using a potassium hydroxide catalyst (instead of the more commonly used sodium hydroxide catalyst) is more amenable to DES formation.
- Develop a protocol for rapid evaluation of parameters: There are many challenges with industrial waste glycerol mixtures, including physical state and variability in its physical and chemical properties. These properties can vary widely, and that variation primarily depends on the feedstock triglycerides, reaction conditions, and separation method from the rawbiodiesel. Our ability to evaluate these parameters was hampered significantly by the shutdown of Springboard Biodiesel' s retail arm. There is no other commercial producer of biodiesel in the North State region of California; however, we have recently secured an agreement with Sierra Nevada Brewing Company, which has a small-scale biodiesel production machine. They have agreed to supply us with 10 gallons of glycerol from their machine every 3-4 weeks. With this new supply stream of biodiesel, this protocol development will now be possible.
- Develop a business plan for a small-scale biodiesel producer: In our initial plans, our students aimed to research and collect data from Springboard Biodiesel to calculate the cost of each DES process. The student team aimed to evaluate potential cost savings associated with decreased need to dispose of waste glycerol, analyze the current market opportunities for DESs, and determine the potential revenues associated with marketing DES as a biodiesel co-product. However, Springboard Biodiesel's decision to terminate their manufacturing and distribution arm made much of this work impossible. Despite the loss of Springboard as a partner, the economics students created a spreadsheet-based model that could be used by another biodiesel producer to determine potential profits from selling DES as a biodiesel co-product. Students determined that DES can be produced at a cost lower than the cost of commonly-used solvents, indicative of a large potential market for their use.
- Showcase research: The initial dissemination of this research was carried out at CSU, Chico's annual This Way to Sustainability Conference on March 24th, 2017. In a session attended by approximately 25 students, faculty, and community members, the P3 students gave an hour-long presentation. The student team is registered for and planning to present their research at the National Sustainable Design Expo in Washington D.C in May. Additionally, our poster has been accepted for presentation at the College ofNatural Sciences' Poster Session on May 10, 2017.
- Integrate research into classroom: Initially, we intended to use these novel solvents in the Department of Chemistry's Integrated Laboratory series during Phase II; instead, PI Ott had the seven students in class (Chem 483) investigate the preparation of DESs during Phase I. These students made significant progress with preparing DESs from both pure and AC glycerol, cleaning up BC glycerol, and investigating a prototype organic reaction in the one of the DES.
- Publish results: We should have sufficient data to publish in the Journal of Chemical and Engineering Data after two to three semesters in which Chem 483 classes synthesize and characterize these DESs with a variety of different HBAs.
Of the six potential HBAs evaluated during Phase I, four were found to result in DESs when complexed with glycerol prepared from AC biodiesel synthesis. All four were characterized by density measurements with appropriate propagation of error. Additionally, the same DESs were prepared with pure glycerol as the HBD for reference. Viscosity measurements were initially hampered by our malfunctioning viscometer, but are in progress in PI Ott's labs. An organic reaction was successfully completed in a DES; optimization of the reaction conditions for this particular reaction is necessary during Phase II.
DESs prepared using the four potential methods were evaluated by the economics students to determine their potential profitability both in the short and long runs. DESs can be prepared from waste glycerol in order to replace CSU, Chico's entire annual volume of volatile organic solvent purchases-58 liters of solvents per year. The only input needed to produce DES is waste glycerol (no cost) and the chemical salts that may be added to the DES at varying concentrations (5%-20%). Costs of DES production increase (linearly) with concentration of salt used; because it is not-yet known which concentrations will yield DESs with desired chemical properties, all concentrations were considered in sensitivity analysis. Students developed a spreadsheet-based model to determine net benefits of replacing traditional solvents with DES in year one; these benefits were found to be positive for all but one of the inputs used at the low concentration (5%). However, because DESs can be reused for up to 5 years (10 semesters), total net benefits from DES production are calculated by comparing the one-time costs of DES production to the (discounted) cost of purchasing traditional solvents in years one through five and disposing of resulting chemical waste on an annual basis.
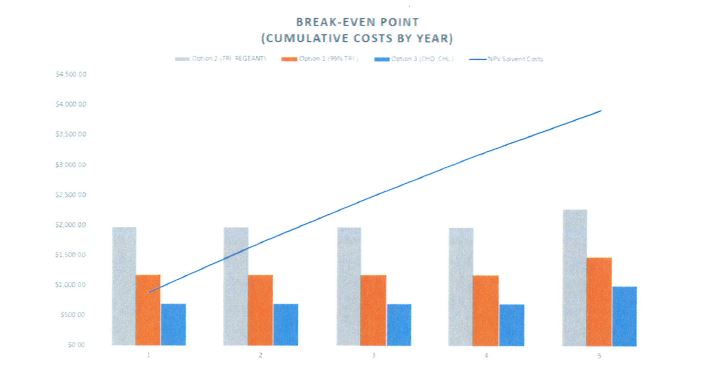
Results below show our most conservative scenario, assuming that high concentrations of input salts are used and that disposal costs for traditional solvents are at their minimum. Table 2 shows the net present value of savings associated with DES production at 20% salt concentration after 5 years. Figure 4 shows the costs of producing DES via each of the potential inputs discounted at a rate of 3%. Vertical bars representing costs at the end of years 1-5; the blue line for net present value of solvent costs shows how total solvent costs increase over time with annual purchase and disposal. Our analysis finds that DES produced using any of the examined inputs (again, with the exception of acetylcholine chloride) will yield positive net benefits within three years at a minimum. Savings will increase if lower concentrations of inputs prove viable.
Results for efficiency of DES in use in the chemistry labs will determine inform final results for potential net benefits associated with the use of DES in the CSU system.
| DES Option | Net Present Value of Savings (r=3%) |
| Triethylamine, 99% Pure | $2645.38 |
| Triethylamine Reagant | $1848.46 |
| Acetylcholine Chloride | $-14649.82 |
| Choline Chloride | $3128.62 |
Table 2. Savings analysis.
Figure 4. Break even analysis.
The measured densities and available viscosity data indicate that using cogenerated glycerol from biodiesel synthesis as a component in DESs results in solvents with comparable physical properties to DESs prepared with pure glycerol. Therefore, our DESs are viable replacements for those prepared with virgin chemicals. Using cogenerated glycerol to prepare DESs reduces the waste stream from biodiesel production and yields a potential value-added product to commercial biodiesel producers. Additionally, since the DESs prepared with Phase I support are reusable, we are preventing waste generation wherever the DESs are used and re-used. If these DESs are used in organic teaching laboratories, thousands of students will be positively impacted by learning about the sustainable principles in practice. Additionally, using these DESs in an organic teaching laboratory is a benefit to the human health of the students, instructional staff, and support staff, as DESs have no measureable volatility and can do no harm via inhalation.
Finally, because DESs are reusable for up to ten semesters, as opposed to volatile organic solvents, which must be disposed of every year, use of DESs may achieve significant reductions in externalities. These externalities include both student's risk of exposure to traditional solvents and emissions/ risk of water contamination that occurs when wastes are transported and either landfilled or incinerated by the waste management company.
Future Activities:
P3 Phase II Project Description
The proposed project is innovative research in sustainable chemistry, investigating DESs prepared with a waste-byproduct. DESs are currently under intense investigation in the chemical literature, garnering interest as a renewable, recyclable, non-volatile, biodegradable alternative to conventional volatile organic solvents. A number of groups are currently reporting on investigations of organic reactions using DESs prepared from pure reagents, but to our knowledge no other groups are using a waste by-product as one of the components in the DES. Initial trials were performed by undergraduate students in Chem 483 during Phase I showed promising results, and during Phase II, the plan is to further research DESs by investigating a variety ofreduction reactions,, 9t o condensation reactions (such as the expand to organometallic reactions. 14Knoevenagle condensation 11 and the Pictet-Spengler reaction12 Diels-Alder reactions,13 and expand to organometallic reactions.
The utilization of a waste product as a component in producing a marketable, useable solvent significantly changes the traditional school of thought for preparing chemical solvents. This sustainable approach reduces waste from the ever-increasing production of biodiesel; produces a green chemical that is renewable, recyclable, non-volatile, and biodegradable; and provides a cost-effective alternative to the hazardous chemical solvents used ubiquitously for chemical research in universities and elsewhere.
In a university laboratory setting, the actual risk of chemical exposure may be higher for students than that estimated at an industry-wide scale, because of the students' relative inexperience with handling volatile organic compounds. We aim to challenge the current standard of using hazard chemical solvents in University laboratories by creating a strong case for substituting with DESs. We will test the production ofDESs, their economic feasibility, and pilot the use ofDESs in our organic teaching labs at CSU, Chico. If we prove successful, we plan to expand our pilot study to other campuses within the 23-campus CSU system. There is literature precedent for using DESs in teaching labs;15 we aim to build on this success of a DES made from virgin chemicals by creating our DESs from reused cogenerated glycerol.
Journal Articles:
No journal articles submitted with this report: View all 2 publications for this projectProgress and Final Reports:
Original AbstractP3 Phase II:
Reusable Biodegradable Solvents from Biodiesel | 2018 Progress Report | 2019 Progress Report | 2020 Progress Report | 2021 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- Original Abstract
- P3 Phase II | 2018 Progress Report | 2019 Progress Report | 2020 Progress Report | 2021 Progress Report | Final Report