Grantee Research Project Results
2017 Progress Report: The Role of Obesity in Biological Responses to Particulate Matter in Mice
EPA Grant Number: R836152C003Subproject: this is subproject number 003 , established and managed by the Center Director under grant R836152
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Center for the Study of Childhood Asthma in the Urban Environment
Center Director: Hansel, Nadia
Title: The Role of Obesity in Biological Responses to Particulate Matter in Mice
Investigators: Polotsky, Vsevolod Y
Institution: The Johns Hopkins University
EPA Project Officer: Callan, Richard
Project Period: September 1, 2015 through August 31, 2019 (Extended to August 31, 2021)
Project Period Covered by this Report: September 30, 2016 through June 30,2017
RFA: Children's Environmental Health and Disease Prevention Research Centers (2014) RFA Text | Recipients Lists
Research Category: Children's Health , Human Health
Objective:
The overarching hypothesis of Project 3 is that obesity exacerbates the particulate matter (PM)-induced AHR due to pathogenic effects of adiposity and comorbid sleep apnea and that the effects of obesity are mediated via IL-6. Furthermore, these detrimental effects of obesity can be attenuated by weight loss and sleep apnea treatment. Our hypothesis will be addressed in two Specific Aims:
Specific Aim 1, we will test causal links between obesity and the PM-induced AHR and inflammation. (A) We will quantify the PM-induced AHR and inflammation in lean and DIO wildtype and IL-6 knockout mice; (B) To link to potential therapeutic actions, we will quantify the ability of weight loss to attenuate the PM-induced AHR and inflammation.
Specific Aim 2, we will examine effects of sleep apnea on the PM-induced AHR and inflammation. We will quantify the PM-induced AHR and inflammation in DIO mice with recurrent upper airway obstruction during sleep caused by genetically induced excessive tongue adiposity. Experiments also will assess the effects of sleep apnea treatment, which will be modelled by supplemental oxygen.
Progress Summary:
Experiments performed during Year 1 of the award have demonstrated that diet-induced obesity (DIO) by feeding a high fat diet (HFD) for 18-20 weeks caused airway hyperresponsiveness (AHR) and neutrophilic inflammation. However, PM exposure did not seem to increase AHR.
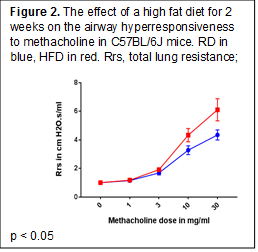
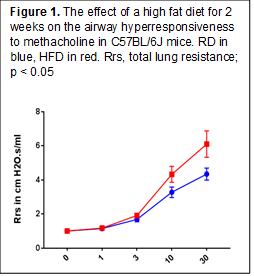
Experiment 1.1. Exposure to HFD for 2 days did not affect body weight, AHR or BAL data, but there was an increase in IL-1β expression in lung tissue (Fig.1). Exposure to HFD for 14 days induced a small but statistically significant weight gain (29.2 ± 2.4 g vs 26.6 ± 2.1 in the RD group, p < 0.05). There was a significant increase in the AHR (Fig. 2) and lung IL-1β expression without any change in the BAL cell count and differential. Conclusion: HFD for 2 weeks increased AHR in association with overexpression of IL-1β in lung tissue.
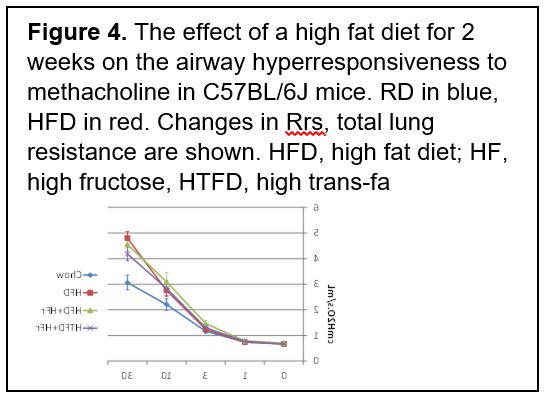
Experiment 1.2. We exposed mice to RD, HFD, High Trans-Fat Diet (HTFD, 4.73kcal/g, 40% kcal from trans-fat) and D-Fructose (HF, 30% solution, 1.2 Kcal/mL) for 8 weeks followed by AHR measurements and BAL analysis as above. As expected, hypercaloric diets induced similar weight gain (Fig. 3). All hypercaloric diets markedly increased AHR (Fig. 4). There was an increase in neutrophils in BAL, especially in the HTFD+HF. The final data analysis is still pending.
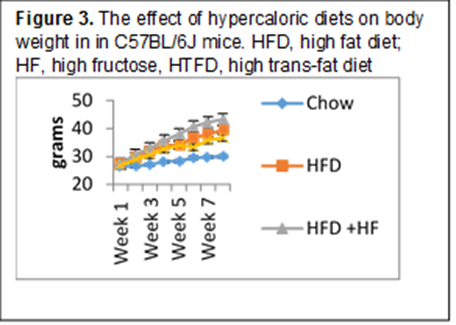
Experiments 1.1 and 1.2: Hypercaloric high fat diets increase AHR. To discriminate effects of diet and obesity, we will repeat Experiment 1.2 with the food restriction protocol matching weights in RD and HFD groups. We also will perform IL-1β in lung tissue. If increases in IL-1β are associated in an increase in AHR, we will perform experiments in IL-1β KO mice to examine the role of this cytokine in HFD induced airway hyper-responsiveness.
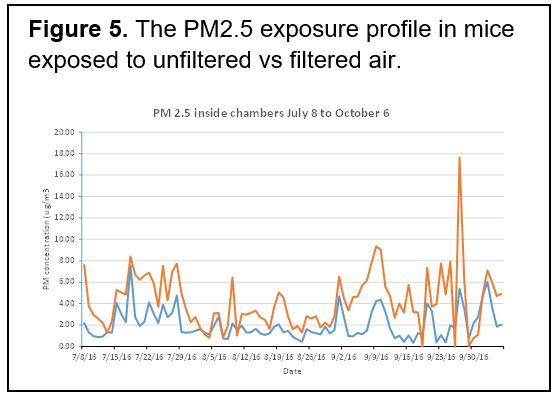
Experiment 2.1. Results: Our experiments demonstrated that the level of PM2.5 air pollution at our downtown Baltimore location is low (Fig. 5). There was no significant effect of this level of pollution on AHR. Conclusion: A route of PM administration should be optimized. Currently, we are testing intranasal PM2.5administration.
Specific Aim 2
We have previously proposed to use genetically engineered mice with excessive tongue adiposity to investigate effects of sleep apnea on AHR. Unfortunately, repeated experiments showed a large degree of variability in the amount of tongue fat induced by PPARγ overexpression. The approach in SA2 was significantly changed. We have developed a completely new mouse model of OSA. We used an alternative approach deploying designer receptors specifically activated by designer drug (DREADD) in hypoglossal motor neurons. DREADDs are G-protein coupled human cholinergic receptors, which have been chemogenetically engineered to recognize clozapine-N-oxide (CNO) but no naturally occurring mammalian ligands. At baseline, only occasional flow limited breaths were observed. In contrast, after CNO administration mice showed severe continuous inspiratory flow limitation, which was particularly striking in NREM sleep (46% of all breaths were flow limited after CNO compared to 7% at baseline). Thus, inhibition of hypoglossal motor neurons causes sleep disordered breathing even in the absence of anatomic pre-disposition (e.g., obesity). The inhibitory DREADD did not induce significant oxyhemoglobin desaturations in lean mice, but we fully expect that it will exacerbate sleep apnea in DIO mice and the effect will depend on the targeted population of motor neurons. We will use this approach in our SA2.
Supplemental Keywords:
Obesity sleep, hyperresponsiveness, mice, particulate matter, PM, asthma, childrenProgress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R836152 Center for the Study of Childhood Asthma in the Urban Environment Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R836152C001 Investigating obesity as a susceptibility factor for air pollution in childhood
R836152C002 Novel exposure metrics for assessing the effects of ultrafine and fine particulate matter on asthma in children
R836152C003 The Role of Obesity in Biological Responses to Particulate Matter in Mice
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.