Grantee Research Project Results
2015 Progress Report: Organotypic Culture Model to Analyze DevelopmentalLimbMalformationsResulting from Toxicant/Teratogen Exposure
EPA Grant Number: R835736C002Subproject: this is subproject number 002 , established and managed by the Center Director under grant R835736
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Mickey Leland National Urban Air Toxics Research Center (NUATRC)
Center Director: Beskid, Craig
Title: Organotypic Culture Model to Analyze DevelopmentalLimbMalformationsResulting from Toxicant/Teratogen Exposure
Investigators: Tuan, Rocky , Alexander, Peter
Current Investigators: Tuan, Rocky
Institution: University of Pittsburgh
Current Institution: University of Pittsburgh , Vanderbilt University
EPA Project Officer: Aja, Hayley
Project Period: December 1, 2014 through November 30, 2018 (Extended to November 30, 2019)
Project Period Covered by this Report: December 1, 2014 through November 30,2015
RFA: Organotypic Culture Models for Predictive Toxicology Center (2013) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Our goal is to develop robust in vitro three-dimensional (3D) organotypic culture models (OCMs) based on human mesenchymal stem cells (MSCs) to first examine critical phenomena of embryonic limb development that are prime targets of limb teratogenesis, and then examine their susceptibility to perturbation by known and candidate teratogens and environmental toxicants.
Progress Summary:
Project 2 has demonstrated successful chondrogenesis and hypertrophy of adult human MSCs in planar high-density culture comprised of 400,000 cells each. Using these cultures, timelines of chondrogenic and hypertrophic (1) gene expression, (2) matrix elaboration and (3) histochemical and immunohistochemical marker detection in both the chondrogenic and hypertrophic cultures are under development. In this process, baseline values for endpoint analyses and detection limits of chondrogenic and hypertrophic OCMs are being determined. In collaboration with Project 5, development of a new mechanical stimulator for the joint segmentation model amenable to microscopy and microfluidics that maintains the region-of-interest in a focal plane is progressing.
The effect of three known teratogens (valproic acid, warfarin and thalidomide) on adult human MSC micromass culture chondrogenesis and hypertrophy also has been tested. Using outcomes based on RT-PCR, immunohistochemistry and biochemistry, effective doses approximating reported maternal plasma levels have been observed. Specific mechanisms of OCM disruption are under investigation. Initial tests for the effect of two candidate teratogens (bisphenol A and mifepristone) on adult human MSC chondrogenesis and hypertrophy also have been performed. Testing of limb developmental stage-specific promoter-reporter constructs and ELISAs to detect extracellular matrix metabolites to monitor OCM development and maturation noninvasively is underway.
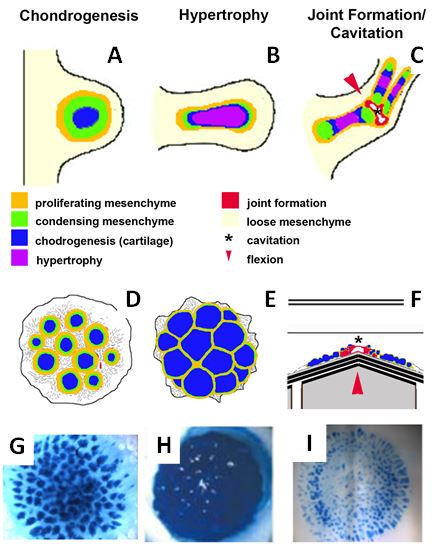
Figure 1: Organotypic culture design. (A-C) Development of the axial cartilaginous skeletal model. (A) The limb bud, the earliest morphogenic indication of limb development, is a protrusion from the lateral plate mesoderm that is initially comprised of the ectoderm overlying specified but undifferentiated mesenchymal cells that proliferate, condense, and differeniate into cartilage. (B) As the limb bud grows and elongates, the cartilage matures and undergoes hypertrophy, a step required for future endochondral ossification. The process of joint formation involves alignment of cells along the site of the future joint forming the interzone that undergoes cavitation (shown in red) resulting in a joint space, proximal and distal joint surfaces and capsular lining. Stages of limb development can be replicated in vitro using the 3D planar high density micromass in various configurations (D-F). (D) Chondrogenesis procedes as a function of commitment to the cartilage lineage at the time of explantation, forming discreet nodules within 3 days of plating in a manner very similar to that in vivo. Hypertrophy in these cultures can be induced through the application of T3 at day 10 of culture, inducing nodule expansion and hypertrophy of the constituent cells. (F) Mechanical stimulation of a chondrogenic culture at day 3 can stimulate joint gene expression along regions (red) of localized stretch (red arrowhead). (G-H) Representative cultures using chick embryonic limb bud undergoing chondrogenesis, hypertrophy, or segmentation stained with Alcian blue – specific for cartilage-specific sulfated proteoglycans.
Future Activities:
Project 2 will adapt chondrogenic and hypertrophic cultures to a format suited for existing microfluidic hardware developed by members of Project 5. This will involve reducing the cell number to 40,000 cells in 2 microliter droplets and culturing the cells on a porous membrane, such as 0.4 micron polycarbonate membranes.
Using these smaller cultures amenable to medium throughput analysis, we will develop tools and techniques for noninvasive analysis. They will involve: (1) determining the utility of tissue-specific promoter-reporter constructs in evaluating chondrogenesis hypertrophy and joint segmentation noninvasively, and (2) correlating changes in extracellular matrix metabolite detection detected by ELISA in the culture effluent (i.e., noninvasively) with changes in tissue development and maturation as detected by destructive endpoint analyses. This is also the OCM format we will use to develop microfluidic modalities for limb development OCMs in collaboration with Project 5.
Two additional aspects of culture development and maturation will be undertaken in collaboration with Project 4. First, the effect of liver conditioned medium in the presence or absence of metabolized (bioactivated) teratogens on OCM development will be assayed using a conditioned medium approach. Second, we will correlate changes in chondrogenesis and hypertrophy endpoint analyses and/or noninvasive matrix metabolite detection and promoter-reporter activity with cell stress and cell death and apoptotic biosensor activity.
Journal Articles:
No journal articles submitted with this report: View all 6 publications for this subprojectSupplemental Keywords:
micromass culture, adult human mesenchymal stem cell chondrogenesis, hypertrophy, joint segmentationRelevant Websites:
Center for Cellular and Molecular Engineering Exit
The Vanderbilt-Pittsburgh Resource for Organotypic Models for Predictive Toxicology Exit
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R835736 Mickey Leland National Urban Air Toxics Research Center (NUATRC) Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R835736C001 Mammosphere Bioreactor For Life-Stage Specific Toxicology
R835736C002 Organotypic Culture Model to Analyze DevelopmentalLimbMalformationsResulting from Toxicant/Teratogen Exposure
R835736C003 Validating a fetal membrane on a chip model for characterizing
reproductive toxicant exposure risks
R835736C004 Organotypic Liver Model for Predictive Human Toxicology and Metabolism
R835736C005 Systems Engineering & Analysis for Organotypic Culture Models
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
1 journal articles for this subproject
Main Center: R835736
169 publications for this center
56 journal articles for this center