Grantee Research Project Results
Final Report: Development of a Heterogeneous Catalyst for Hydroformylation in Supercritical CO2
EPA Grant Number: R828206Title: Development of a Heterogeneous Catalyst for Hydroformylation in Supercritical CO2
Investigators: Abraham, Martin A. , Davies, Julian A. , Mason, Mark R.
Institution: University of Toledo
EPA Project Officer: Hahn, Intaek
Project Period: July 1, 2000 through June 30, 2003 (Extended to June 30, 2004)
Project Amount: $315,000
RFA: Technology for a Sustainable Environment (1999) RFA Text | Recipients Lists
Research Category: Pollution Prevention/Sustainable Development , Sustainable and Healthy Communities
Objective:
Approach:
This proposal seeks to eliminate potentially hazardous organic solvents through two novel developments:1. Perform the target reaction in the benign reaction solvent supercritical CO2, and
2. Develop a heterogeneous catalyst for the target reaction.
The use of CO2 as a reaction solvent offers optimal environmental performance because CO2 does not deplete the ozone layer, does not contribute to ground-level smog, and will not contribute to global warming. The use of CO2 by-product from existing commercial and natural sources will ensure that no net increase in global CO2 results from the use of this technology.
One limitation of heterogeneous catalysis has been the inability to control selectivity. We believe, however, that modification of the catalyst and the support for optimization with scCO2 can allow the development of active and selective catalysts for the hydroformylation reaction. Use of supported metal-phosphine ligands will allow control of selectivity, similar to that seen in liquid-phase catalysis. Modification of the catalyst support, and the use of shape-selective catalysts with uniform molecular-size pores, provides further opportunity to achieve selectivity control.
Summary/Accomplishments (Outputs/Outcomes):
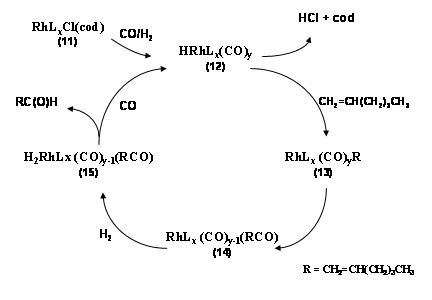
Expected Results:
The work completed through this research will demonstrate the production of commercially important chemical intermediates without the use of organic solvents. The proposed process considers an environmentally benign synthesis that can be adapted for the production of many important chemicals. These process modifications decrease the potential for volatile organic compounds (VOCs) emission in large-scale production processes, providing one means for the chemical industry to be responsive to pollution prevention requirements.Journal Articles on this Report : 12 Displayed | Download in RIS Format
| Other project views: | All 12 publications | 12 publications in selected types | All 12 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Abraham MA, Nguyen N. “Green engineering: defining the principles”— results from the Sandestin Conference. Environmental Progress & Sustainable Energy 2003;22(4):233-236. |
R828206 (Final) |
Exit Exit |
|
|
Bektesevic S, Tack T, Mason MR, Abraham MA. Analysis of the hydroformylation reaction over an immobilized catalyst in supercritical carbon dioxide. Industrial & Engineering Chemistry Research 2005;44(14):4973-4981. |
R828206 (Final) |
Exit Exit Exit |
|
|
Bektesevic S, Kleman AM, Marteel-Parrish AE, Abraham MA. Hydroformylation in supercritical carbon dioxide: catalysis and benign solvents. The Journal of Supercritical Fluids 2006;38(2):232-241. |
R828206 (Final) |
Exit Exit Exit |
|
|
Hemminger O, Marteel A, Mason MR, Davies JA, Tadd AR, Abraham MA. Hydroformylation of 1-hexene in supercritical carbon dioxide using a heterogeneous rhodium catalyst. 3. Evaluation of solvent effects. Green Chemistry 2002;4(5):507-512. |
R828206 (Final) |
Exit |
|
|
Kleman AM, Abraham MA. Asymmetric hydroformylation of styrene in supercritical carbon dioxide. Industrial & Engineering Chemistry Research 2006;45(4):1324-1330. |
R828206 (Final) |
Exit Exit Exit |
|
|
Marteel AE, Tack TT, Bektesevic S, Davies JA, Mason MR, Abraham MA. Hydroformylation of 1-hexene in supercritical carbon dioxide: characterization, activity, and regioselectivity studies. Environmental Science & Technology 2003;37(23):5424-5431. |
R828206 (Final) |
Exit Exit Exit |
|
|
Marteel AE, Davies JA, Olson WW, Abraham MA. Green chemistry and engineering: drivers, metrics, and reduction to practice. Annual Review of Environment and Resources 2003;28:401-428. |
R828206 (Final) |
Exit |
|
|
Marteel A, Davies JA, Mason MR, Tack T, Bektesevic S, Abraham MA. Supported platinum/tin complexes as catalysts for hydroformylation of 1-hexene in supercritical carbon dioxide. Catalysis Communications 2003;4(7):309-314. |
R828206 (Final) |
Exit |
|
|
Snyder G, Tadd A, Abraham MA. Evaluation of catalyst support effects during rhodium-catalyzed hydroformylation in supercritical CO2. Industrial & Engineering Chemistry Research 2001;40(23):5317-5325. |
R828206 (Final) |
Exit Exit Exit |
|
|
St John Sutton MG, Plappert T, Abraham WT, Smith AL, DeLurgio DB, Leon AR, Loh E, Kocovic DZ, Fisher WG, Ellestad M, Messenger J, Kruger K, Hilpisch KE, Hill MRS, Multicenter InSync Randomized Clinical Evaluation (MIRACLE) Study Group. Effect of cardiac resynchronization therapy on left ventricular size and function in chronic heart failure. Circulation 2003;107(15):1985-1990. |
R828206 (Final) |
Exit Exit Exit |
|
|
Tadd AR, Marteel A, Mason MR, Davies JA, Abraham MA. Hydroformylation of 1-hexene in supercritical carbon dioxide using a heterogeneous rhodium catalyst. 2. Evaluation of reaction kinetics. Industrial & Engineering Chemistry Research 2002;41(18):4514-4522. |
R828206 (Final) |
Exit Exit Exit |
|
|
Tadd AR, Marteel A, Mason MR, Davies JA, Abraham MA. Hydroformylation of 1-hexene in supercritical carbon dioxide using a heterogeneous rhodium catalyst. 1. Effect of process parameters. The Journal of Supercritical Fluids 2003;25(2):183-196. |
R828206 (Final) |
Exit Exit Exit |
Supplemental Keywords:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
