Grantee Research Project Results
2014 Progress Report: Impacts of Anthropogenic Emissions in the Southeastern U.S. on Heterogeneous Chemistry of Isoprene-Derived Epoxides Leading to Secondary Organic Aerosol Formation
EPA Grant Number: R835404Title: Impacts of Anthropogenic Emissions in the Southeastern U.S. on Heterogeneous Chemistry of Isoprene-Derived Epoxides Leading to Secondary Organic Aerosol Formation
Investigators: Surratt, Jason D.
Institution: University of North Carolina at Chapel Hill
EPA Project Officer: Chung, Serena
Project Period: April 1, 2013 through March 31, 2016 (Extended to March 31, 2017)
Project Period Covered by this Report: April 1, 2014 through March 31,2015
Project Amount: $300,000
RFA: Anthropogenic Influences on Organic Aerosol Formation and Regional Climate Implications (2012) RFA Text | Recipients Lists
Research Category: Early Career Awards , Air Quality and Air Toxics , Air , Climate Change
Objective:
The underlying hypothesis of this study is that anthropogenic emissions enhance isoprene secondary organic aerosol (SOA) formation through the heterogeneous chemistry of isoprene-derived epoxides, possibly leading to light-absorbing SOA in the southeastern United States. The specific objectives to evaluate this hypothesis include: (1) leveraging our ongoing Look Rock, TN, field site during the community-led Southern Oxidant and Aerosol Study (SOAS) in summer 2013 to evaluate how isoprene SOA formation chemistry varies between regional and urban influenced air masses; (2) evaluate the effects of relative humidity, aerosol acidity, and seed aerosol type on the heterogeneous chemistry of isoprene-derived epoxides leading to SOA and how this might yield light-absorbing aerosol constituents (i.e., brown carbon); (3) evaluate gaseous yields of epoxides from isoprene oxidation under varying initial levels of nitric oxide; (4) experimentally determine the isoprene epoxydiols (IEPOX) and methacrylic acid epoxides (MAE) reaction probability per collision with aerosol particles, γ(IEPOX) and γ(MAE), and its dependence upon aerosol composition in order to help improve parameterization of isoprene-derived SOA formation in models; and (5) determine the source of isoprene SOA at the Look Rock, TN, ground site observed during SOAS 2013 that occurs through non-epoxide routes.
Progress Summary:
A suite of offline and real-time gas- and particle-phase measurements was deployed at Look Rock, TN, during the 2013 SOAS to examine the effects of anthropogenic emissions on isoprene-derived SOA formation. High- and low-time-resolution PM2.5 samples were collected for analysis of known tracer compounds in isoprene-derived SOA by gas chromatography/electron ionization-mass spectrometry (GC/EI-MS) and ultra performance liquid chromatography/diode array detection-electrospray ionization-high-resolution quadrupole time-of-flight mass spectrometry (UPLC/DAD-ESI-HR-QTOFMS). Source apportionment of the organic aerosol (OA) was determined by positive matrix factorization (PMF) analysis of mass spectrometric data acquired on an Aerodyne Aerosol Chemical Speciation Monitor (ACSM). Campaign average mass concentrations of the sum of quantified isoprene-derived SOA tracers contributed to ~ 9% (up to 28 %) of the total OA mass, with isoprene-epoxydiol (IEPOX) chemistry accounting for 97% of the quantified tracers. PMF analysis resolved a factor with a profile similar to the IEPOX-OA factor resolved in an Atlanta study, and was therefore, designated IEPOX-OA. This factor was strongly correlated (r2 > 0.7) with 2-methyltetrols, C5-alkene triols, IEPOX-derived organosulfates, and dimers of organosulfates, confirming the role of IEPOX chemistry as the source. On average, IEPOX-derived SOA tracer mass was ~ 26% (up to 49 %) of the IEPOX-OA factor mass, which accounted for 32% of the total OA. A low-volatility oxygenated organic aerosol (LV-OOA) and an oxidized factor with a profile similar to 91Fac observed in areas where emissions are biogenic-dominated, also were resolved by PMF analysis, whereas no primary organic aerosol (POA) sources could be resolved. These findings were consistent with low levels of primary pollutants, such as nitric oxide (NO ~ 0.03 ppb), carbon monoxide (CO ~ 116 ppb), and black carbon (BC ~ 0.2 μg m-3). Particle-phase sulfate is fairly correlated (r2 ~ 0.3) with both methacrylic acid epoxide (MAE)/hydroxymethyl-methyl-lactone (HMML)-(henceforth called methacrolein (MACR)-derived SOA tracers) and IEPOX-derived SOA tracers, and more strongly correlated (r2 ~ 0.6) with the IEPOX-OA factor, in sum suggesting an important role of sulfate in isoprene SOA formation. Moderate correlation between the MACR-derived SOA tracer 2-methylglyceric acid with sum of reactive and reservoir nitrogen oxides (NOy; r2 = 0.38) and nitrate (r2 = 0.45) indicates the potential influence of anthropogenic emissions through long-range transport. Despite the lack of a clear association of IEPOX-OA with locally estimated aerosol acidity and liquid water content (LWC), box model calculations of IEPOX uptake using the simple GAMMA model, accounting for the role of acidity and aerosol water, predicted the abundance of the IEPOX-derived SOA tracers 2-methyltetrols and the corresponding sulfates with good accuracy (r2 ~ 0.5 and ~ 0.7, respectively). The modeling and data combined suggest an anthropogenic influence on isoprene-derived SOA formation through acid-catalyzed heterogeneous chemistry of IEPOX in the southeastern United States. However, it appears that this process was not limited by aerosol acidity or LWC at Look Rock during SOAS. Future studies should further explore the extent to which acidity and LWC as well as aerosol viscosity and morphology becomes a limiting factor of IEPOX-derived SOA, and their modulation by anthropogenic emissions.
We have demonstrated the formation of light-absorbing (290 < λ < 700 nm) SOA constituents from reactive uptake of trans-β-IEPOX onto preexisting sulfate aerosols as a potential source of secondary BrC. IEPOX-derived BrC generated in controlled chamber experiments under dry, acidic conditions has an average mass absorption coefficient of ∼ 300 cm2 g−1. Chemical analyses of SOA constituents using UV−visible spectroscopy and high-resolution mass spectrometry indicate the presence of highly unsaturated oligomeric species with molecular weights separated by mass units of 100 (C5H8O2) and 82 (C5H6O) coincident with the observations of enhanced light absorption, suggesting such oligomers as chromophores, and potentially explaining one source of humic-like substances (HULIS) ubiquitously present in atmospheric aerosol. Similar light-absorbing oligomers were identified in fine aerosol collected in the rural southeastern United States during the SOAS campaign, supporting their atmospheric relevance and revealing a previously unrecognized source of oligomers derived from isoprene that contributes to ambient fine aerosol mass.
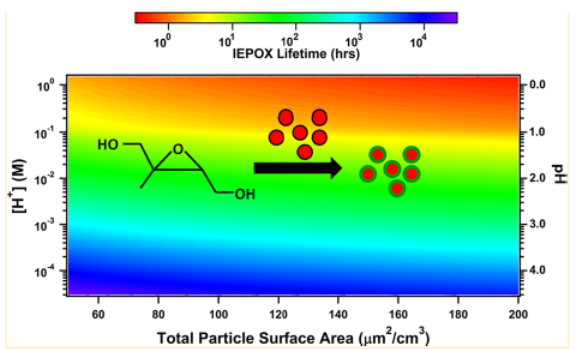
We also report measurements of γIEPOX for trans-β-IEPOX, the predominant IEPOX isomer, on submicron particles as a function of composition, acidity, and relative humidity (RH). Particle acidity had the strongest effect. γIEPOX is more than 500 times greater on ammonium bisulfate (γ ~ 0.05) than on ammonium sulfate (γ ≤ 1 x 10−4). We could accurately predict γIEPOX using an acid-catalyzed, epoxide ring-opening mechanism and a high Henry’ s law coefficient (1.7 x 108 M/atm). Suppression of γIEPOX was observed on particles containing both ammonium bisulfate and poly(ethylene glycol) (PEG-300), likely due to diffusion and solubility limitations within a PEG-300 coating, suggesting that IEPOX uptake could be self-limiting. Using the measured uptake kinetics, the predicted atmospheric lifetime of IEPOX is a few hours in the presence of highly acidic particles (pH < 0) but is greater than 25 h on less acidic particles (pH > 3). This work highlights the importance of aerosol acidity for accurately predicting the fate of IEPOX and anthropogenically influenced biogenic SOA formation. Figure 1 shows the IEPOX lifetime against reactive uptake as a function of aerosol acidity and total particle surface area. The conclusion from this figure is that IEPOX uptake onto acidic particles is competitive with its other major sinks, including reactions with OH radicals in the gas phase and deposition.
Figure 1. Lifetime of IEPOX uptake onto acidic sulfate aerosol as determined from our flow reactor measurements. This matrix should be useful to models like the U.S. EPA CMAQ model.
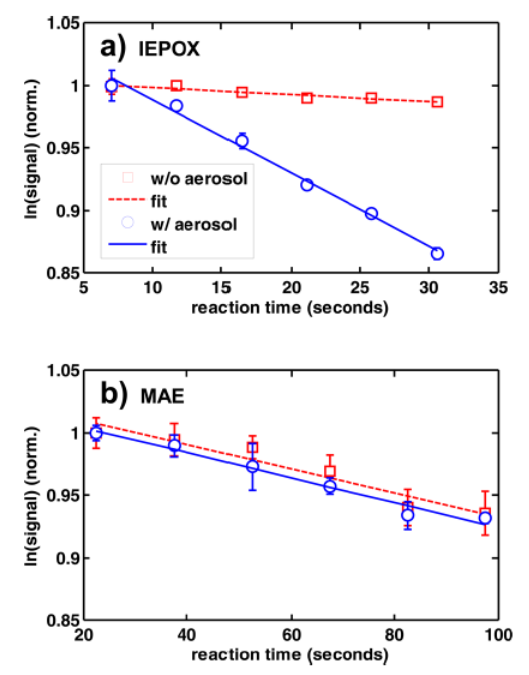
In addition, a combination of flow reactor studies and chamber modeling is used to constrain two uncertain parameters central to the formation of secondary organic aerosol (SOA) from isoprene-derived epoxides: (1) the rate of heterogeneous uptake of epoxide to the particle phase and (2) the molar fraction of epoxide reactively taken up that contributes to SOA, the SOA yield (ϕSOA). Flow reactor measurements of the trans-β-isoprene epoxydiol (trans-β-IEPOX) and methacrylic acid epoxide (MAE) aerosol reaction probability (γ) were performed on atomized aerosols with compositions similar to those used in chamber studies. Figure 2 shows how we determined the γ ranges for trans-β-IEPOX and MAE as 6.5 x 10-4−0.021 and 4.9−5.2 x 10-4, respectively. Through the use of a time-dependent chemical box model initialized with chamber conditions and γ measurements, ϕSOA values for trans-β-IEPOX and MAE on different aerosol compositions were estimated between 0.03−0.21 and 0.07−0.25, respectively, with the MAE ϕSOA showing more uncertainty.
Figure 2. Average of the logarithm of the epoxide signal vs reaction time and associated linear fit without aerosols (red squares,±2σ; red dashed line) and with aerosols present in the flow reactor (blue circles, ±2σ; blue solid line) for (a) trans-β-IEPOX and (b) MAE on a (NH4)2SO4 + H2SO4 aerosol. Initial values have been normalized to 1 for ease of comparison.
Lastly, we have constrained condensed-phase formation kinetics of SOA components from IEPOX uptake chemistry. Heterogeneous reactions of IEPOX on atmospheric aerosols form various aerosol-phase components or “tracers” that contribute to the SOA mass burden. A limited number of the reaction rate constants for these acid-catalyzed aqueous-phase tracer formation reactions have been constrained through bulk laboratory measurements. We have designed a chemical box model with multiple experimental constraints to explicitly simulate gas- and aqueous-phase reactions during chamber experiments of SOA growth from IEPOX uptake onto acidic sulfate aerosol. The model is constrained by measurements of the IEPOX reactive uptake coefficient, IEPOX and aerosol chamber wall losses, chamber-measured aerosol mass and surface area concentrations, aerosol thermodynamic model calculations, and offline filter-based measurements of SOA tracers. By requiring the model output to match the SOA growth and offline filter measurements collected during the chamber experiments, we have derived estimates of the tracer formation reaction rate constants that have not yet been measured or estimated for bulk solutions.
Future Activities:
We currently are working on the data analyses of filters we had collected from the CTR ground site during SOAS. We also are writing up data for publication that summarizes some new findings on ISOPOOH + OH chamber experiments we have done in our indoor smog chamber. These show that ISOPOOH + OH yields SOA through the non-IEPOX route in substantial amounts. Many of these SOA constituents also are found at the SOAS ground sites, including the LRK and CTR sites. We also are looking at how SOA coatings on sulfate aerosol affect the reactive uptake of IEPOX, as this more closely mimics the aerosol conditions observed during 2013 SOAS.
Journal Articles on this Report : 10 Displayed | Download in RIS Format
| Other project views: | All 43 publications | 23 publications in selected types | All 23 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Budisulistiorini SH, Li X, Bairai ST, Renfro J, Liu Y, Liu YJ, McKinney KA, Martin ST, McNeill VF, Pye HOT, Nenes A, Neff ME, Stone EA, Mueller S, Knote C, Shaw SL, Zhang Z, Gold A, Surratt JD. Examining the effects of anthropogenic emissions on isoprene-derived secondary organic aerosol formation during the 2013 Southern Oxidant and Aerosol Study (SOAS) at the Look Rock, Tennessee ground site. Atmospheric Chemistry and Physics 2015;15(15):8871-8888. |
R835404 (2014) R835404 (2015) R835404 (Final) R835401 (2014) R835401 (Final) R835410 (2013) R835410 (2014) R835410 (2015) R835410 (Final) |
Exit Exit Exit |
|
|
Gaston CJ, Riedel TP, Zhang Z, Gold A, Surratt JD, Thornton JA. Reactive uptake of an isoprene-derived epoxydiol to submicron aerosol particles. Environmental Science & Technology 2014;48(19):11178-11186. |
R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit Exit |
|
|
Hu WW, Campuzano-Jost P, Palm BB, Day DA, Ortega AM, Hayes PL, Krechmer JE, Chen Q, Kuwata M, Liu YJ, de Sa SS, McKinney K, Martin ST, Hu M, Budisulistiorini SH, Riva M, Surratt JD, St. Clair JM, Isaacman-Van Wertz G, Yee LD, Goldstein AH, Carbone S, Brito J, Artaxo P, de Gouw JA, Koss A, Whisthaler A, Mikoviny T, Karl T, Kaser L, Jud W, Hansel A, Docherty KS, Alexander ML, Robinson NH, Coe H, Allan JD, Canagaratna MR, Paulot F, Jimenez JL. Characterization of a real-time tracer for isoprene epoxydiols-derived secondary organic aerosol (IEPOX-SOA) from aerosol mass spectrometer measurements. Atmospheric Chemistry and Physics 2015;15(20):11807-11833. |
R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit Exit |
|
|
Kristensen K, Cui T, Zhang H, Gold A, Glasius M, Surratt JD. Dimers in α-pinene secondary organic aerosol: effect of hydroxyl radical, ozone, relative humidity and aerosol acidity. Atmospheric Chemistry & Physics 2014;14(8):4201-4218. |
R835404 (2013) R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit |
|
|
Lin Y-H, Budisulistiorini SH, Chu K, Siejack RA, Zhang H, Riva M, Zhang Z, Gold A, Kautzman KE, Surratt JD. Light-absorbing oligomer formation in secondary organic aerosol from reactive uptake of isoprene epoxydiols. Environmental Science & Technology 2014;48(20):12012-12021. |
R835404 (2013) R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit Exit |
|
|
Rattanavaraha W, Chu K, Budisulistiorini SH, Riva M, Lin Y-H, Edgerton ES, Baumann K, Shaw SL, Guo H, King L, Weber RJ, Neff ME, Stone EA, Offenberg JH, Zhang Z, Gold A, Surratt JD. Assessing the impact of anthropogenic pollution on isoprene-derived secondary organic aerosol formation in PM2.5 collected from the Birmingham, Alabama, ground site during the 2013 Southern Oxidant and Aerosol Study. Atmospheric Chemistry and Physics 2016;16(8):4897-4914. |
R835404 (2014) R835404 (2015) R835404 (Final) R835401 (Final) R835410 (2013) |
Exit Exit Exit |
|
|
Riedel TP, Lin Y-H, Budisulistiorini SH, Gaston CJ, Thornton JA, Zhang Z, Vizuete W, Gold A, Surratt JD. Heterogeneous reactions of isoprene-derived epoxides: reaction probabilities and molar secondary organic aerosol yield estimates. Environmental Science & Technology Letters 2015;2(2):38-42. |
R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit Exit |
|
|
Riedel TP, Lin Y-H, Zhang Z, Chu K, Thornton JA, Vizuete W, Gold A, Surratt JD. Constraining condensed-phase formation kinetics of secondary organic aerosol components from isoprene epoxydiols. Atmospheric Chemistry and Physics 2016;16(3):1245-1254. |
R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit Exit |
|
|
Riva M, Budisulistiorini SH, Zhang Z, Gold A, Surratt JD. Chemical characterization of secondary organic aerosol constituents from isoprene ozonolysis in the presence of acidic aerosol. Atmospheric Environment 2016;130:5-13. |
R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit Exit |
|
|
Zhang H, Zhang Z, Cui T, Lin Y-H, Bhathela NA, Ortega J, Worton DR, Goldstein AH, Guenther A, Jimenez JL, Gold A, Surratt JD. Secondary organic aerosol formation via 2-methyl-3-buten-2-ol photooxidation: evidence of acid-catalyzed reactive uptake of epoxides. Environmental Science & Technology Letters 2014;1(4):242-247. |
R835404 (2013) R835404 (2014) R835404 (2015) R835404 (Final) |
Exit Exit Exit |
Supplemental Keywords:
atmosphere, particulates, biogenic volatile organic compounds, VOCs, terpenes, SOA, SOAS;Relevant Websites:
UNC Gillings School of Public Health: Jason D. Surratt, PhD Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.