Grantee Research Project Results
Final Report: Producing and Characterizing Bacillus Subtilis Biosurfactants with Potentially Lower Environmental Impact for Salt Water Applications
EPA Grant Number: R835181Title: Producing and Characterizing Bacillus Subtilis Biosurfactants with Potentially Lower Environmental Impact for Salt Water Applications
Investigators: Lamsal, Buddhi , Somasundaran, P. , Nyman, John A. , Green, Christopher C. , LeBlanc, Brian D.
Institution: Iowa State University , Columbia University in the City of New York , Louisiana State University - Baton Rouge
EPA Project Officer: Aja, Hayley
Project Period: April 1, 2012 through March 31, 2015 (Extended to March 31, 2016)
Project Amount: $500,000
RFA: Environmental Impact and Mitigation of Oil Spills (2011) RFA Text | Recipients Lists
Research Category: Aquatic Ecosystems , Ecological Indicators/Assessment/Restoration
Objective:
Specific Objective #1: Evaluate fermentation and purification strategies to produce effective and low-toxicity biosurfactants, their isoforms, and elucidate their structure: This objective entails optimizing fermentation conditions by utilizing different energy sources for Bacillus subtilis, such as fibrous feedstocks like switchgrass, bagasse, etc. We hypothesize that different fermentation strategies and operational parameters can affect isoforms of biosurfactant produced resulting in different surface properties.
Specific Objective #2: Evaluate dispersant activity of biosurfactants and their isoforms: This objective entails studying the structural aspects of biosurfactants and their isoforms produced by Bacillus subtilis (surfactin and FA-Glu) to understand the structure-property relationships of various FA-Glu isoforms and developing formulations that potentially could have applications in oil-spill cleanup.
Specific Objective # 3: Evaluate environmental effects of biosurfactants and their isoforms: This objective involves analysis of toxicity of biosurfactants produced by Bacillus subtilis (FA-Glu and surfactin) on small fish (Fundulus grandis) common to estuaries on the northern Gulf of Mexico.
Summary/Accomplishments (Outputs/Outcomes):
Specific Objective #1:
Accomplished: Scale up study in 5-L bioreactor on fibrous biomass based hydrolysates has been completed and techno-economic evaluation is being conducted. Isoform fractionation of three surfactin isoforms has also been completed at a purity of 72%.
1.1 Five liter fermentation of Bacillus subtilis strains of fibrous biomasses
Post completion of 50-mL shake flask fermentation experiments, based on a combined pretreatment (liquid ammonia and ultrasonication) optimization on six fibrous biomass hydrolysates (switchgrass, alfalfa, soy hulls, soy fiber, DDGS and bagasse), concentration of two biosurfactants FA-Glu and surfactin were determined. Switchgrass, alfalfa and soy hulls turned out to be the best performing biomasses and were selected for scale up to 5-L bioreactor fermentation experiments.
Five-liter bioreactor fermentation experiments were performed with Bacillus subtilis strains producing FA-Glu and surfactin, with 2% (w/v) carbon based growth media from biomass hydrolysate, with glucose as control. Pretreatment of 300 g biomass was conducted at optimized conditions for switchgrass, alfalfa and soy hulls (1:11 solid loading of biomass and 15 % w/v liquid ammonia at 60°C for 12 h and subsequent ultrasonication for 3 min at 170 µm amplitude) to produce sufficient hydrolysates for 2% (w/v) carbohydrate concentration. The 5-L bioreactor experiments were conducted at 2.5 mL/mL/min airflow rate and agitation speed of 75 rpm. Fermentation experiments for both Bacillus subtilis strains were completed for all three selected fibrous biomasses with glucose as a control. Post fermentation analysis and bacterial growth analysis has shown that scale up of pretreated hydrolysates as growth components in media for Bacillus subtilis strains were successful in achieving similar yields (Table 1,Table 2) as the shake flask fermentations. The maximum yields achieved for both shake flask level and 5-L bioreactor fermentations were found to be approximately 3 g/L and 0.3 g/L for surfactin and FA-Glu respectively. Among all the fibrous biomasses, soy hulls performed the best in terms of amount of total product made An initial techno-economic evaluation of our experiments has shown that surfactin cost estimate is $45.7/g compared to the market price of $13.94/mg ($13,940/g).
1.2 Fractionation of surfactin isoforms
The isoform fractionation experiments were conducted on 95% pure surfactin produced from three 15-L glucose (2% w/v) fermentation experiments on Bacillus subtilis. Methanol-dissolved surfactin was passed through a preparatory C-18 column (50 x 250 mm) and peaks for each isoform (992 Da, 1006 Da, 1020 Da, 1034 Da, 1048 Da) were collected and 72% purity in the third phase of fractionation on the three most abundant isoforms (992 Da, 1020 Da, 1034 Da) was achieved. These isoforms fractions were collected and detected in a negative ion mode mass spectrometer to confirm the molecular weights and relative purities of isoforms in each fraction.
Specific Objective #2:
Accomplished: Surface tension of surfactin isoforms was measured
Three surfactin isoforms obtained from 1.2 were shipped to LSU and CU for molecular/structural characterizing and toxicity testing. The critical micelle concentration (CMC) values of the fractionated isoforms of surfactin were determined. The surface tension was measured at 25±1 oC using the Wilhelmy vertical plate technique with a sandblasted platinum plate as the sensor [1]. The pull exerted on the sensor was determined with a Cahn microbalance kept in a draft-free enclosure. For each measurement, the sensor was in contact with the solution for 30 min for equilibration. The CMC was obtained from the intersection of the sloped and plateau portions of the surface tension vs. concentration curve. The slope of the curve was used to calculate the adsorption density at the interface with the Gibbs equation [2] (Table 3).
The CMC values of surfactin and fatty acyl glutamic acid (FA-Glu) were measured in deionized double-distilled water (DDDW) and in 12 g/L NaCl solutions prepared using Instant Ocean® salt [3]. The CMC of Surfactin and FA-Glu in DDDW was measured as 0.013 g/L and 0.35 g/L, respectively; under saline conditions the CMC value was measured to be 0.4 g/L. Generally, the CMC values, whether determined under saline conditions or not follows a relationship given as below:
logC0=-k log(C0+Ca)+k2
.................................................(1)
Where, where C0 and Ca are the critical micelle concentration and the concentration of the added salt, respectively; k1 and k2 are constant. Therefore, depending on k1 and k2, minor variations as observed here are likely. Fundamentally, such variations can be attributed to packing of surfactant and self-assembly forces that leads to the formation of the micelles. This can be attributed to packing of surfactant and self-assembly forces that leads to the formation of the micelles.
In May 2015, three isoforms of surfactin were obtained and the CMC values of these isomers were determined by using the procedure given above. Fig. 1 shows that the CMC values of the surfactin isoforms were all around 0.015 g/L. However, in order to dissolve the surfactant fractions into solution, NaOH was added and pH level was brought up from a starting pH of around 6 to as high as 7.5. The isoform 2 in particular required higher concentration of NaOH compared to isoform 1 and 3. (Fig 1)
Specific Objective #3:
Accomplished: Dose response curves for surfactin isoforms on Gulf killifish (Fundulus grandis) were established.
Ninety-six hour bioassays to determine dose response curves for fractionated isomers of surfactin have been completed with 8 to 10 day old Gulf killifish (Fundulus grandis). Non-fractionated samples of surfactin will serve as a benchmark for toxicity of a mixture of Surfactin isomers. Unlike our previously reported work with FA-Glu, the surfactin assays were all conducted at a single salinity of 12%.
Newly fertilized Gulf killifish eggs were incubated at room temperature (25°C) for 14 days. Larvae were used in Surfactin bioassays at 10 days after hatch. A preliminary range finding bioassay was conducted on each of the three Surfactin isomers to center on an inflection point of the dose-response curve. A definitive 96-hr bioassay was conducted using 5 concentrations equally centered on the estimated LC-50 values from the preliminary examination. A control group was maintained at each bioassay requiring > 90% survival and the assays was rerun if this survival in the control group was not maintained. All individuals were fed approximately 5 newly hatched Artemia nauplii at 48 hours of the bioassay. Total ammonia nitrogen, nitrite, salinity, pH, and temperature were recorded daily. Treatment and control waters were changed daily and dead larvae were removed and recorded at this time.
The treatment solutions were created using desiccated surfactin isoforms weighed to the nearest mg to create 1 L stock solutions. In order to dissolve the initial Surfactin into the stock solutions, each weighed powder was initially dissolved in DMSO (1 mL) with a similar amount added to the control stock solution (1 L).
The acute toxicity of FA-Glu is presented in (Table 3) and details previous reports that indicate changes in acute toxicity in response to changes in environmental salinity. The surfactant, Dioctyl Sulfosuccinates (DOSS) is also presented to represent the comparative toxicity of a common industrial surfactant. All testing on the Surfactin isomers are complete and indicate that toxicity appear to be similar (Table 4). At this time, statistical tests of equality and parallelism have not been performed.
TABLES AND FIGURES
| Biomass | Surfactin | Fa-Glu |
|---|---|---|
| Switchgrass (SW) | 1.84 | 0.127 |
| Alfalfa (AA) | 2.38 | 0.092 |
| Baggase (BG) | 1.33 | 0.171 |
| Dry distillers grain solubles (DD) | 1.21 | 0.109 |
| Soy Fiber (Soy Fiber) | 2.49 | 0.228 |
| Soy hulls (SH) | .030 | 0.249 |
| Gkycise (control) | 3.1 | 0.327 |
| Biomass | Surfactin | FA-Glu |
|---|---|---|
| Switchgrass (SW) | 2.11 | 0.217 |
| Alfalfalfa | 1.80 | 0.176 |
| Soy Hulls (SH) | 2.9 | 0.282 |
| Glucose | 2.70 | 0.257 |
| Surfactant | CMC (DDDW) g/L | CMC (saline conditions) g/L |
|---|---|---|
| FA-Glu (received in 2014) | 0.35* | 0.4 |
| Surfactin (received in 2014) | 0.013 | To be determined |
| * The cmc of FA-Glu in DDDW were ~0.35 g/L, which is higher (0.1/g/L than FA-Glu sample received from on of the earlier batches. ϯ The replicates of the cmc values were within an error range of ±3% | ||
| Bioassays (salinity) | LC50 value (mg/L) | 95% lower-upper confidence interval (mg/L) |
|---|---|---|
| FA-Glu at 4% | 126.0 | 106.0 - 138.7 |
| FA-Glu at 12% | 101.1 | 96.2 - 122.9 |
| FA-Glu at 24% | 68.2 | 64.0 - 72.5 |
| DOSS at 12% | 24.8 | 18.6 - 34.9 |
| Isoforms (tested at 12% salinity) | LC50 value (mg/L) | 95% lower - upper confidence interval (mg/L) |
|---|---|---|
| Isoform 1 - M.W. 992 | 7.122 | 4.639 - 10.543 |
| Isoform 2 - M.W. 1020 | **17.1 | ** 11.3 - 35.4 |
| Isoform 3 - M.W. 1034 | 9.941 | 8.7 - 11.3 |
| ** Preliminary Finding - Expecting LC50 value to be between 10 and 16 | ||
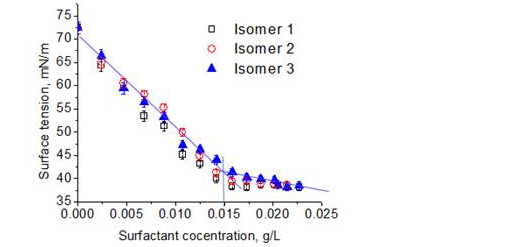
Figure 1. Surfactant concentrations vs surface tension for 3 different
isoforms (also called isomers) of surfactin. Molecular weights of
Isomer 1, 2 and 3 are 992 Da, 1020 Da and 1034 Da
Outreach activity:
1. Columbia University: Miss. Suji Balfe, a high school junior from Madison High School, New Jersey, participated in this research program where she worked on determining the CMC of surfactin and its isoforms.
2. Louisiana State University: Community outreach and education was carried out within this performance period with a number of different materials including oral presentations, videos, press releases, printed materials, visuals aids at demonstration booths, and a web presence. Andy Nyman, Bran Wagner, and Chris Green created a series of information oral presentations answering questions such as: What makes a substance toxic? What does a toxicologist do? How does toxicology apply to everyday life? Presentations also created a background and introduction to toxicology by highlighting the History and introduction to Toxicology. Additional titles of these oral presentations included Acute and chronic toxicology, Introduction to surfactants, and review of the research performed in this grant titles laboratory studies of petroleum based surfactants and microbial-based surfactants.
Informational workshops at libraries in Louisiana and recorded metrics of attendance and participation were conducted. The research team at LSU was invited to present their work at a public meeting organized by the Gulf of Mexico Research Initiative and the Sierra Club. The team also presented and interacted with individuals in the Seafood and Fishery Industries at information Fisheries Expo organized by NOAA and the Louisiana SeaGrant.
Printed materials for distribution at the previously mentioned events to complement our efforts have been created. Brochures that detailing importance of fish physiology and how surfactants in everyday life and their use have been produced along with videos to detail how toxicologist determine acute toxicity and have created a brief video to inform people about the use of aquatic organisms in toxicology testing.
Conclusions:
Future Work:
Completion of FA-Glu fractionation into constituent isoforms would be performed and sent to toxicity and surface tension studies to LSU and Colombia respectively. LSU will conduct assays to equally combine all three isomers for a new bioassay to confirm that the 3 isomers together present similar acute toxicity to the non-fractionated source material. Parallel bioassay on original non-fractionated surfactin to compare LC values from the same cohort of test organisms will be conducted. At Colombia, determination of the cmc values of the isomers of the surfactin isomers under saline conditions will be performed along with the determination of the surfactant packing in the micelles devised with individual surfactants, their isomers and surfactant mixtures. Electron spin resonance (ESR) technique will be used to determine the rotational coefficient of a DS-16 probe, which provides a measure of the surfactant packing. Beside use of ESR technique, beta-parameter will also be derived from the CMC values.
References:
1. Shanahan MER. A simple analysis of local wetting hysteresis on a Wilhelmy plate. Surf Interface Anal 1991;17(7):489-495.
2. Menger FM, Galloway AL, Chlebowski ME. Surface tension of aqueous amphiphiles. Langmuir 2005;21(20):9010-9012.
3. Atkinson MJ, Bingman C. Elemental composition of commercial sea salts. J Aquaricult Aquat Sci 1997;8(2):39-43.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other project views: | All 5 publications | 3 publications in selected types | All 3 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Lamsal B, Patra P, Sharma R, Green C. Production of Non-Toxic Biosurfactan-Surfactin-Through Microbial Fermentation of Biomass Hydrolysates for Industrial and Environmental Applications. TENSIDE SURFACTANTS DETERGENTS 2019;56(5):357-366. |
R835181 (Final) |
Exit |
|
|
Ren K, Lamsal B, Mendonca A. Stability of Emulsions and Nanoemulsions Stabilized with Biosurfactants, and their Antimicrobial Performance against Escherichia coli O157:H7 and Listeria monocytogenes. TENSIDE SURFACTANTS DETERGENTS 2019;56(5):436-446. |
R835181 (Final) |
Exit |
|
|
Sharma R, Lamsal B, Colonna W. Pretreatment of fibrous biomass and growth of biosurfactant-producing Bacillus subtilis on biomass-derived fermentable sugars. BIOPROCESS AND BIOSYSTEMS ENGINEERING 2016;39(1):105-113. |
R835181 (Final) |
Exit Exit |
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
