Grantee Research Project Results
2011 Progress Report: Enabling Potable Reuse Of Wastewater Using Forward Osmosis: A Sustainable And Affordable Alternative To Reverse Osmosis
EPA Grant Number: R834872Title: Enabling Potable Reuse Of Wastewater Using Forward Osmosis: A Sustainable And Affordable Alternative To Reverse Osmosis
Investigators: McCutcheon, Jeffrey R
Institution: University of Connecticut
EPA Project Officer: Packard, Benjamin H
Project Period: June 1, 2011 through May 31, 2016
Project Period Covered by this Report: June 1, 2011 through May 31,2012
Project Amount: $300,000
RFA: Advancing Public Health Protection through Water Infrastructure Sustainability (2009) RFA Text | Recipients Lists
Research Category: Drinking Water , Water
Objective:
The objective is to evaluate forward osmosis for wastewater reuse potential.
This project has been undertaken by the Sustainable Water and Energy Learning Laboratory (SWELL). This is a collaborative research-learning environment where graduate and undergraduate students work together to develop materials that enable sustainable technologies that produce both water and power. During Year 1 of this project, two graduate students have been partially funded by this project and several undergraduates have participated.
This project aims to evaluate the promise of forward osmosis (FO) as a technology to enable direct potable reuse of water. FO is an emerging membrane technology platform that offers a low cost alternative to RO as a means of recapturing wastewater for direct potable reuse. The barriers to direct potable reuse are great, mostly stemming from the guaranteed removal of contaminants that can cause harm to humans even at low levels when exposure is chronic. Some of these contaminants are not removed effectively using membrane processes because of their chemistry. The goal of this project is to develop new membranes that function in FO and test those membranes for removal of various contaminants.
Progress Summary:
After year 1, we have established three new membrane platforms for use in forward osmosis.
Platform 1: Nanofiber Supported TFC Membranes
This novel membrane design is the signature accomplishment of SWELL. This membrane could represent the next generation of membranes for not only FO, but for other membrane processes as well. Nanofiber nonwovens produced by electrospinning are employed as supports for thin film composite membranes. During year one of this project, we have successfully formed a polyamide layer in situ through a conventional interfacial polymerization approach. Images of these membranes can be seen in our recent publication in the Journal of Membrane Science (see section 7 of this report), though we provide some of the most recent images here for this report in Figure 1.

Figure 1 - Diagram showing how polyamide layers are formed onto nanofiber supports for use in forward osmosis
As seen in figure 1, we have developed a method for forming this layer which serves as the selective barrier in the membrane. The nanofibers act only as a support, but unlike those in RO membranes, these supports offer much less resistance to solute transport.
The process by which these layers are deposited are shown in Figure 2.
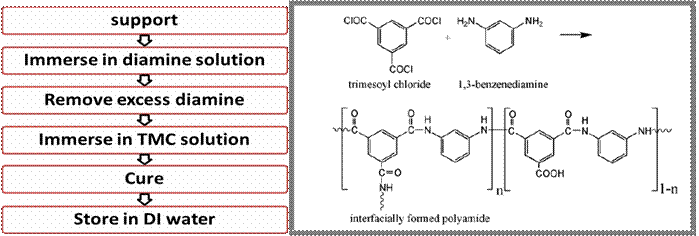
Figure 2 – Interfacial polymerization process (left) and chemistry (right)
In this process, the support material is saturated with an amine solution (typically m-phenylene diamine) and then exposed on one side to an organic phase (such as hexane), which contains a dissolved acid chloride. The acid chloride and the amine react to form a highly crosslinked aromatic polyamide, which serves as the selective layer for the membrane. In our nanofiber membranes, the polyamide will sometimes wrap around the fibers, as shown in Figure 1, or sometimes “float” above the membrane, as shown here.

Figure 3 – SEM image showing delamination of the polyamide selective layer from the nanofiber support
We have developed methods to avoid this delamination and in fact the presence of the polyamide can actually work to increase the strength of the membrane, as shown in Figure 4.
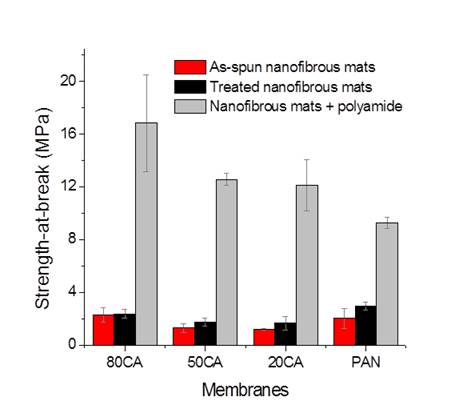
Figure 4 – Tensile strength measurements of nanofibers, treated without formation of PA, and then the TFC membrane.
These membranes have exhibited dramatic increases in strength after deposition of the polyamide layer. The water flux measurements were equally as exciting. We would normally spin the nanofibers onto a support (such as a polyester nonwoven). This facilitates the polyamide formation process and general handling. However, the PET could also be removed for individual tests. Figure 5 shows flux data from both the full membrane support including the PET and the membrane with the PET layer removed. The control test was a commercial FO membrane available from Hydration Technologies Innovations, a company that has pioneered FO technology for the past 10 years.
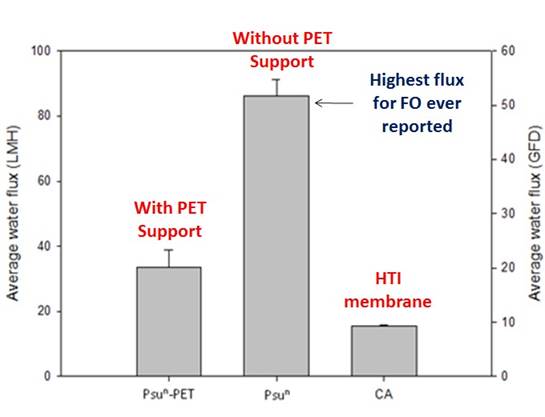
Figure 5 – Flux performance of the nanofiber membrane with a PET support (needed for spinning), after the support was removed, and the HTI membrane as a control. Tests were run at 25 °C with a 1.5 M NaCl draw solution.
These results show that under the same conditions, our membrane exhibited a 2-5 fold increase in flux performance than the HTI membrane. The 90 LMH test was the highest flux ever reported for an FO membrane for any condition.
After year one, we believe that this membrane was well on its way to being an excellent platform for future membrane designs for FO.
Platform 2 – Polydopamine modified RO membranes for forward osmosis
During year 1 of this project, we also demonstrated a new type of membrane modification to make RO membranes function in FO. Poor RO membrane performance in FO has long been attributed to thick, relatively tortuous and low porosity support layers. Another problem with these layers is that they are comprised of relatively hydrophobic polymers, like polysulfone. If the support does not wet out, then it will not be able to transport solutes and therefore perform poorly in FO. In this part of the project, RO membrane support layers were modified with polydopamine (PDA) in order to hydrophilize their structures. These hydrophilic structures would be easier to “wet out” and therefore provide better mass transfer.
The PDA modification process for membranes was pioneered by Benny Freeman at the University of Texas for use as an anti-fouling modification technique. Here, we modify only the support layers using the scheme illustrated in Figure 6.
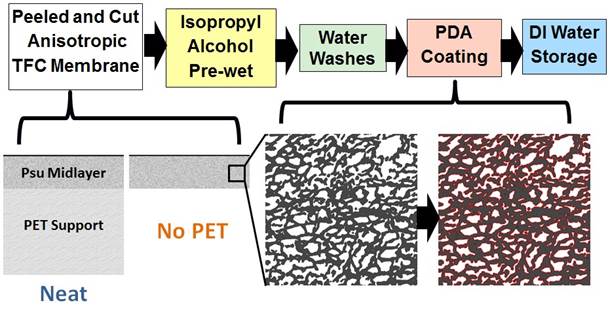
Figure 6 – PDA modification procedure.
The PET is removed in this process because the polysulfone (PSu ) midlayer is very hydrophobic. Work in this area showed that by hydrophilizing the PSu layer, flux could be increased by a factor of up to 12 (Figure 7).
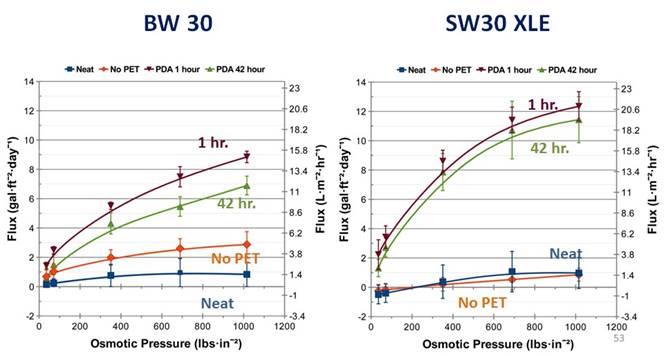
Figure 7 – Flux performance of PDA modified RO membranes (BW 30 and SW30 XLE from Dow Water & Process SolutionsTM). Two coat times are shown (1 hour and 42 hours). Tests were run at 25°C and represent an average of three tests.
These results are remarkable in that without modification, the membrane fluxes were measured to be very low in spite of high osmotic pressure gradients across the membrane. This two-step modification resulted in membrane performance that matches HTI membrane water flux performance.
This is a promising technique because it allows for the use of a manufactured membrane with demonstrated excellent selectivity. If we can make these membranes work with a simple modification, it could provide a mass produced membrane to help propel FO commercialization.
Platform 3 – Nylon 6,6 supported TFC membrane.
An entirely unanticipated membrane was developed under the support of this program. We established a collaboration with 3M Purification Inc. (3M PI, formerly Cuno). This group supported my candidacy for the 3M Nontenured Faculty Award. I received the award in 2010 and it has supported a growing collaboration between 3M and UConn, which resulted in these exciting findings.
3M manufactures a microfiltration membrane comprised of nylon 6,6. These membranes are tiered structures, with highly defined regions making their characterization quite simple. A top and cross section image of one of their membranes is shown in Figure 8.
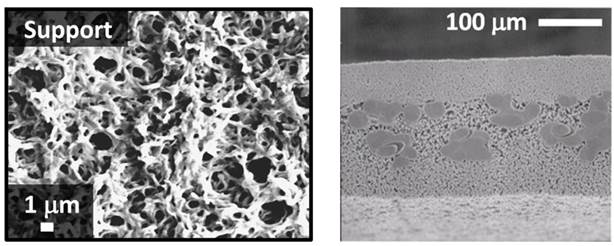
Figure 8 – SEM images of the BLAO1 micrfiltration membrane from the top (left) and the cross section (right).
What we believed that these membranes would serve as are another type of support for a TFC membrane. The material is hydrophilic and the membranes exhibit high porosity.
We proceeded to form polyamide layers onto these membranes using the method described in Figure 2. The images of the results are shown in figure 9.

Figure 9 – Images of the top (left) and cross section (right) of the polyamide layer formed on the 3M nylon 6,6, microfiltration membrane.
Once these membranes were formed, we tested them for their performance using RO so that we could test for salt rejection and water permeability coefficient. The results are shown in Figure 10 and Table 1.
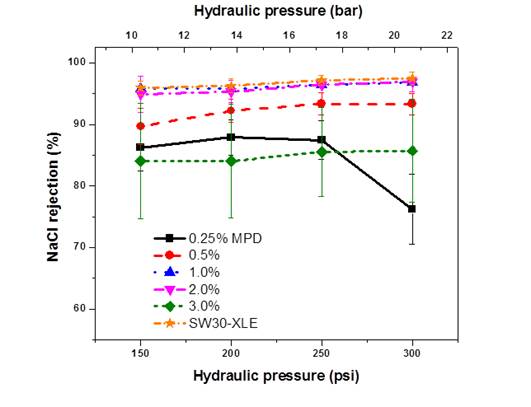
Figure 10 – Reverse osmosis data showing NaCl rejection of TFC membranes formed on the 3M BLAO1 membrane. This membrane has a pore size of 1 µm. Each set of data points represents a different concentration of amine in the aqueous phase during interfacial polymerization
Based on the results shown in figure 10, we can see that there is an optimal condition for forming polyamide layers. The concentration of the amine does make an impact, and higher concentrations, with the exception of 3%, exhibit increasing rejection and pressure tolerance. We note, however, that higher amine concentrations also reduce permeability, as shown in Table 1.
Table 1 – Water permeability coefficient of membranes formed with different amine concentrations in the aqueous phase
| Hydraulic permeability | ||
|---|---|---|
| (gfd/psi) | (LMH/bar) | |
| 0.25 % MPD | 0.080 | 1.96 |
| 0.5% | 0.049 | 1.21 |
| 1.0% | 0.038 | 0.94 |
| 2.0% | 0.031 | 0.76 |
| 3.0% | 0.026 | 0.64 |
| SW30-XLE | 0.044 | 1.08 |
We also ran a control of SW30-XLE so that we could compare the performance of these membranes with a commercial membrane. Remarkably, our membranes showed comparable performance to the SW30-XLE in terms of both salt rejection and water permeance. This is remarkable because this is the first time a polyamide layer has been formed on a support of this kind. We had expected pinholes at worst and poor permselectivity at best given the vastly different chemistry and structure of this support. These results represent the first time that a polyamide layer has been deposited onto a hydrophilic support and reported. Osmotic tests are forthcoming in Year 2 of the grant, as are additional studies on using other membranes like this as a support for TFC membranes.
The outputs of this research have been the following:
Development of new membrane platforms
Three membrane platforms have been developed based on the fundamental approach as described in the proposed work. This development would not have been possible without EPA funding and the EPA now can say it has funded critical research toward the development of FO technology
Education of students
During year 1, one graduate student was trained toward her Ph.D. Two undergraduates were also involved with the project, one of which was working toward an undergraduate thesis.
Industrial collaborators
During year 1, industrial collaborations were established with Oasys Water (a small startup commercializing forward osmosis for produced water treatment) and 3M. These strong relationships could ultimately lead to commercialization avenues for technology that EPA supported in early development
Academic collaborators
UConn established active collaborations with The University of Texas at Austin and The University of California Los Angeles. The PDA modification work was done with Professor Benny Freeman Texas and the interfacial polymerization was done with help from Professor Eric Hoek at UCLA.
Invention disclosure
Three invention disclosures were made to the UCONN Office of Technology and Commercialization. These were joint with UCLA, Texas and 3M.
Future Activities:
The plan for the period starting 5/31/2012 and ending 5/30/2013 will be to continue developing these membrane platforms and to publish their performance results in short order. Each platform is progressing well at the time of this report writing and the next report will exhibit some of those results.
We will also work to establish protocols for conducting the eventual membrane fouling studies that will be necessary when we work with actual wastewater and other contaminants that may be difficult to measure at low concentrations. We have already established protocols for measuring trace inorganic contaminants like arsenic, selenium, cadmium, and lead. We will, however, need to establish protocols for organic contaminants as well.
Future Activities:
- Optimization of membrane fabrication procedure
- Development of alternative materials for membranes designed for forward osmosis
- Elucidating transport mechanisms that could impact trace contaminant removal during wastewater reuse
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 82 publications | 14 publications in selected types | All 12 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Bui N-N, Lind ML, Hoek EMV, McCutcheon JR. Electrospun supported thin film composite membranes for engineered osmosis. Journal of Membrane Science 2011;385-386:10-19. |
R834872 (2011) R834872 (2012) R834872 (2013) R834872 (2014) R834872 (Final) |
Exit Exit Exit |
|
|
Huang L, Bui N-N, Manickam SS, McCutcheon JR. Controlling electrospun nanofiber morphology and mechanical properties using humidity. Journal of Polymer Science Part B: Polymer Physics 2011;49(24):1734-1744. |
R834872 (2011) R834872 (2012) R834872 (2013) R834872 (2014) R834872 (Final) |
Exit Exit |
Supplemental Keywords:
Forward osmosis, engineered osmosis, membrane, thin film composite, nanofiber, electrospinningRelevant Websites:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
