Grantee Research Project Results
2016 Progress Report: Vascular Response to Traffic-Derived Inhalation in Humans
EPA Grant Number: R834796C004Subproject: this is subproject number 004 , established and managed by the Center Director under grant R834796
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: Predictive Toxicology Center for Organotypic Cultures and Assessment of AOPs for Engineered Nanomaterials
Center Director: Faustman, Elaine
Title: Vascular Response to Traffic-Derived Inhalation in Humans
Investigators: Kaufman, Joel D. , Larson, Timothy V.
Institution: University of Washington
EPA Project Officer: Callan, Richard
Project Period: December 1, 2010 through November 30, 2015 (Extended to November 30, 2017)
Project Period Covered by this Report: August 1, 2015 through July 31,2016
RFA: Clean Air Research Centers (2009) RFA Text | Recipients Lists
Research Category: Human Health , Air
Objective:
Project 4 examines the acute vascular effects of commute traffic exhaust exposures in human subjects, in a multi-pollutant context. This double-blind, randomized, controlled crossover trial will test whether traffic-derived mixed pollution atmospheres of diesel exhaust and gasoline engine exhaust, experienced through travel on roadways in a passenger car, causes an increased vascular response (brachial artery vasoconstriction, increased blood pressure, reduced retinal arteriolar diameter) compared with filtered air (FA) in healthy subjects. Nested aims include: whether specific exhaust-related monocytic gene expression effects are mediated by lipid peroxidation; whether traffic-related pollutants' vasoconstrictive effects are increased in subjects with a common SNP variant in the gene coding for TRPV1; and whether monocyte DNA methylation in specific genes is modified with exposure to typical, roadway-derived exposures.
Progress Summary:
Project 4 was launched in Year 4 of the Center. In this project, we use a "typical commute" study design and pertinent experience in human exposure studies to advance the Center's research agenda with a double-blind, controlled exposure crossover clinical trial in 16 subjects, randomized to order. Using an innovative approach in which contrasts of in-vehicle exposure and potential participant susceptibility by genotype are nested in the experiment, we can address several hypotheses in this study. Building on our prior work, we will use a typical commute model to confirm or determine whether traffic (e.g., mixed on-road environment with diesel and gasoline engine exhaust components) derived aerosols exert demonstrable and important acute vascular effects in human subjects, and whether traffic-derived aerosols acutely induce increased lipid peroxidation, response to oxidized phospholipids, and result in measurable impacts on gene expression and DNA methylation, in pathways that are related not only to the triggering of acute cardiovascular events, but also to the development and progression of atherosclerosis. Of course, all of the outcomes we measure are completely transient and reversible, and exposures are designed to be those of a typical urban commute path.
Screening
Subjects were screened to determine eligibility. At screening, subjects were required to be in the normal range for BMI, blood sugar, cholesterol and triglyceride levels, lung function, blood pressure, and ECG. Subjects also fill out questionnaires describing past illness, health history, traffic and chemical exposure, smoking history, and occupation. Buccal swab samples were collected in order to achieve a balance of the TRPV1 (SNP I-585V, rs8065080) gene, which our prior work suggests modifies vascular response to traffic-related air pollution.
Eligible subjects complete three monitoring sessions consisting of three 2-hour commutes that travel I-5, extending from North Seattle to roadways in South Seattle (e.g. Duwamish Valley). During each drive, subjects are accompanied by research staff responsible for collecting subject health measurements and monitoring conditions of the drive. Each drive is separated by at least 3 weeks. Two of the monitoring sessions experience an unfiltered pollution exposure, and one is filtered to remove pollutants; the order of the sessions is randomized and the scenario is conducted in a double-blinded manner. During drives, the cabin air and HEPA filters are configured to reflect the randomized exposure conditions (i.e., on-road ambient or filtered air exposure). The cabin ventilation controls are adjusted such that air is entrained and directed to the floor vents, and the temperature inside the vehicle is comfortable for the occupants. Van windows remain closed during the drive and subjects wear N95 masks while transitioning from the lab to the UW van regardless of drive condition.
Enrollment
To date, all 16 subjects have been enrolled with successful genotype-stratification of recruitment. 14 have completed all three drives, and 2 will complete the study by the end of July 2016.
Health Measurements
Subjects complete health measurements at baseline, during the drive, immediately after the drive, 3 hours later, 5 hours later and 24 hours later. These health measurements include: questionnaires, blood markers, Holter ECG, ambulatory blood pressure, 24 hour urine, brachial artery reactivity, retinal photography, and Finometer measurements. The frequency of health measurements are shown in Table 1. All subjects provided a urine sample for a cotinine test and, if female, a pregnancy test.
| AM/PM | AM | PM | |||||||||||
| Time (hour) | 10:00pm-7:00 am | 7:00 | 8:00 | 9:00 | 10:00 | 11:00 | 12:00 | 1:00 | 2:00 | 3:00 | 4:00 |
| 9:00 am (24hr) |
| Overnight fast | X |
|
|
|
|
|
|
|
|
|
|
| X |
| Urine collection |
| X | X | X | X | X | X | X | X | X | X | X | X
|
| Vitals BP, PR, RR |
| X |
| XX | XXX |
| X |
| X |
| X |
| X |
| ABP |
|
|
|
|
|
| X | X | X | X | X | X | X |
| Holter Monitor 11 min record |
| XX | X | X | XX | X | XX | X | X | X | XX | X | XX |
| Blood Draw
|
|
| X |
|
|
|
|
|
|
| X |
| X |
| BAR Roosevelt |
|
| X |
|
| X |
|
|
|
|
|
|
|
| Symptom Questions |
|
| X |
| X |
| X |
| X |
| X |
| X |
| Commute Drive |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Finometer
|
|
| X | X | X | X | X |
| X |
| X |
| X |
| Retinal Photography |
|
| X |
|
| X |
|
|
|
|
|
|
|
| Lunch
|
|
|
|
|
|
| X |
|
|
|
|
|
|
Air Monitoring
This study involves in-vehicle monitoring for 48 drives involving 16 participants in Seattle. Each day of monitoring includes the following suite of monitors in order to collect real-time measurements of the pollutants: PM2.5 (Nephelometer, Radiance Research), black carbon (microAethelometer, Aeth Labs), particle count (P-Trak, TSI Inc), PAHs (PAS 2000CE, EcoChem), NO2 (CAPS, Aerodyne Research Inc), NOX (UV absorbance Model 410, 2B Technologies), ozone (chemiluminescence 3.02P, Optec), CO (CO T15n, Langan), CO2 (CO2 K-30-FS Sensor, CO2 Meter.com), temp/RH (Precon HS-2000, Kele Precision Mfg), location (GPS BU-353, US GlobalSat). Filters and air monitors inside the car are powered by gel cell batteries connected to power inverters.
Drive Route
A consistent route is used for each subject exposure drive.
Preliminary Results
Preliminary results for 8 participants (24 drives) are presented in Table 2. As demonstrated here, filtered drives showed an order of magnitude reduction in particles and back carbon compared with unfiltered air drives. As anticipated, reductions in NO2 and NOX were more modest.
| Unfiltered Air Drives | Filtered Air Drives | |||
| Mean | 10th - 90th | Mean | 10th - 90th | |
| Travel speed (km/hr) | 43.4 | 0.4 - 87 | 39.5 | 0.4 - 85 |
| P-Trak count, (pt/cm3) | 29,579 | 7,612 - 58,360 | 2,418 | 372 - 5,242 |
| Neph b-scat (m^-1) | 2.5E-05 | 1.2E-05 - 3.8E-05 | 1.8E-06 | 3.1E-07 - 4.5E-06 |
| BC (ng/m3) | 4,017 | 1,113 - 7,405 | 302 | 20 - 693 |
| particle-bound PAH (ng/m3) | 163 | 38 - 331 | 148 | 32 - 335 |
| CO2 (ppm) | 1,434 | 1,031 - 1,897 | 1,853 | 1,074 - 3,825 |
| CO (ppm) | 1.4 | 0.2 - 2.6 | 1.1 | 0.5 - 1.9 |
| NO2 (ppb) | 25 | 12 - 37 | 16 | 5 - 30 |
| NOX (ppb) | 135 | 41 - 245 | 87 | 17 - 182 |
| Figures 1 and 2 display the particle number and black carbon results for a single participant in the study. |
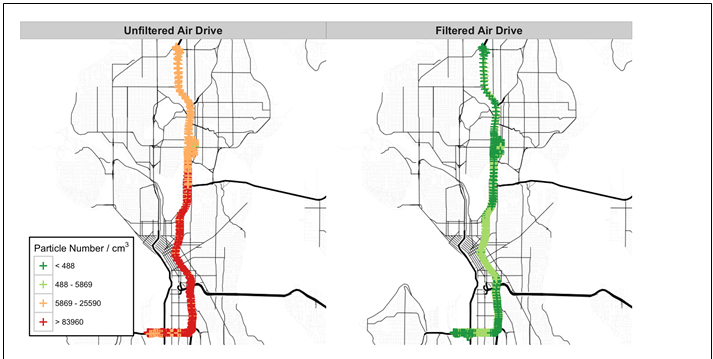 Figure 1. Comparison in measured particle number for unfiltered and filtered drives for a single participant. |
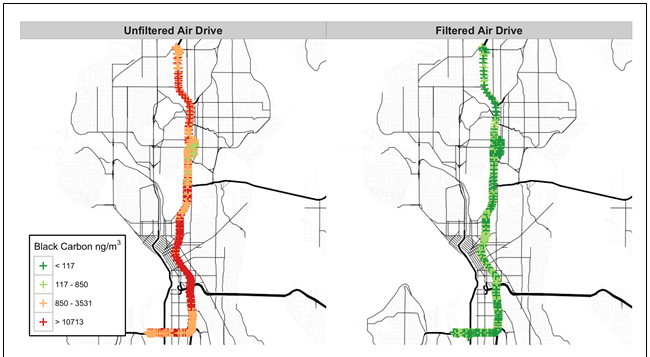 Figure 2. Comparison in black carbon for unfiltered and filtered drives for a single participant. |
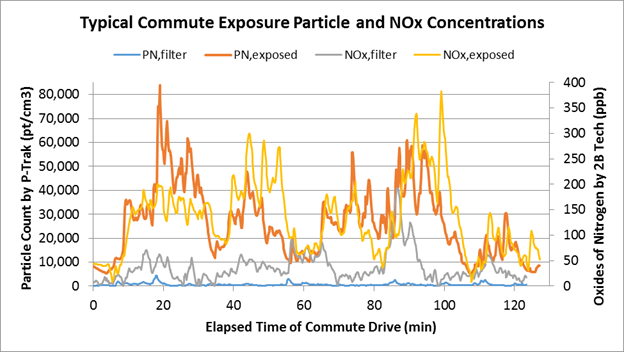
This same participant's measured particle count and total oxides of nitrogen (NOX) concentrations are shown as a time series plot for both roadway exposure and filtered air conditions in Figure 3. This variation in levels throughout the exposure period is typical of measured pollutant concentrations which are affected by vehicle operating conditions and the proximity and volume of traffic.
Figure 3. Selected time series variation of particle count (by P-Trak) and NOX (by 2B Tech analyzer) during an approximate 2-hour exposure drive for both exposure and filtered air.
Future Activities:
We plan to complete the commute exposure study this summer and will move forward with health analyses starting this fall.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other subproject views: | All 2 publications | 2 publications in selected types | All 2 journal articles |
|---|---|---|---|
| Other center views: | All 197 publications | 94 publications in selected types | All 93 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Cosselman KE, Navas-Acien A, Kaufman JD. Environmental factors in cardiovascular disease. Nature Reviews Cardiology 2015;12(11):627-642. |
R834796 (2015) R834796 (Final) R834796C004 (2015) R834796C004 (2016) R834796C004 (Final) |
Exit Exit |
Supplemental Keywords:
Health, Air, Scientific Discipline, ENVIRONMENTAL MANAGEMENT, Health Risk Assessment, Risk Assessments, Air Quality, mobile sources, Biochemistry, air toxics, Risk Assessment, aerosol particles, vehicle emissions, bioavailability, atmospheric chemistry, motor vehicle emissions, particulate matter, motor vehicle exhaust, ambient particle health effects, cardiotoxicity, air quality models, air pollutants, atmospheric aerosols, atmospheric particulate matter, vascular dysfunction, ambient air quality, air pollution, cardiovascular disease, exposure assessment, airway diseaseRelevant Websites:
University of Washington Center for Clear Air Research Exit
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R834796 Predictive Toxicology Center for Organotypic Cultures and Assessment of AOPs for Engineered Nanomaterials Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R834796C001 Exposure Mapping – Characterization of Gases and Particles for ExposureAssessment in Health Effects and Laboratory Studies
R834796C002 Simulated Roadway Exposure Atmospheres for Laboratory Animal and Human Studies
R834796C003 Cardiovascular Consequences of Immune Modification by Traffic-Related Emissions
R834796C004 Vascular Response to Traffic-Derived Inhalation in Humans
R834796C005 Effects of Long-Term Exposure to Traffic-Derived Particles and Gases on Subclinical Measures of Cardiovascular Disease in a Multi-Ethnic Cohort
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2015 Progress Report
- 2014
- 2013 Progress Report
- 2012 Progress Report
- 2011 Progress Report
- Original Abstract
2 journal articles for this subproject
Main Center: R834796
197 publications for this center
93 journal articles for this center