Grantee Research Project Results
2013 Progress Report: Risk Assessment for Manufactured Nanoparticles Used in Consumer Products (RAMNUC)
EPA Grant Number: R834693Title: Risk Assessment for Manufactured Nanoparticles Used in Consumer Products (RAMNUC)
Investigators: Zhang, Junfeng , Georgopoulos, Panos G. , Di Giulio, Richard T. , Lioy, Paul J. , Isukapalli, Sastry S. , Kipen, Howard , Chung, Kian Fan , Garfunkel, Eric , Lee, Ki-Bum , Mainelis, Gediminas , Porter, Alexandra , Ryan, Mary P. , Schwander, Stephan K. , Tetley, Teresa D
Current Investigators: Zhang, Junfeng , Tetley, Teresa D , Chung, Kian Fan , Georgopoulos, Panos G. , Lioy, Paul J. , Schwander, Stephan K. , Ryan, Mary P. , Isukapalli, Sastry S. , Di Giulio, Richard T. , Porter, Alexandra , Garfunkel, Eric , Mainelis, Gediminas , Kipen, Howard , Lee, Ki-Bum
Institution: University of Medicine and Dentistry of New Jersey , Duke University , Imperial College
EPA Project Officer: Aja, Hayley
Project Period: April 1, 2011 through June 30, 2014 (Extended to June 30, 2016)
Project Period Covered by this Report: July 1, 2012 through June 30,2013
Project Amount: $1,999,995
RFA: Environmental Behavior, Bioavailability and Effects of Manufactured Nanomaterials - Joint US – UK Research Program (2009) RFA Text | Recipients Lists
Research Category: Chemical Safety for Sustainability
Objective:
Our U.S.-U.K. center, "Risk Assessment for Manufactured Nanoparticles Used in Consumer Products (RAMNUC)," provides a systematic, multidisciplinary approach for predicting potential human and environmental risks associated with the use of selected consumer products that incorporate zinc oxide and silver as well as a diesel fuel additive containing cerium dioxide nanoparticles. The overall hypothesis of the RAMNUC project is that “manufactured nanoparticles (MNPs) at the point of exposure for both humans and aquatic animals will substantially differ in both physicochemical and toxicological properties from the MNPs at the source (synthesized in the laboratory or acquired commercially).”
Our project includes both experimental and computational tools to analyze the selected MNPs in consumer products and diesel fuel additive using both in vitro and in vivo experiments to assess intra- and extra-cellular bioavailability, bioreactivity and toxicity. We have been characterizing MNPs for their physical (e.g., size, shape, electric charge and state of agglomeration/aggregation) and chemical properties (e.g., composition and surface functionalization). Thus, our in vitro and in vivo studies are producing mechanism-based results relating toxic effects to the physicochemical properties of the MNPs. Another aspect of our project includes an exposure simulation study that will produce realistic estimates of MNP exposures to consumers.
Data generated from these experiments are being integrated into mechanism-based computational modules of our two existing source-to-exposure-to-dose-to-effects modeling systems, allowing for rational extrapolation and generalization in MNP risk assessment. Built upon an inter-institutional structure, the RAMNUC consortium serves as a model for systematically addressing complex problems associated with MNP risk assessment. The results will contribute to the limited knowledge about health risks associated with the use of nanotechnology-based consumer products.
Progress Summary:
This center project has eight specific aims. Below is a summary of the progress made in Project Year 2 by each aim.
Aim 1 (Risk Assessment Framework):
This aim relates to the development of a generalized conceptual risk assessment framework for risks from MNPs and adaptation of these modules using data from Aims 2 through 7. In years 1 and 2, this framework has been developed by adapting existing frameworks and incorporating MNP life cycle analysis approaches for the following stages of MNPs’ life cycle: (a) sources, (b) environmental fate and transport, (c) concentrations in environmental media, (d) human exposure, (e) dose at target organs and bioavailability. We are expanding upon this framework by defining a computational structure for studying toxicodynamic responses following exposures to MNPs. This is being actively developed in collaboration with the RAMNUC team and utilizes other data and mechanistic information from ongoing related research efforts.
Computational Implementation and Application of the risk assessment framework for MNPs:
- Modules for estimating realistic environmental and occupational exposures to MNPs are being developed: (a) modules for environmental levels of silver nanoparticles and carbon nanotubes have been developed by evaluating and applying this framework in complementary research efforts, and (b) modules for environmental levels of cerium dioxide (CeO2) and zinc oxide (ZnO) are being developed as part of Aim 1.
- Modules for consumer exposures and exposures in occupational settings have been developed by adapting existing consumer exposure models. In year 2, initial data from exposure simulation studies (Aim 3) have been incorporated into these modules to extrapolate risk assessment.
- Modules for estimating consumer exposure to nanospray products have been developed and linked with the environmental modules from Year 1. Application case studies so far in Year 2 focused on silver nanoparticles.
- Modules for size-dependent alveolar, tracheobronchial and pharyngeal depositions have been developed and integrated with the consumer exposure modules (Year 2) by linking the modules with existing deposition model, Multiple-Path Particle Dosimetry Model (MPPD v2.11).
- Screening level assessment of cerium dioxide (CeO2) and zinc oxide (ZnO) were performed via PRoTEGE Tier 1 (Prioritization/Ranking of Toxic Exposures with GIS Extension).
- Modules for screening toxicokinetics and toxicodynamics of MNPs are being developed within the generalized framework. These are being evaluated with available in vitro and in vivo measurements.
Aim 2 (Nanoparticle Synthesis and Characterization):
This aim focused on the preparation of well-defined Ag and ZnO MNPs for cell exposure and toxicity assays. Appropriate protocols have been developed and characterization of the particles by size (and dispersion), phase and chemical purity has been achieved.
Particle Synthesis and Characterization
Throughout Year 2 we continued to synthesize and characterize controlled Ag and ZnO nanoparticles (NPs) and nanowires (NWs). Citrate stabilized silver NPs with sizes of ~10 nm diameter and ~50 nm diameter were synthesized using chemical bath reduction The as-deposited ZnO NWs had a uniform diameter of 150 nm and are 2.5 μm and >5 µm long. Silver NWs with a length of 3.4±1.7 µm were produced by chemical bath deposition. Longer AgNWs have also been produced 1D structures were synthesized by preventing growth in the {100} direction by capping the (100) planes of the wires with PVP. Longer AgNWs (>5 µm) have been produced by template-assisted electrodeposition using alumina membranes. The Ag, ZnO and CeO2 NPs have then been distributed to our Center’s in vitro labs in London and New Jersey.
Dissolution of Nanoparticles in situ, in vitro and in vivo
Another sub-aim that we have been addressing during Year 2 is to assess whether, and by which mechanism, the physiological properties (i.e. aggregation state, morphology and chemistry) of the Ag nanostructures (both wires and particles) change in various cell culture media, proteins, components of the lung lining fluid, and human and rodent cells (lung epithelial, THP-1 and airway smooth muscle cells). Using a combination of in-situ synchrotron based Xray scattering (SAXS) and spectroscopic methods, coupled with high resolution imaging, we have demonstrated that the rate of Ag+ ion release from 20nm diameter citrate capped silver NPs is strongly pH-dependent. As predicted, the Ag NPs dissolve more rapidly in acidic pH, whereas they were comparatively stable at extracellular pH over periods of many days. However, the amount Ag+ dissolution is strongly dependent on the cell culture media. In particular, there is a strong interaction with S-containing groups in the media, although we observe that the Ag NWs did not readily sulphidize in media containing the sulphur-rich protein cysteine (in contrast to literature data). We found that DPPC, a phospholipid that is the primary component of lung lining fluid, delayed the release of Ag+ ions, but did not significantly alter the total amount of Ag+ released from the AgNPs after two weeks. In addition, DPPC was found to improve the dispersion of the AgNPs, inhibiting aggregation and coarsening. Transmission electron microscopy (TEM) images revealed that the AgNPs become wrapped with a DPPC layer serving as a semi-permeable protective layer. Hence, we have shown that lung lining fluid, and particularly DPPC can modify the particle aggregation state and the kinetics of Ag+ ion release of AgNPs inhaled into the lung. The biostability and distribution of AgNWs and AgNPs were characterized as a function of time, following incubation of the Ag nanowires with type 1 epithelial cells. TEM imaging and analysis demonstrated that the AgNWs penetrated through the plasma membrane of type 1 epithelial cells and distributed within the cytoplasm and endosomes. The AgNWs were dissolved inside the cells and subsequently transformed into the highly insoluble Ag2 S. We suggest that transformation of the nanowires to sulfide acts as an Ag+ ion ‘trap’ that will significantly limit the short-term nano-Ag toxicity and their impact on environment. Finally, we have successfully imaged the biodistribution of the Ag nanoparticles inside lung tissue following intratracheal installation and have shown that a similar process of Ag2 S formation occurs inside the lung tissue.
Characterization of Diesel Exhaust Particles
In Years 1 and 2, our major focus was centered on the characterization of diesel exhaust particles (DEPs) with and without the CeO2 additive using a commercial product called Envirox (EnviroxTM, Energenics Ltd., UK). DEPs were generated from an internal combustion engine under Aim 4 of our project and we have investigated whether the addition of Envriox, at varying doping concentrations, into diesel fuel altered the physicochemical properties (size, shape, agglomeration, chemical composition and surface charge) of DEPs.
It was found that the addition of Envirox significantly affected the various physicochemical properties of the resulting DEPs. The DEPs obtained after the addition of Envirox were significantly smaller in size as evidenced by the measurement of airborne particles and dynamic light scattering analysis (Figure 1), which could be due to the more effective combustion of the diesel fuel in presence of CeO2. The morphology of the DEPs was further analyzed using scanning electron microscopy (SEM) of particles collected on filters. In addition, it was noticed that the DEPs had a highly negative surface charge as evidenced by zeta potential measurements. This can be attributed to the presence of oxides as a result of combustion. However, there was no significant change in the surface of the DEPs with an increase in the Envirox concentration. We also performed the elemental analysis of the DEP obtained from the combustion of the diesel fuel (with and without Envirox) and found that the percentage of carbon was reduced from 90% to 79% on increasing the Envirox concentration to 10 times the manufacturer recommended value. In addition, the percentage of nitrogen increased correspondingly, which was further supported by an increase in the gaseous NOx amount with increase in Envirox concentration (as elaborated in Aim 4).
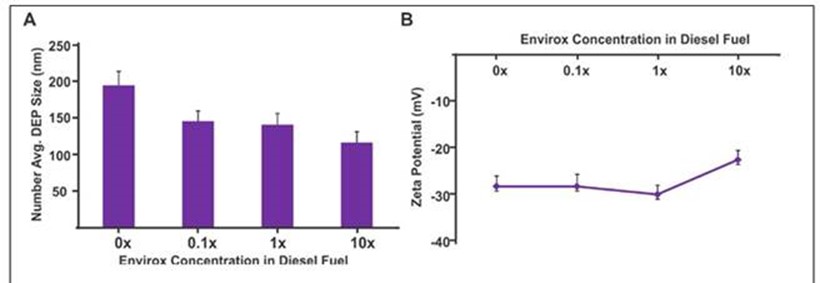
Figure 1. Hydrodynamic size and surface charge of the DEPs with and without addition of
Envirox. (A) DLS analysis clearly shows the reduction in size of the diesel exhaust particles
with an increase in the Envirox concentration in the fuel. (B) The Zeta potential analysis
of the DEPs shows that there was no significant change in the negative surface chanrge of
the DEPs with increse in the amount of Envirox . Results are presented as means ± SE.
0, 0.1x, 1x and 10x means diesel containing Envirox at 0, 0.0, 1 and 10 times the manufacturer
recommended doping concentration, respectively.
Aim 3 (Exposure Simulation Studies):
This aim was designed to investigate exposure to nanoparticles from commercially obtained consumer sprays that incorporate ZnO and Ag MNPs. Analysis of 4 sprays was suggested in our grant proposal; however, at the commencement of the project, those products were no longer available. We identified a new set of 19 nanotechnology consumer products to be analyzed, 13 containing silver and 6 with zinc. The aerosols produced by those sprays and potential exposures were determined using three different aerosolization methods. C-Flow® and Collison® nebulizers were used to aerosolize the sprays and analyze the range of particle sizes that could be produced. A test system was also built to analyze aerosol production using sprayers included with each product. Scanning Mobility Particle Sizer (SMPS) and Aerodynamic Particle Sizer (APS) were used to analyze particle size distribution and concentration of the aerosols generated from the products.
Production of nano-sized and micro-sized particles by the two nebulizing methods and by the product sprayer was found during experimentation with all products. The concentration in the nano-sized region (1-100 nm) ranged from 102 to 106 #/cm3 for Ag and 102 to 105 #/cm3 for Zn. Collison® nebulizer produced a higher number of nano particles per cubic centimeter than C-Flow® and included sprayers. Use of sprayers supplied with the products tended to produce higher concentration of particles above 1 µm. These larger particles are likely nanoparticle agglomerates or aggregates with particles from product matrix.
In order to analyze the agglomeration status and fraction of nanoparticles in the produced aerosol, we constructed an electrostatics-based collector to capture aerosolized particles on TEM grids. The device allows capturing of wide particle size distributions as well as particles of only a certain size when used in conjunction with SMPS. Initial images from the TEM analysis showed individual nano-sized particles and micro-sized agglomerates.
The following experiments are currently being initiated for characterization:
- More detailed analysis of nanoparticles and their agglomerates captured on TEM grid.
- Simulated use of sprays in a room; determination of resulting personal exposures.
- Selection and characterization of sprays for in vitro and in vivo experiments.
Aim 4 (Diesel Exhaust Particle Collection):
The purpose of this aim was to collect and characterize diesel exhaust particles from an internal combustion engine combusting diesel fuels with and without Envirox to allow comparisons of resultant exhaust particle properties. In order to investigate the effect of CeO2 additives on the physicochemical properties of the resulting DEPs, we collected diesel exhaust particles from burning a normal ultralow sulfur diesel fuel (purchased in New Jersey) with Envirox at 0, 0.1, 1, and 10 times the manufacturer recommended doping concentration, corresponding to 0, 0.9, 9.0, and 90 CeO2 μg/ml, respectively. We measured the diesel exhaust for (1) particle size distributions by particle number and mass, (2) total particle number concentration, (3) CO concentrations, (4) CO2, (5) NO/NO2/NOx, (6) black carbon, (7) gas-phase PAHs, (8) particle phase PAHs, (9) particle-phase elements, and (10) gas-phase aldehydes.
Results show that the addition of Envirox™ resulted in emission rate reductions for CO2, CO, total particulate mass, formaldehyde, acetaldehyde, acrolein, and several polycyclic aromatic hydrocarbons, mostly in a CeO2-concentration-dependent manner. However, the use of fuel additive at manufacturer-recommended concentration resulted in increases in emission rate of NOx (+9.3%) and that of total particle number (+32%). Furthermore, increasing fuel CeO2 concentrations led to decreases in the size of particles emitted. Given health concerns related to ultrafine or nanoparticles and NOx, our findings may complicate the risk-benefit analysis of Envirox™ and possibly other ceria-based fuel additives. These findings are reported in a manuscript that has been submitted to a peer-reviewed journal. Currently we are preparing another manuscript reporting findings on physicochemical characteristics (elemental composition, morphology, oxidizing potential, surface charge, hydrodynamic size) of diesel exhaust particles in relation to CeO2 concentrations in the fuel.
In Year 2, we continued to collect diesel exhaust particles for experiments conducted in Aims 2-6.
Aim 5 (in vitro Studies):
The in vitro studies have been conducted in parallel in two laboratories (the Tetley Lab in London and the Schwander Lab in New Jersey).
Tetley Lab
In Year 2, further in vitro cytotoxicity tests of MNPs were carried out in the unique human pulmonary alveolar epithelial type 1-like cell line (TT-1) and primary human lung fibroblasts (HLF).
Concentrations of ZnO nanoparticles (15, 30 and 250nm) higher than 10µg/ml induced superoxide release in TT-1 cells at 4 hours. Lower concentrations of ZnO did not induce superoxide release. Pre-treating TT-1 cells with antioxidants glutathione (5mM) or N- acetylcysteine (1mM) reversed ZnO nanoparticle toxicity at concentrations of 10-50µg/ml at both 4 and 24 hours. ZnO at this range of concentrations stimulated IL-6 and IL-8 release in TT-1 cells pre-treated with antioxidants at levels comparable with non-pre-treated cells exposed to sub-toxic (>10µg/ml) concentrations of ZnO. ZnO activated the oxidative-stress mitogen activated protein kinase pathways, namely P38, ERK and JNK at concentrations of 10µg/ml and 20µg/ml at 4 hours. Inhibition of P38 MAPK reversed ZnO toxicity at 4 hours without affecting either IL-6 or IL-8 release. The effect of surfactant on ZnO cytotoxicity was assessed by incubated particles with Curosurf prior to exposures. Preliminary studies show that surfactant may have a protective effect against ZnO cytotoxicity. These studies are continuing.
In vitro test using diesel exhaust particles (DEP) from the four fuels (0x, 0.1x, 1x, and 10x Envirox) were carried out in primary human lung fibroblast (HLF) cells. The highest dose of DEP, 100µg/ml, caused no reduction in cell viability 48 hours and approximately 20% cell death at 72 hours. This was independent of Envirox concentrations. At 72 hours DEP induced significant proliferation of HLF cells at concentrations of 25-100µg/ml. Envirox concentrations 1x and 10x stimulated the highest levels of proliferation (~70%). 100µg/ml of DEP inhibited collagen production in HLF cells at 72 hours, an effect that was Envirox-concentration dependent.
Schwander Lab
In Year 2, studies focused on assessing bioreactivity of DEPs (from the four fuels) in THP-1 cells and primary human peripheral blood mononuclear cells (PBMC). We have evaluated batch- to-batch variability using three sets of DEP samples collected and extracted at different time points (namely Collections 1, 2, and 3). Three doses at 0, 10 and 50 μg/mL were used in our experiments.
Exposure of both THP-1 cell line and primary PBMC to identical concentrations of DEP within each fuel type from collections #1, 2 and 3 resulted in different bioreactivity as determined by IL-1β, TNF-α, IL-6, IFN-γ, and IL-12p40 mRNA expression (qRT-PCR) and protein production (ELISPOT assays). Marked differences in bioreactivity from the three collections were observed despite following the same protocols for DEP collection, extraction, and resuspension in water. Examination of the physicochemical properties from collections #1, 2 and 3 identified differences in particle size (nm) and particle charge (Zeta potential, mV), which turned out to be the primary determinants of the differences observed in bioreactivity in our cell culture system. The observed batch-to-batch variability of our study material may be due to variations in environmental (e.g., weather condition affecting combustion) and experimental (e.g., length of storage of DEP filters before extraction) conditions among the three collections over a long time period or other yet unknown factors.
Our findings point to the potential risks of over- or underestimation of bioreactivity effects (and by inference of public health risks) resulting from the use of pools from different particle collections without taking into account potential batch-to-batch variability of DEPs collected under different combustion conditions and prepared under different experimental conditions.
As part of our commitment to assessing MNP effects on antimicrobial immune effector functions, we also infected THP-1 cells and PBMC with avirulent M.tb strain (H37Ra) and examined whether DEPs from different CeO2-concentration fuels would have different impact on M.tb-induced gene and protein expressions. M.tb infections were done at a multiplicity of infection (MOI) 10, i.e. exposing monocytes within PBMC to 10 M.tb bacteria / cell. This work will be completed in Year 3.
Aim 6 (in vivo Studies):
Year 1 was primarily used to obtain in vitro toxicity data first to guide the exact nanoparticle types to be used in the animal in vivo studies. In Year 2, we initiated studies to examine the effects of Ag nanoparticles and DEPs in vivo in the lungs.
We studied the effect of intratracheally-administered silver nanospheres (AgNPs; 20nm and 110nm PVP- or citrate-capped) in Brown-Norway (BN) and Sprague-Dawley (SD) rats. We found that the 20nm citrate-capped AgNPs were more proinflammatory than the 110nm PVP- capped and that there was a different response of the lung toxicity and inflammatory changes in the 2 rat strains. A preexisting eosinophilic lung inflammation in BN rats was associated with a late eosinophilic response with bronchoconstriction and bronchial hyperresponsiveness. This indicated that AgNPs may worsen pre-existing eosinophilic inflammation in the lungs leading to greater lung toxicity, and that the smaller sized citrate-capped AgNPs were the most toxic. We are currently studying the toxicity of intratracheally-administered silver nanowires (AgNWs) in SD rats compared to AgNPs of 20nm size.
We are also now comparing the effects of intratracheally-administered AgNPs with those induced by a PALASTM spark generator in Dr Smith’s lab that delivers an aerosol concentration of 1.1 mg/m3 inhaled via the nasal route by SD rats at two exposure durations of 1.3 hours and 3 hours, producing estimated doses of 10µg and 20µg per rat respectively (equivalent to 0.04 and 0.07 mg/kg rat). Lung inflammation with a small but significant increase in neutrophils over controls for both dose levels was seen. Further studies are currently progressing with chronic exposure.
Studies of DEP± CeO2 administered intratracheally in mice
These have just started in our lab using the DEP generated at Rutgers University (from Aim 4). The primary question is whether CeO2 in diesel exhaust particles produces particles that have enhanced or decreased toxicity in the lungs.
Aim 7 (Ecotoxicity Studies):
This aim has not started according to the defined time line of our project. Experiments will begin in Year 3 as described below under the “Future Work” section.
Aim 8 (Refinement of Computational Modules):
This aim was initiated in Year 2 and involves the refinement of the risk assessment framework by parameterization of computational modules developed as part of Aim 1 with data collected from Aims 2-6. We are currently modifying the already developed modules addressing the specific experimental scenarios from Aims 2 through 6.
- We are focusing on parameterized particle size distribution for the consumer exposure modules according to data from Aim 3 (exposure simulation studies).
Future Activities:
Project implementation is on schedule as planned in our proposal and we do not foresee any major changes in project activities for the next reporting period, July 1, 2013 to June 30, 2014. Our planned activities are described below by specific aims.
Aim 1
The activity for this aim has been completed in Year 2.
Aim 2
For year 3, we will continue to characterize our consortium’s nanoparticles for various physicochemical properties, including the following techniques TEM, SEM, Raman, XANES, XRD, Zeta-potential, UV-Vis and others. The information gathered from our experiments will be used to help probe the investigation of in vitro and in vivo toxicity of such nanomaterial-based commercial products. Tests for the next year include the continuation of (1) cell exposures to compare ZnO, Ag and CeO2 nanoparticles with similar diameter to test the effect of redox activity on the cellular reactivity of the particles and their effects on cell viability, (2) transmission electron microscopy to determine uptake of NPs by cells (following i), and (3) determination of the dissolution behavior of the various particles in lung cells (ICP, UV-vis, DLS, XAS).
Aim 3
Future tasks will be the collection of the released MNPs for their subsequent analysis, such as using TEM and similar techniques. In separate tests, the airborne particles from selected sprays will be collected for ensuing in vitro investigation of the particle toxicity (Aim 5). In year 3, the following specific experiments that have been recently initiated will continue: use of sprays using their original packaging (sprayers) in a room to simulate and determine potential personal exposures.
Aim 4
The primary work under this aim has been completed and will now focus on completion of the second manuscript and assisting Aims 2-6 in determining the reasoning behind the toxicological effects being examined through our ongoing in vitro and in vivo experiments.
Aim 5
In Year 3, we will continue to test the effects of CeO2 and diesel exhaust particles provided from Aim 4. We will also continue to test synthesized ZnO nanoparticles and nanowires provided by Aim 2. We will assess oxidative stress and cell signaling pathways on primary human alveolar epithelial cells and alveolar macrophages.
In other experiments, we will continue assessing the effects of particles on M.tb-induced gene and protein expression eventually broadening the breadth of our gene expression studies to TLR pathway related genes and a greater number of inflammation related cyto-, and chemokines supernatants of cell cultures. We will also continue to assess the toxicity of the MNPs in THP-1 and primary blood cells using several toxicity assays and examine whether MNPs affect growth control of M.tb in peripheral blood mononuclear cells (PBMCs).
Aim 6
In Year 3, we will continue the in vivo experiments of intratracheally administered AgNWs, spark generated nanoparticle studies and DEP± CeO2 administered intratracheally in mice. We have in mind these studies to be completed by the end of Year 3; however, it is very much possible that they may extend through to the early part of Year 4.
We will also start experiments with the consumer products containing silver and zinc oxide nanoparticles. Proposed in vivo studies of consumer products have been discussed, including a face-to-face meeting to discuss the choice of consumer products and delivery system with Dr. Gedi Mainelis who has characterized the consumer products containing silver and zinc oxide nanoparticles (See Aim 3). The proposed exposure route is inhalation from a nebulized aerosol (produced using a Collison nebulizer) in a nose-only exposure system. Preliminary studies to produce nebulized aerosols for the exposure system have been undertaken at the PHE lab.
All of these experiments will include measurement of lung inflammation and oxidative stress.
Aim 7
Our aquatic toxicity experiments will begin in Year 3 and will be led by R. Di Guilio at Duke University. The goal of this aim is to determine the effects of the selected MNPs as released into the environment on aquatic organisms by evaluating MNPs collected in media under simulated conditions of MNP disposal in wastewater. The experiments under this aim will allow the completion of the “life-cycle” analyses for the three MNPs being studied in the RAMNUC project.
Aim 8
This aim involves refinement of the risk assessment framework by parameterization of computational modules developed as part of Aim 1 with data collected from Aims 2-7 and will continue to be actively pursued in Years 3-4. We are currently customizing the modules developed as part of Aim 1 so that they can address specific experimental scenarios from Aims 2 through 6. Specific efforts include customization of these modules to: (a) estimate cerium oxide levels corresponding to controlled experimental settings in Aims 3 and 4, and (b) describe the in vitro toxicodynamic responses corresponding to experimental settings of Aim 5 and in vivo toxicodynamic responses corresponding to experimental settings of Aim 6.
- Continue to develop modules for estimating population exposure from cerium dioxide (CeO2) in diesel fuel and diesel exhaust particles. The modules will be parameterized based on data collected from Aims 4 through 6.
Journal Articles on this Report : 6 Displayed | Download in RIS Format
| Other project views: | All 47 publications | 28 publications in selected types | All 28 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Nazarenko Y, Zhen H, Han T, Lioy PJ, Mainelis G. Potential for inhalation exposure to engineered nanoparticles from nanotechnology-based cosmetic powders. Environmental Health Perspectives 2012;120(6):885-892. |
R834693 (2011) R834693 (2012) R834693 (2013) R834693 (2014) R834693 (2015) R834693 (Final) |
|
|
|
Nazarenko Y, Lioy PJ, Mainelis G. Quantitative assessment of inhalation exposure and deposited dose of aerosol from nanotechnology-based consumer sprays. Environmental Science: Nano 2014;1(2):161-171. |
R834693 (2013) R834693 (2014) R834693 (2015) R834693 (Final) |
Exit Exit |
|
|
Royce SG, Mukherjee D, Cai T, Xu SS, Alexander JA, Mi Z, Calderon L, Mainelis G, Lee K, Lioy PJ, Tetley TD, Chung KF, Zhang J, Georgopoulos PG. Modeling population exposures to silver nanoparticles present in consumer products. Journal of Nanoparticle Research 2014;16(11):2724. |
R834693 (2013) R834693 (2014) R834693 (2015) R834693 (Final) |
Exit Exit |
|
|
Seiffert J, Hussain F, Wiegman C, Li F, Bey L, Baker W, Porter A, Ryan MP, Chang Y, Gow A, Zhang J, Zhu J, Tetley TD, Chung KF. Pulmonary toxicity of instilled silver nanoparticles: influence of size, coating and rat strain. PLOS ONE 2015;10(3):e0119726 (17 pp.). |
R834693 (2013) R834693 (2014) R834693 (2015) R834693 (Final) |
Exit Exit Exit |
|
|
Subramaniam P, Lee SJ, Shah S, Patel S, Starovoytov V, Lee K-B. Generation of a library of non-toxic quantum dots for cellular imaging and siRNA delivery. Advanced Materials 2012;24(29):4014-4019. |
R834693 (2011) R834693 (2012) R834693 (2013) R834693 (2014) R834693 (2015) R834693 (Final) |
Exit Exit |
|
|
Zhang J, Nazarenko Y, Zhang L, Calderon L, Lee KB, Garfunkel E, Schwander S, Tetley TD, Chung KF, Porter AE, Ryan M, Kipen H, Lioy PJ, Mainelis G. Impacts of a nanosized ceria additive on diesel engine emissions of particulate and gaseous pollutants. Environmental Science & Technology 2013;47(22):13077-13085. |
R834693 (2013) R834693 (2014) R834693 (2015) R834693 (Final) |
Exit Exit |
Supplemental Keywords:
Consumer products, manufactured nanoparticles, diesel exhaust particles, inhalation exposureProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- Final Report
- 2015 Progress Report
- 2014 Progress Report
- 2012 Progress Report
- 2011 Progress Report
- Original Abstract
28 journal articles for this project
