Grantee Research Project Results
Final Report: Analysis and Fate of Single-Walled Carbon Nanotubes and Their Manufacturing Byproducts in Estuarine Sediments and Benthic Organisms
EPA Grant Number: R833859Title: Analysis and Fate of Single-Walled Carbon Nanotubes and Their Manufacturing Byproducts in Estuarine Sediments and Benthic Organisms
Investigators: Ferguson, P. Lee , Chandler, G. Thomas
Institution: University of South Carolina at Columbia
EPA Project Officer: Hahn, Intaek
Project Period: July 1, 2008 through June 30, 2011
Project Amount: $399,628
RFA: Exploratory Research: Nanotechnology Research Grants Investigating Fate, Transport, Transformation, and Exposure of Engineered Nanomaterials: A Joint Research Solicitation - EPA, NSF, & DOE (2007) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
Research supported by this grant extends previous EPA-supported work in our laboratories on estuarine fate and effects of SWNT. Specifically, we have utilized our stock of well-characterized SWNT and 14C-SWNT to fully develop and validate a new trace-analytical method aimed at specific detection of SWNT in complex mixtures. This method takes advantage of the unique and intrinsic NIR fluorescence behavior of pristine SWNT materials. The 14C-SWNT radiotracer was utilized to track recoveries and analytical yield during the sample preparation steps and to standardize near infrared fluorescence (NIRF)-based quantitative analyses. We have then tested our newly developed NIRF methods using SWNT standard-addition experiments with relevant sediments collected from areas likely to receive SWNT-input through manufacture or use of these materials. We have also conducted parallel experiments using both radiotracer and unlabeled SWNT aimed at tracking the disposition of these carbon nanomaterials (and potential transformation products) among sediment, porewater, and meiobenthic organisms in sediment-microcosms collected from a pristine salt marsh estuary. Specific objectives of the project were to:
- Develop and validate sensitive and selective analytical methods based on near-infrared fluorescence (NIRF) spectroscopy for assaying the fate and transport of single-walled carbon nanotubes in estuarine sediment, water, and fauna.
- Utilize the developed NIRF methods in parallel with radiotracer experiments (14C-SWNT) to determine the partitioning, biological uptake, and transformation of SWNT and their nanocarbon manufacturing byproducts in estuarine sediment microcosms under laboratory conditions.
Summary/Accomplishments (Outputs/Outcomes):
I. Development and initial application of Near Infrared Fluorescence Spectroscopy as a sensitive analytical tool for quantifying SWNT in the aquatic environment
The first task of this project centered on development and validation of a NIRF spectroscopy method for detecting, characterizing, and quantifying SWNT in estuarine sediments and biota (addressing Objective #1, above). The goal was to achieve high sensitivity (part-per-billion level) while maintaining analytical tolerance for complex background (e.g., sediment extracts).
Several analytical methods have been applied to characterize SWNT under well-defined laboratory settings, including TEM and Raman spectroscopy for qualitative analysis, as well as UV-Vis spectrometry, size exclusion chromatography, thermogravimetry-mass spectroscopy and quartz crystal microbalance (QCM-D) for quantitative analysis.[1-4] These methods are limited by high detection limits e.g., UV-Vis spectroscopy detection in the mg L-1 range as well as matrix effects in natural samples; they are typically useful only for analysis of SWNT in pure form or in very simple solutions.[4-12]
SWNT can be classified as either semiconducting or metallic, depending on their electronic structure as described by the chiral wrapping-angle index (n,m).28 The distribution of allowed SWNT chiralities is approximately 2:1 semiconducting:metallic, that implies approximately 2/3 of SWNT species are semiconducting, and all current synthetic methods lead to more or less complex mixtures of both types.[13] Synthesis of isochiral SWNT materials is currently not possible, and extensive purification is required to prepare mixtures enriched in semiconductive or metallic species.[14-16] Individualized, semiconducting SWNT in aqueous surfactant suspension give distinct fluorescence emission in the near infrared (NIR) range when excited by visible/NIR light (600-800 nm); these unique spectral features can be used to determine structural information. [17, 18] Fluorescence has been observed directly across the band gap of semiconducting carbon nanotubes.[13] The energy of this transition is highly dependent on nanotube diameter.[17] Consequently, at different wavelengths, various distinct Exx transitions will occur in a sample containing different (n,m) SWNT, allowing a characterization of the bulk SWNT sample.[13]
Although the fluorescence of SWNT in aqueous suspensions is unique, it can be quenched by aggregation or bundling, chemical functionalization, or acidification. For reliable detection and quantification of SWNT by near-infrared fluorescence spectroscopy (NIRF), nanotubes need to be individualized in suspension. This can be achieved through high power sonication in the presence of surfactants, DNA or polymers.[17, 19-22] Importantly, the fluorescence quantum yield of SWNT can vary considerably depending on the suspension matrix.[13] A centrifugation step can be performed after sonication to remove larger agglomerates as well as impurities. Assuming that impurities are less well dispersed or have a significantly higher sedimentation rate in the surfactant solution than the SWNT, a decrease of the non-SWNT carbon fraction in a sonicated suspension is achieved through centrifugation.[23] Moore, et al. showed that in an aqueous SDS solution, metal catalyst impurities affiliate more with larger SWNT bundles than individualized or smaller SWNT bundles.[19] Consequently, bundles as well as metal catalyst residues can be effectively removed by centrifugation after sonication.
To date, NIR fluorescence microscopy has been applied to gain qualitative information of the (n,m) composition of mixed SWNT bulk samples, as well as to detect and image SWNT in biological samples such as macrophage cells and Drosophila melanogaster.[23, 24] Most biological and environmental samples show extremely weak or essentially no background photoluminescence in the NIR region. Consequently, NIR emission spectra from SWNT in biological samples can give very high signal-to-noise, enhancing sensitivity even in complex samples. In this project, we illustrate the utilization of the unique electronic properties of semiconductive SWNT to develop a method for extraction, characterization and quantification of these important manufactured nanomaterials from natural sediments using estuarine sediments as model sediments. To our knowledge, this is the first report of quantification of SWNT in sediments by extraction and NIRF measurement. As only semiconducting SWNT fluoresce, proximal metallic SWNT can act as a photon sink and might broaden the absorbance spectra; therefore, it was crucial to develop methods for efficiently extracting and disaggregating SWNT mixtures prior to NIRF analysis.[17] SWNT used in this study show a low degree of functional groups, low metal content and high quality of semiconducting SWNT, all of which serve to minimize those interferences.
Four different types of CoMoCat SWNT (SouthWest NanoTechnologies Inc., SWeNT, OK, USA) were tested in this study: SG65 SWNT (lot-no. 000-0031), SG76 SWNT (lot-no. 000-0020), CG200 SWNT (lot-no. 400) and CG100 SWNT (lot-no. 000-0012). The SWNT were produced by chemical vapor deposition. [25] All materials had a carbon content > 90% by weight and relative purity of >90%.[25] All SWNT were used as received without any further purification. As indicated by the manufacturer, SG65 SWNT contains > 90% semiconducting tubes. SG65 SWNT were characterized by NIRF and optical absorption spectroscopy (diameter and chirality determination), SEM, Raman spectroscopy (SWNT quality) and XPS (surface functionalization); results of SWNT characterization are provided in Figure S1, Supporting Information. The 14C-labeled SWNT were received from Research Triangle Institute (RTI), NC. The as-produced 14C-labeled SWNT material from RTI was purified by acid treatment and electrophoresis as described previously.[26] Compared to the CoMoCat SWNT, the 14C-SWNT were oxidized and showed a much higher degree of dispersibility; characteristics of this 14C-SWNT material have been previously reported. [5]
All chemicals were analytical grade purchased from Acros (Deoxycholic acid sodium salt SDC, 99%; benzalkonium chloride BAC, and Sodium dodecyl benzene sulfonate SDBS, Sodium dodecyl sulfate SDS, Cetyltrimethyl ammonium bromide CTAB), Sigma Aldrich (Pluronic-F127, Gum Arabic GA), Anatrace (Sodium cholate SC, sol-grade) and ICN Biomedicals, Inc. (TritonX-100). Natural estuarine sediment was collected from a pristine salt marsh estuary – Bread and Butter Creek (BBC) within the North Inlet Estuarine Research Reserve on the coast of South Carolina, USA. This sediment was well characterized (98% silt/clay, total organic carbon content fOC = 2.6%, total black carbon content fBC= 0.6%[5]) and has been previously used for estuarine ecotoxicity bioassays and microcosm studies.[5] Subtidal sediment was collected from central Long Island Sound, NY (sand:silt:clay ratio 8:77:14, fOC = 2.0%, fBC = 0.26 ± 0.02%).40 [27, 28]
SWNT SG65 suspensions were prepared in 2% w/v surfactant in deionized water (c0 = 1 mg SWNT mL-1 surfactant solution) by ultrasonication for 10 min at 50 Watt power input (microtip, Sonifier 450, Branson Ultrasonics, Danbury, CT, USA) in a salt-water ice bath and centrifugation for 10 or 30 min at 17860 x g. The supernatant was diluted for spectroscopic characterization. The pH of all samples was determined to be neutral. NIR absorbance and fluorescence spectra of SWNT suspensions were measured using a Nanospectralyzer NS1 (Applied Nanofluorescence, Houston, TX) with three discrete laser excitation wavelengths (wavelength/power: 638 nm/32 mW, 691 nm/31 mW, 782 nm/74 mW). The integration time was 100 ms with 100 spectral averages. Fluorescence emission spectra were scanned and recorded from 900 – 1300 nm after stepwise excitation by the three lasers. These three excitation spectra can be used to create 3D excitation/emission contour plots using a spectral modeling approach based on the known electronic transitions of individual semiconductive SWNT chiral species (discussed below).28
In an aqueous suspension of dispersed SWNT, fluorescence intensity is influenced both by SWNT (n,m) chirality and by their respective concentrations. Individual SWNT (n,m) species give different fluorescence signal strengths even when present at equal mass concentrations. In order to interpret the data and determine the relative concentrations of each SWNT species, we have applied a set of correction factors that reflect the different intrinsic quantum yield of each species. These calibration factors were determined previously by photophysical measurements on individual SWNT in SDC and other surfactants[29] and were used in concert with the raw fluorescence spectra to calculate diameter and (n,m) distributions of SWNT species in samples. Quantitative calibration curves were developed based on total emission power and total quanta (peak area of each individual species). The measured NIR fluorescence intensity (emission power per wavenumber) was dependent on the optical excitation power, the SWNT concentration, sample matrix and the sample quality. We tested this parameter for quantitative purposes. Standards as well as samples were treated by using the same preparation protocol (prepared with the same surfactant and batch of SWNT).
The concentration of SWNT stock solution was determined by UV-Vis spectroscopy (SpectraMax 5 Multimode Plate reader, Molecular Devices Inc.) comparing the absorbance at 775 nm before and after centrifugation.[30] No loss to settling of surfactant-wrapped SWNT was observed over a period of 3 months. Moreover, the stability of SWNT stock suspensions was studied by NIRF spectroscopy with no observed change in NIRF signal for more than 3 months, indicating that SWNT dispersed in 2% w/v SDC were stable in suspension. For quantitative calibrations, serial dilutions of SWNT stock suspension in 2% w/v SDC or BBC control sediment extract were prepared from 25 ng mL-1 to 30 µg mL-1 in triplicates. The -calibration curve for each of the three excitation lasers was constructed by plotting the total emission power as a function of SWNT concentration. Calibration curves for each (n,m) species present in the sample were derived from fitted spectra. The concentration of (n,m) species was then calculated based on relative abundance in the sample, thus permitting a quick determination of SWNT structure distribution in an extracted sample.
Various surfactants (Triton X-100, GA, SDS, SDBS, CTAB, SC and SDC) in combination with high power sonication were tested for their ability to extract SWNT from sediment. Spiking step: Natural sediment (1.5 g wet sediment: BBC [0.75 g dry weight] or LIS [0.6 g dry weight]) was suspended in 8 mL of 30 practical salinity unit [psu] synthetic seawater (Instant Ocean®) simulating conditions in an estuarine sediment. The sediment slurry was stirred vigorously during SWNT suspension addition: 13 µg (20,000 DPM) for 14C-labeled SWNT or 0.1 - 50 µg for non-radiolabeled SWNT. The slurry was shaken for various times (24 h up to 30 days) on an orbital shaker or on a jar roller at 4°C and centrifuged 15 min at 1880 x g to remove the water phase. Extraction step: The extractant (surfactants dissolved in water, V = 3 mL, 1 – 10% w/v) was added to the sediment. This slurry was sonicated at high power (variable power input, 26 – 50 W) for 10 minutes followed by a centrifugation step (17,860 x g for 10 min). The supernatant was diluted for analysis by scintillation counting for 14C-SWNT (cocktail: Ecoscint XR, National diagnostic, scintillation Counter: Tri-Carb 2800TR, Perkin Elmer) or NIRF spectroscopy for non-labeled SWNT. Subsequent extractions were performed on the sediment residue. Concentration step (applied when necessary): Four sequential sediment extracts were combined (2.5 mL each, total 10 mL) and transferred to an ultracentrifuge tube containing a 1 mL underlayer of 60% Iodixanol (OptiPrep Density Gradient Medium, Sigma Aldrich). The samples were ultracentrifuged at 207,000 x g for 12 h. Concentrated SWNT solution was collected from the interface between the surfactant solution and the Iodixanol density cushion (fraction size: 1 mL, representing a 10-fold concentration). Unspiked sediment samples were treated using the same procedure. Benthic organisms were extracted in 2 mL 2% w/v SDC by ultrasonication (40 Watts power input), centrifuged at 17860 x g for 10 minutes at 22°C and the supernatant measured and quantified by NIRF spectroscopy.
Analytical performance of the method was validated by analysis of precision among triplicate measurements, accuracy in measuring the spiked concentration (validated against recovery of 14C-labeled SWNT), linear range, matrix effects (consistency of response between SWNT standards and standard-addition calibrations in sediments), and instrument/method detection limits. Data analyses, including analysis of variance (ANOVA), were conducted with an a = 0.05.
Disaggregation and individualization of SWNT in suspension was required prior to NIRF analysis. Several synthetic and natural surfactants were evaluated for their ability to disaggregate and individualize SG65 SWNT in aqueous suspension, including SDBS, GA, Pluronic-F127, SC and SDC. For this work, SG65 SWNT suspensions (c0 = 1 mg mL-1) were prepared by high power sonication and ultracentrifugation. The supernatant was removed and used for spectroscopy analysis (UV-Vis and NIRF).
For the tested surfactants (2% w/v) at different SWNT concentration, SDC-dispersed SWNT exhibited the most highly resolved NIRF spectral features (absorbance and emission) as well as the highest fluorescent yield relative to SDBS and SC (Figure S1e,f; Supporting Information). Additionally, 2% w/v SDC solutions proved to be the most effective dispersant for SWNT, as suspensions of SWNT in SDC were more resistant to ultracentrifugation. These results indicated that SDC can disperse SWNT more efficiently than other widely used surfactants. These observations were in agreement with UV-Vis-NIR absorbance measurements of surfactant wrapped SWNT by [19]. The authors showed that UV-Vis-NIR absorbance spectra of SWNT in SDC showed the most resolved spectra compared to other widely used anionic, cationic and neutral surfactant implying a higher yield of individualized SWNT in suspension.[19]
Prior to analysis of SWNT in sediments by NIRF spectroscopy, an effective method for extracting and stabilizing SWNT in aqueous suspension was developed. Various surfactants (BAC, TritionX-100, GA, SDS, SDBS, CTAB, SC and SDC) in combination with high power sonication were tested for their ability to extract SWNT from a sediment matrix (BBC sediment). Experiments were performed with both oxidized, radiolabeled (14C-SWNT) and pristine (SG65) SWNT to evaluate analytical performance and to validate sample preparation methods. It was proposed that fluorescence intensity is influenced by sample preparation method.[31-33] By employing 14C-labeled material the extraction method can be evaluated independent of the optical properties of SWNT material. Since SWNT were transferred to an aqueous solution, sediment matrix effects on scintillation measurements were minimized. In contrast to the optical NIRF measurements, scintillation measurements were not influenced by the agglomeration state of SWNT.
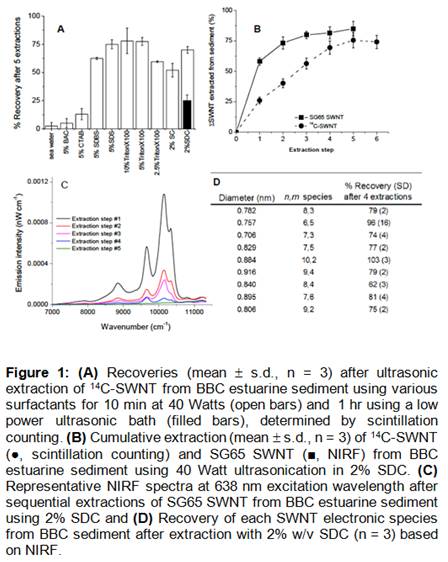
The extraction efficiency for 14C-labeled SWNT depended strongly on the surfactant, surfactant concentration and sonication procedure (Figure 1a). SWNT could not be extracted from the sediment by seawater, indicating a high affinity of SWNT to the sediment matrix.[34, 35] For surfactant concentration of 5% w/v, the ability of each surfactant to extract SWNT from sediment from the least efficient to the most efficient was as follows: cationic (BAC and CTAB) << anion surfactants (SDBS and SDS) < neutral surfactant (Triton X-100). Moreover, results showed that comparable extraction efficiencies (> 65% with 5 sequential extractions) were achieved by using SC and SDC at lower concentration (2% w/v). Additionally, the results showed that sonication conditions had a strong influence on 14C-SWNT recovery. SWNT-recovery increased from 35 ± 10% to 75 ± 4% by using high power (probe) compared to low power (sonic bath) sonication (Figure 1a). High power sonication was necessary to separate SWNT from sediment as well as individualize SWNT in suspension for NIRF-analysis. The extraction efficiency for the different surfactants corresponded to their ability to stabilize SWNT in aqueous suspension. Aromatic surfactants such as SDBS and Triton X-100 stabilized SWNT better than conventional linear surfactants SDS (contain an alkyl chain). For Triton X-100, the stabilizing mechanism is postulated as the formation of hemi-micelles covering the SWNT surface with the benzene rings providing p-p stacking between the surfactant molecule and nanotube core.[19] Bile salts such as SC and SDC showed a high potential for individualizing and stabilizing SWNT in aqueous suspensions.[33] It has been proposed that these surfactants can very effectively accommodate the curvature of the SWNT surface due to the slightly bent but rigid steroid ring. [33]
Based on these results (recovery above 75% for 14C-labeled SWNT), SDC and TritonX-100 were selected to further test their ability to extract SWNT from sediments prior to NIRF spectroscopic analysis. For NIRF measurements, spiked sediment extracts were measured relative to a sediment extract from non-spiked sediment. Cumulative recoveries for SG65 SWNT from BBC sediment obtained by 4 sequential extraction steps with 2% w/v SDC (81 ± 5%) were slightly higher compared to recoveries of 14C-SWNT (75 ± 4%) using 2% w/v SDC (Figure 1b) and significantly higher compared to recoveries of SG65 (57 ± 10%) utilizing 10% w/v Triton X-100 (ANOVA, a = 0.05,). Results demonstrated that 4 sequential extractions were sufficient to extract SG65 SWNT from the sediment (Figure 1b, c). After 5 extraction steps, the solution did not yield recognizable characteristic spectral features of SWNT (Figure 1c). Based on the response for individual (n,m) SWNT species in SG65, it was possible to determine the recovery for (n,m) SWNT species (relative abundance > 1%). The recovery based on SWNT diameter is shown in Figure 1d, illustrating that recoveries of > 75% were yielded for all of the (n,m) SWNT species (except (8,4), which was present in the SWNT mixture at only low abundance as shown in Figure S2). There was no evidence that extraction efficiency was systematically influenced by the diameter or electronic structure of SWNT. Although recoveries varied among the individual SWNT species tested, there was no clear trend according to either diameter or chirality. Repeatability among the three replicate extractions was good (< 5%) for all SWNT species except for the (6,5), which was the dominant species in the mixture. The cause of the higher variability in recovery of this species from sediment was not known.
NIRF spectra of SWNT extracted from sediment using 10% w/v Triton X-100 showed low intensity, peak broadening and limited peak resolution. For Triton X-100, the recovery was much lower than expected based on the results with 14C-SWNT (Figure 1a). The lower apparent recovery of SG65 in Triton X-100 relative to SDC might be explained by the lower efficiency of this surfactant for disaggregating SWNT in suspension since agglomeration state plays a key role in NIRF response. Triton X-100 was a suitable surfactant for extraction of SWNT from sediment; however, it was not effective in disaggregating SWNT for NIRF analysis. Summarizing, our results revealed that SDC proved to be an excellent extractant for both the oxidized 14C-SWNT as well as the pristine SG65 SWNT indicated its general utility for extracting nanotubes with varying surface chemistry from sediment.
Solutions containing only SDC (2% w/v) did not show any absorbance or emission in the NIR region. After sonication and a short centrifugation step, control sediment extracts displayed a yellowish to brownish color depending on the sediment type and mass extracted. Optical absorbance spectra of these extracts revealed a significant optical density of these solutions in the region of maximum SWNT NIRF emission. NIRF spectra of BBC control sediment extracts with no added SWNT showed indistinct features, with a generally rising baseline in each laser excitation channel in the region between 9500 – 11500 cm-1 (emission power value: from 0.000005 to 0.00003 nW cm-1). The emission peaks of several (n,m) species, including the dominant species in SWNT: (6,5); (7,3) and (7,5), fall within this region.[29] For quantification, NIRF spectra in sediment extracts were referenced against control sediment extracts containing no added SWNT. We attempted to matrix-match blank reference samples and samples of interest by quantitatively comparing optical densities in the range from 9500 to 11000 cm-1 to minimize matrix effects.
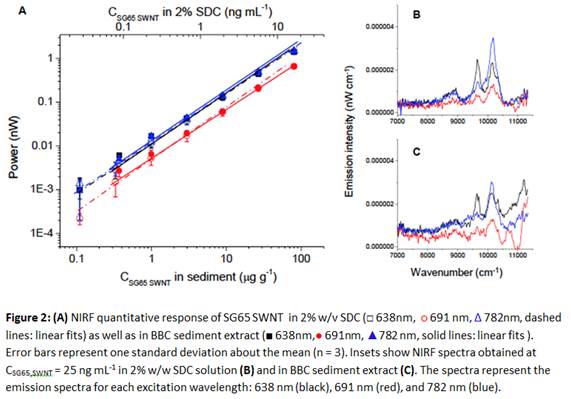
Figure 2 illustrates the quantitative response of the NIRF spectroscopy method for SG65 SWNT in sediment extract compared to 2% w/v SDC. Quantitation of SWNT in 2% w/v SDC suspension as well as in sediment extract was reliable and linear over more than three orders of magnitude (Figure 2). However, matrix effects decreased sensitivity somewhat in sediment relative to pure SDC solutions as the comparison of the NIRF spectra obtained at CSWNT SG65 = 25 ng mL-1 in 2% w/v SDC and in sediment extract revealed (Figure 2b and 2c). NIRF spectra for the 638 nm and 782 nm excitation wavelengths yielded characteristic spectral features of SG65 SWNT, indicating the presence of these SWNT, whereas for the 691 nm excitation wavelength unique identification of SWNT species was not possible. Instrument detection limits (IDL) were calculated as the SWNT concentration in 2% SDC solution that gave spectral peaks greater than 3x the amplitude of noise within the same spectral region in measured blank samples, based on data presented in Figure 2 (n ³ 4). IDL as determined to be 15 ng mL-1 in 2% w/v SDC.
Method detection limits (MDL) proved to be dependent on the recovery of SWNT from samples and on the level of interferences, e.g., internal filter and quenching effects by the components of the sediment matrix such as natural organic matter and colloids. MDLsediment was calculated using the same approach described above for IDLs, except in this case the measured spectra were obtained for SWNT in estuarine sediment extracts (n = 3) instead of 2% SDC solutions and the noise levels were determined from control (unspiked) sediment extracts. The initial MDLsediment was determined to be 525 ng g-1 in sediment after correction for recovery and sediment mass. In order to increase the sensitivity of SWNT analysis in sediment extracts, a concentration/clean-up step was developed, achieving a 10-fold concentration factor. With the concentration step the final MDLsediment was determined to be 62 ng g-1 in sediment. In addition to concentrating the SWNT sample, this ultracentrifugation step served as a matrix purification method, since dissolved substances (e.g., DOM and small organic molecules) were not sedimented by ultracentrifugation. This concentration step was included in the sample analysis method in cases where we found evidence for the presence of SWNT in the samples based on observed spectral features (peaks), but where signals were below the initial MDL.
The work presented here was focused on estuarine sediments/water; however, we have found that the method performs quite well in freshwater samples of sediment, water, and tissue. For example, we have also utilized our method to analyze wastewater produced during commercial SWNT production, using ultracentrifugation to concentrate water samples from 24 to 4 mL. This method enabled us to achieve an MDLwater of 1 ng L-1 (in the waste water).
It is important to note that unlike thermogravimetric methods e.g. CTO-375 and thermogravimetry-mass spectrometry, NIRF spectroscopy is uniquely suited to detect and quantify only carbon nanotubes in environmental samples. In particular, NIRF spectroscopy does not suffer from interferences caused by the presence of black carbon (BC), e.g., soot and chars in the samples. We have tested diesel soot as a model BC and have observed no emission in the NIR region. NIRF emission is a unique property of semiconducting SWNT, which is not shared by any other allotrope of black carbon.
After developing a highly sensitive and robust method for quantifying SWNT in sediments, we then performed a whole-sediment microcosm exposure experiment, designed to assess effects and uptake of SWNT in estuarine meiobenthic organisms at the community-scale (addressing Objective #2). Twenty intact meiofaunal microcosm cores (120 mm diameter) were collected in June 2009 from Oyster Landing in the North Inlet National Estuarine Research Reserve, Georgetown, South Carolina. The cores were pressed approximately 10 cm into the mudflat at low tide, excavated on one side, and removed intact using a double O-ring seal slipped into the bottom of each core. In the laboratory, microcosm cores were moved to an O2 saturated, carbon-polished flow-through seawater system (30 ppt; 22°C, pH 8.0 ± 0.3) under mild airlift mixing (Figure 3). The microcosms were acclimated for 7 days under dripping flow with outflow of meiobenthos prevented by 63 mm mesh inserts sealed at a diagonal in each core. All visible macrobenthos (~99% Nassarius snails) were removed with forceps within 24 hr. Pre-washed, sterile, 63 mm press-sieved Spartina marsh mud was spiked with two types of CoMoCat SWNTs (SG65 and CG200 grades, from SouthWest Nanotechnologies) at nominal concentrations of 0, 1 and 10 mg-SWNT/Kg-dry wt. These sediments were rolled on a jar mill for 7 days at 4C to yield a homogeneous SWNT-sediment mixture (slurry = 9.4% solids, 3.5% OC; median diameter 0.4 mm). Aliquots of 240-mL SWNT-spiked or SWNT-free sediment slurry were pipetted into triplicate randomized microcosms to yield a final 1.5-cm surface layer after settlement. One hundred fifty juvenile Streblospio worms were added to each microcosm on day zero for future SWNT body-burden analysis. Meiobenthos and small macrobenthos (i.e., polychaete worms and juvenile Mercenaria clams) moved into the new oxic horizon within 24 hours.
Microcosm communities were exposed to SWNT-spiked sediment for 24 days with feedings of thermally shocked phytoplankters (15-mL X 107 cells/mL) every fourth day, and harvest of the entire oxic-layer community was performed on day 24 by vacuum. Sediment was collected and analyzed for SWNT concentrations using NIRF spectroscopy. On day 24, all Streblospio were collected alive, placed into algal-enriched sediments, and allowed to feed/purge their guts for 4 hours. These worms were then analyzed for SWNT tissue body burdens using NIRF spectroscopy as described above. All other meiofauna were sieved from sediments and incubated (3 hrs) in the viability stain “CellTracker Green,” fixed in 4% phosphate-buffered glutaraldehyde, and later scored to species, lifestage and sex, as dead or alive, using a fluorescence stereomicroscope with BODIPY cube set.
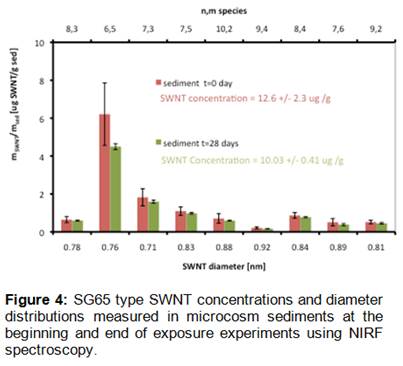
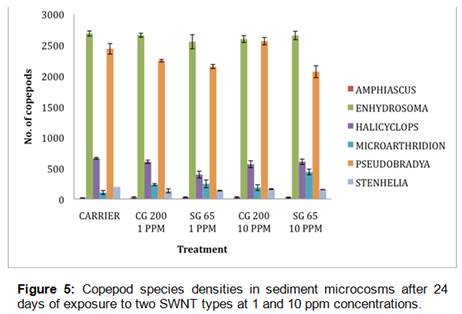
Sediment analyses confirmed that SWNT concentrations remained relatively constant throughout the microcosm exposure duration (28 days) and that nanotube diameter distributions did not change over the course of the experiment (Figure 4). Analyses of worm body burdens from microcosm experiments showed that SWNT accumulation was below the instrument detection limits for the NIRF method, indicating that Streblospio may not bioaccumulate SWNT to a great extent from contaminated sediments. Further, SAS ANOVA and multiple comparison tests (Tukey’s T-test) across microcosm treatments for each species of meiofauna found no significant detrimental effects of any SWNT type or concentration on any meiofaunal taxon at the 0.05 significance level (Figure 5). The CG200 type SWNT at 10 ppm did show a stimulatory effect on densities of Amphiascus tenuiremis copepodites relative to controls, but in comparison to other copepod species, A. tenuiremis recruitment was sharply lower over all microcosms irrespective of treatment. Taken together, these results seem to indicate relatively benign behavior of SWNT in the context of estuarine sediments and associated benthic meiofauna.
II. Fate of SWNT in a wetland mesocosm ecosystem
Building further upon our successful development and validation of a sensitive and selective NIRF spectroscopy method for detecting, characterizing, and quantifying SWNT in estuarine sediments and biota (Objective #1), we have applied this method to determine the fate of SWNT in wetland mesocosm environments after application of a SWNT dose to the water column. We have also performed experiments aimed at understanding the uptake and bioaccumulation potential of short tubular nanocarbon (STNC) SWNT manufacturing byproducts in estuarine meiofaunal organisms using a sediment microcosm bioassay approach in combination with 14C-STNC materials (Objective #2).
The analytical method we use for SWNT detection is based on the intense bandgap photoluminescence produced by pristine, isolated semiconductive SWNT (e.g., those with diameters and chiralities described by n,m circumference vector values such that n ≠ m and [n-m]/3 is not an integer) in the near infrared (~900 – 1,600 nm) range when excited by red light (600-700 nm). These spectral features are diagnostic of SWNT structure and therefore we have shown that NIRF spectroscopy is capable of detecting semiconductive SWNT in complex, carbon-rich backgrounds such as biological tissue because the intrinsic emission characteristics of these materials are very unique and do not overlap commonly occurring organic compounds.
Using this method, we have performed the first study addressing the fate and transport of SWNT in a highly complex terrestrial/aquatic ecosystem. We have utilized tightly controlled and highly instrumented wetland ecosystems (i.e., mesocosms) located in the Duke Forest, Durham, NC. The experimental design was configured to study the impact of nanomaterials on a complex ecosystem and to address questions including fate and transport, effect on community structure, effects on biogeochemical function and effects on productivity of the ecosystem. In Summer 2010, one wetland mesocosm (12’x4’, V = 4.4 m2, Soil depth ~ 9”, water level variable) consisting of aquatic and terrestrial compartments was amended with gum arabic (GA)-coated CoMoCat SWNT (c SWNT,0 = 2.5 mg L-1 in 0.5% GA). The distribution of CoMoCat SWNT in the mesocosm was followed during 6 months using NIRF spectroscopy. Samples from representative biotic and abiotic compartments were analyzed to evaluate the transport and fate of SWNT in the ecosystem.
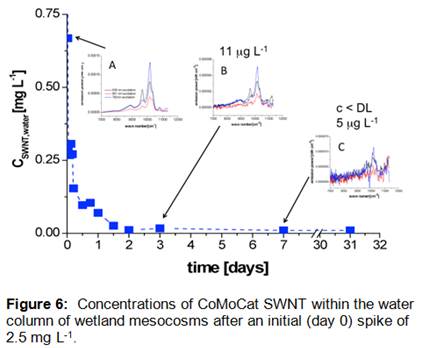
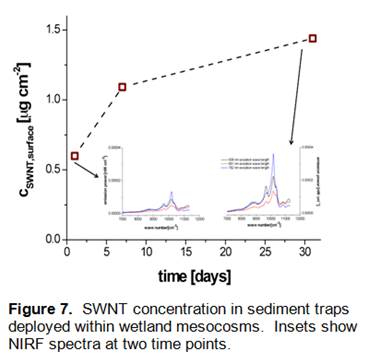
Results (Figure 6) indicate that Concentrations of CoMoCat SWNT within the water column attenuated rapidly, with near total disappearance observed within a period of 2 days (Cw,t/Cw,0 < 0.01) after spiking. Insets show NIRF spectra of CoMoCat SWNT extracted using 2% sodium deoxycholate surfactant solutions from water samples at 0.5h (A), 3 days (B) and 7 days (C). As illustrated in Figure 7, the loss of SWNT from the wetland mesocosm water was accompanied by an increase in concentration within sediment traps, which were deployed within the mesocosm to catch settling particles that might contain SWNT. This increase of SWNT concentration in sediment traps was observed over a period of 30 days, revealing that loss of SWNT from the water column was likely due to association of these particle-reactive nanoparticles to the surface of suspended sediment particles. CSWNT,surface concentration was normalized to the total surface area of the sediment traps.
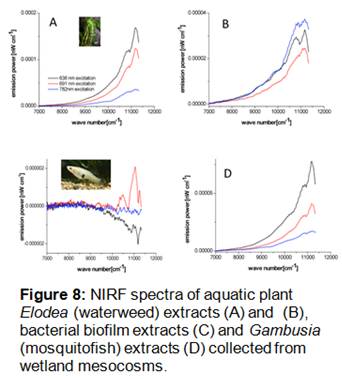
NIRF analysis of plant, biofilm, and fish samples collected from the wetland mesocosms after 8 months showed no evidence of SWNT accumulation in these biological compartments (Figure 8). Spectra for these samples (particularly from waterweed extracts) were characterized by relatively high spectral baseline and lack of SWNT-specific photoluminescence bands. Based on these results, it is unlikely that SWNT were accumulated at significant levels by biological organisms within the wetland mesocosms. These results are consistent with our previous findings that SWNT were not readily bioaccumulated by estuarine meiofaunal copepods and polychaete worms.
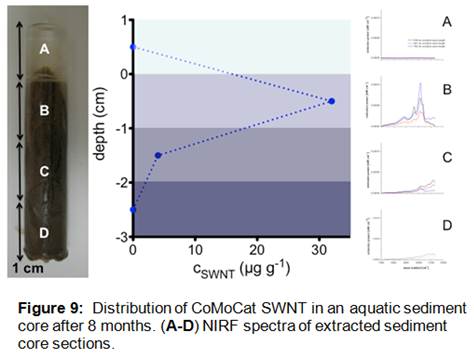
Analysis of sediment core samples taken from the wetland mesocosms 8 months after the addition of SWNT showed clear evidence of residual SWNT in the top two centimeters of the core (Figure 9). NIRF spectra from these samples were characterized by intense bandgap photoluminescence features consistent with the emissions of SWNT. These data are consistent with the rapid loss of SWNT from the mesocosm water column (Figure 6) as well as the accumulation of SWNT within deployed sediment traps (Figure 7). Based on quantification of the SWNT concentrations within the top two centimeters of the collected sediment cores, we have estimated a total mass-balance of SWNT within the wetland mesocosms as 60%. Although it is unclear at this time what the fate of the remaining 40% of the initially added SWNT may have been, the results shown here clearly indicate that the most important process impacting the fate of SWNT in aquatic systems is likely association with suspended particles and subsequent deposition and sequestration within bedded sediments. These results have implications for the long-term fate of SWNT in aquatic systems, as such deposition and burial may lead to preservation of these nanomaterial contaminants and corresponding long environmental persistence.
III. Effects and accumulation of SWNT manufacturing byproducts in estuarine meiobenthic copepods
Previous work in our laboratories has found that seawater- and sediment-associated single-walled carbon nanotubes (SWNT) of multiple types (carboxylated, metallic, semi-conducting) are practically non-toxic and non-bioaccumulated by meiobenthic copepods, nematodes and foraminifera (i.e., the major taxonomic groups comprising the marine meiobenthos) up to and including 10 mg-SWNT/mL seawater or g-sediment. However, we have also found that metal-free “as prepared” arc-discharged SWNT and also the smallest nanocarbon “fluorescent fraction” byproduct of arc discharge synthesis caused significant developmental toxicity to the model meiobenthic copepod Amphiascus tenuiremis at 1 mg-SWNT/mL-seawater.
We have therefore conducted follow-up experiments to examine the uptake and developmental toxicity of small-sized short tubular nanocarbon (STNC) manufacturing byproducts to A. tenuiremis and multiple other meiobenthic taxa found naturally in spiked/manipulated whole-sediment meiofaunal microcosms, and in seawater alone.
SWNT nanosized manufacturing byproducts (e.g., short tubular nanocarbons, (nanohorns), fluorescent nanocarbons) may be discharged to aquatic environments via industrial effluent or landfill leachate. Brackish water in estuarine systems causes aggregation of byproducts into various clumped size-fractions of 1-20 microns, followed by sedimentation to the benthos. The meiobenthos (principally copepods, nematodes, foraminifera) feed selectively within this size range, so the likelihood of SWNT and byproduct exposure via contact and ingestion is high, as is the likelihood of meiobenthos-based nanocarbon feeding and re-packaging in fine sediments. Short tubular nanocarbons are 10-30 nm long and are produced as a byproduct of incomplete SWNT synthesis. This fraction has no practical application to date, and is subject to waste discharge. Several studies now suggest that pure SWNTs may not be bioavailable but smaller nanocarbons might be (e.g., short tubular nanocarbons (STNC), carbon dots, amorphous nanocarbon).
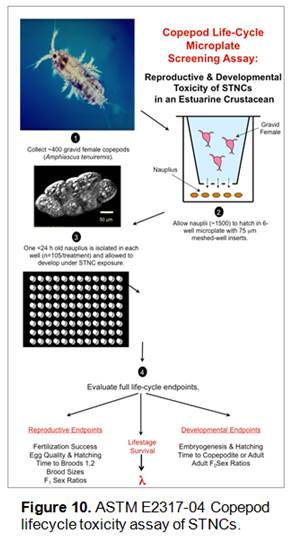
We have therefore (1) evaluated the meiofaunal and small macrofaunal uptake of 14C labeled “short tubular nanocarbons” from muddy estuarine sediments using a sediment microcosm bioassay system, and (2) tested the lifecycle toxicity of STNCs in seawater suspension to the meiobenthic copepod (below) Amphiascus tenuiremis using ASTM method E-2317-04. This work directly addressed Objective #2 of our project.
Meiobenthic copepods are a keystone taxon in most estuaries globally, and serve as a critical food source for shrimps, fish and crabs. Amphiascus tenuiremis is a mud dwelling harpacticoid copepod that also cultures well in seawater alone. In 30 g·L-1 seawater, at 25ºC, A. tenuiremis passes through three distinct life-stages, beginning with the larval-naupliar stage, followed by the copepodite stage, and ending in an adult stage in approximately 15-18 days. Sexually dimorphic adults then mate and produce one clutch every 3-4 days over a 20-30 day period. Each clutch averages 6-8 embryos in seawater culture, or 12-16 embryos in sediment culture, with five to seven clutches per lifetime. We have a detailed life table for this species that allows fine-scale population growth modeling and prediction.
To begin the experiment, 15 intact muddy sediment cores were collected early summer from a common 1 m2 area of salt-marsh mudflat in North Inlet, SC, placed on running seawater in laboratory, and fed phytoplankters for 3 months acclimation. On day 7 post-collection, 100 gravid A. tenuiremis were added to each microcosm to establish a synchronous-aged cohort for toxicity and bioaccumulation studies. Pre-washed, sterile, < 63-mm press-sieved Spartina marsh mud was spiked with 7.24 mL 14C-STNC at 187 mg-STNC/g-sed dry wt. Sediments were rolled on a jar mill for 14 days at 4C to yield a homogeneous STNC-sediment slurry (8.7% solids; median f 4 mm). At 3 months, a 1 cm layer of 14C STNC amended sediments was layered on top of each microcosm and incubated under mild aeration and dripping flow for 14 days. 113 mL STNC or STNC-free sediment slurry was pipetted into seven randomized microcosms (n = 3 controls; n = 4 treatments). Meiobenthos and small macrobenthos moved up into the STNC layer and were harvested after 14 d exposure. Ten 1 cm2 subcores were taken from each microcosm for faunal counts.
All fauna were depurated in clean sediments for 4-6 h, digested in Scintigest, and assayed for 14C STNC uptake by liquid scintillation counting. Any potential chemiluminescence was quenched by addition of 0.07 mL acetic acid per LSC vial.
Copepod lifecycle toxicities of cold STNCs were tested via ASTM E2317-04 (Figure 10). Briefly, using 96-well microplates, < 24h old naupliar larvae were placed individually in wells loaded with 250 μL of a STNC solution in 30 g·L-1 seawater (n = 30 nauplii/plate/exposure, 3 replicates per exposure). STNC exposures were: 0 mg·L-1, 0.1 mg·L-1, 0.3 mg·L-1, 0.6 mg·L-1, and 1 mg·L-1. Treatments were prepared from a concentrated, dialyzed STNC:DI water stock by dilution with 0.2 μm filtered seawater. Control solutions were 0.2 μm filtered 30 g·L-1 seawater alone. Dialyzed STNC stock concentrations were measured gravimetrically. The diluted stock was vortexed and pipetted immediately into microwells. Copepods were removed and placed into new microwells containing fresh STNC solutions every 4 days to insure consistent STNC exposure. Immediately following STNC:seawater renewal, each microwell was loaded with 2 μl of a 107 algal cells mL-1 food stock. Copepods were monitored daily for ASTM E2317-04 endpoints (see Figure 10).
| TAXON | CONTROL (ng 14C-STNC/g) | TREATMENT (ng 14C-STNC/g) |
|---|---|---|
| Mercenaria mercenaria (203 mm) (clam) | < LOD (30 dpm) | 402 ± 297 |
| Mercenaria mercenaria (depurated 3-4 h) | < LOD (30 dpm) | 10.2* |
| Streblospio benedicti (polychaele 10-15 mm) | 37 ± 27 | 357 ± 190 |
| Neris succinea (polychaaela worm) | 80 ± 83 | 71 ± 48 |
| Nassarius obsoleta (3-4 mm) (mud snail) | < LOD (30 dpm) | 172 ± 144 |
| Nassarius obsoleta (depurated 3-4 h) | < LOD (30 dpm) | 48.5* |
| Amphiascus tenulremis (male & female copepods) | < LOD (30 dpm) | 0.091 ± 0.1 ng/copepod = (~91 ± 100 ng-STNC/g dry) |
| Amaphiascus tenulremis (depurated 3-4 h) | < LOD (30 dpm) | 0.0012 ng/copepod* |
Table 1. Taxon-specific 14C-STNC body burdens (ng/wet) after 14 days exposure (mean ± 1SD).
Values are blank corrected. Coepod mean dry weight per copepod is 0.9 - 1.2 mg. 50% of
recovered live fauna were depurated 3-4 hours in cold sediments enriched with centrifuge
phytoplankter food. Time zero sediment conc. = 208 mg/g. * n = 1 replicate due to low tissue
mass constraints.
Results of STNC bioaccumulation assays indicated that after exposure to 200 mg/g concentrations, organisms accumulated only low nanogram body burdens for most fauna (Table 1). Mercenaria clam juveniles (undepurated) had highest mean STNC burden at 402 ng/g, but shed almost all of this burdent after a 3-4 hour feeding on cold sediments. Similarly, Streblospio worms shed 90%, mud snails shed 72%, and copepods 98% of initially measured burdens. Burdens were therefore largely tied to ingested sediments in the gastrointestinal tract.
| Short Tublar Nanocarbon Sediment adn Water Concentrations | ||
|---|---|---|
| T=0 | T=14 d | |
| Control Sediments (3 microcosm x 3 reps) | < LOD (45 dpm) | < LOD (45 dpm) |
| Treatment Sediments (microcosm-C1 surface) | 208 (± 10.8) | 12.8 (± 10.4 |
| 7.0 (± 3.9) | ||
| 8.4 (± 7.1) | ||
| 11.5 (± 12 dpm) | ||
| Overlying H20 (n=3 x 6 reps over 14d | < LOD (123dpm)* | < LOD (123 dpm)* |
| Deep Sediments | < LOD (45 dpm)* | < LOD (45 dpm)* |
Table 2. Mocrocosm mean STNC concentrations (blank corrected) in overlying water, top
1-cm "fluff" layer, and the deeper sediment horizon. n= triplicates/microcosm/tim.
Time-zero spiked sediment concentration = 14C activity at time of sediment addition to microcosm.
STNC concentrations in 0-1 cm deep surface sediments declined to ~10 mg/g on average after 14 days of re-working by meio- and small macrobenthos (Table 2). Surface concentrations became highly variable (patchy) in horizontal space (i.e., high sample-to-sample variance within a 150 cm2 area). STNC concentrations “downcore” in deeper sediments were below LOD’s presumably due to dilution by a much larger deep sediment volume (>12 fold dilution volume) in this microcosm design and also lack of deep transport of STNCs within the vertical horizon of the core by bioturbation.
Measured concentrations of 14C-STNC in overlying water remained below detection throughout the experiment. This indicates that STNCs did not evolve appreciably from sediments into the water column, consistent with very strong association of these nanoparticulate contaminants with sediment particles. This result is in accordance with the results reported above for SWNT in wetland mesocosm environments.
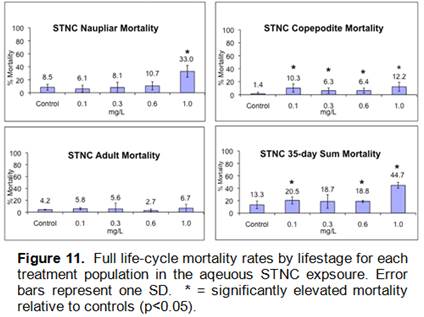
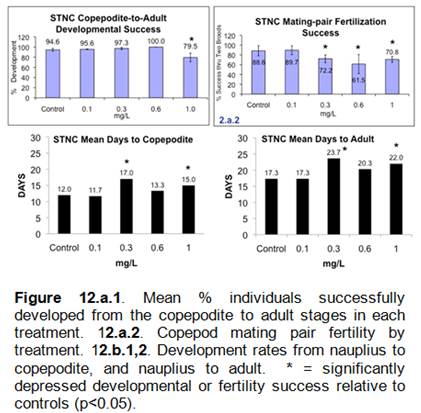
Aqueous STNC suspensions in microplates were chronically toxic to copepods for multiple endpoints (Figures 10,11), but no toxicity or biologically significant tissue accumulation was observed for any copepods in native microcosm sediments.
Overall, these results indicate that SWNT and STNCs are recalcitrant to biological uptake from both sediment and water by aquatic invertebrates. While some toxicity was observed for STNCs in estuarine copepods using a full life cycle test, these nanomaterials can be characterized as only moderately toxic. It is clear that sediment association and associated burial and preservation will be an important environmental fate process for carbon based nanomaterials. Both NIRF spectroscopy and radiolabeling approaches offer high sensitivity analysis and tolerance of complex environmental media when tracking environmental fate and biological uptake of carbon nanomaterials. NIRF spectroscopy in particular shows great promise for surveying environmental media for SWNT contamination.
IV. Bioaccumulation and toxicity of SWNT to benthic organisms at the base of the marine food chain
Due to sample preparation and analytical detection challenges, very few ecotoxicological studies have examined the effects of pristine, non-functionalized SWNT with minimal metal impurities. Also, sensitive and specific analytical methods have not previously been available for quantitatively measuring SWNT in environmental samples (e.g., sediments, tissues). However, our development of NIRF spectroscopy as a sensitive and selective analytical method for quantifying SWNT in the aquatic environment has enabled detailed study of the fate and effects of these nanomaterials. We have therefore utilized several benthic organisms at the base of the marine food chain including the estuarine amphipod, Ampelisca abdita, and mysid shrimp, Americamysis bahia, to investigate the potential bioavailability, bioaccumulation and toxicity of four distinct SWNT materials through a sediment exposure route. Further, one SWNT material was used to explore the effect of exposure route (e.g., sediment and/or food source) on SWNT bioavailability, bioaccumulation and toxicity. Sediment and food exposures were selected rather than an aqueous route because SWNT are not very water soluble and are more likely to associate with particles including food and suspended sediment particles when in aqueous solution [36-38]. NIRF spectroscopy was utilized to determine the concentration and specific SWNT species (n,m) present in each environmental matrix (sediment, tissues). In addition to evaluating pristine semiconducting SWNT, [14C]SWNT were used to analyze the potential bioavailability, bioaccumulation and toxicity of carboxylic acid-functionalized SWNT in the estuarine amphipod L. plumulosus via sediment and/or food exposures. Taken together, these studies provide an indication of whether SWNT are likely to enter the marine food chain.
SWNT used in the current work were CoMoCAT nanotubes (SG65, SG76, CG100) from SouthWest NanoTechnologies, Inc (SWeNT), a proposed standard reference material SWNT from the Organization for Economic Co-operation and Development (OECD), and [14C]SWNT produced by the arc-discharge method (Research Triangle Institute). Materials from SWeNT are well-characterized, high purity mixtures of SWNT with either narrow diameter distributions and specificity (SG65, SG76) or broad diameter distributions (CG100)[39-42]. For characterization of the SWNT stocks, stable suspensions of SWNT were prepared in 2% w/v sodium deoxycholate (SDC) using high power sonication at 50% amplitude for 10 minutes in a salt-water ice bath (Branson 450D Sonifier) followed by centrifugation at 17,860 g and 22°C for 30 minutes (accuSpin™ 1R, Fisher Scientific) to remove non-suspended SWNT bundles. Toxicity and bioaccumulation test suspensions of the unlabeled SWNT materials were prepared at 1 mg SWNT/mL 2% w/v SDC with no centrifugation step. Stock suspensions were resonicated prior to amendment to sediment or food sources if not used immediately after preparation.
The [14C]SWNT were purified using nitric acid to introduce oxygen containing groups such as carboxyl to defect and end sites [43-45]. The [14C]SWNT stock was 1.15 mg [14C]SWNT/mL deionized water with an activity of 1.63 µCi/mL. This stock was resonicated for 10 minutes at 80-100 watts (Fisher Scientific Model 100 Sonic Dismembrator) prior to use.
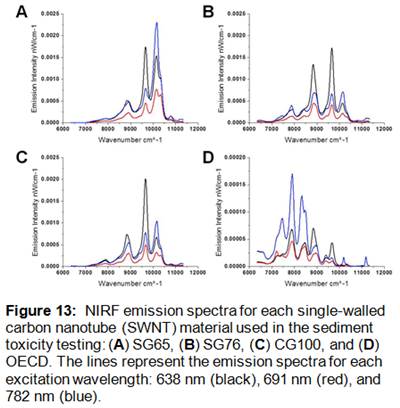
Stock SWNT suspensions and sample extracts (i.e., sediment, food and organism) were analyzed for near infrared fluorescence and absorbance as described in detail above using a NS1 Nanospectralyzer (Applied NanoFluorescence, LLC). This instrument employs three excitation lasers (638 nm, 691 nm, 782 nm) and detects fluorescence emission from 880-1580 nm with an InGaAs array detector cooled to -18 °C. The measurement parameters were as follows: 100 ms fluorescence integration time with 100 spectral averages for each excitation wavelength, and 60 ms absorbance integration time with one spectral average. Resulting NIRF emission spectra were analyzed with spectral fitting software to determine the specific SWNT chirality (n,m) species present. This determination is based on data collected by Weisman and Bachilo [29] demonstrating that each SWNT species has a unique diameter, chiral wrapping angle, and excitation-emission spectrum. The unique spectral signature allows for identification of individual species in SWNT mixtures. NIRF and absorbance spectra were measured to obtain quantitative and qualitative characterization information for each SWNT stock suspension (Figure 13).
We have found that 2% w/v SDC is useful in extracting and resuspending SWNT from sediment, water and biota samples [46]. Sediment extractions were performed using approximately 1.5 g wet sediment. The sample was washed with 6 mL of deionized water twice by vortexing and centrifuging the sample for 10 minutes at 13,520 g and 20°C (Eppendorf centrifuge 5804 R). Each wash was collected and saved. The final pellet was resupended in 2.5 mL 2% w/v SDC by ultrasonication for 10 minutes at 50% amplitude in a salt water ice bath. This sample was centrifuged for 15 minutes at 13,520 g and 20°C and the supernatant was saved. The extraction was repeated two-three times. A known, equal volume of each extract was combined for analysis and quantitation by NIRF spectroscopy. The samples were measured against a sediment blank extract in order to account for interference in the spectra.
Brine shrimp and algae were extracted by centrifuging the sample at 13,520 g for 10 minutes at 22°C and resuspending the pellet in 2 mL 2% w/v SDC by ultrasonication. The suspension was centrifuged at 17,860 g for 10 minutes at 22°C and the supernatant was measured and quantified by NIRF spectroscopy. Amphipods and mysids were extracted using the same method without the initial centrifugation step. If SWNT were not detected, the extracts were concentrated via ultracentrifugation for 2 hours at 369,734 g and 20°C (SW 60 rotor, L8-80 Ultracentrifuge, Beckman Coulter) and re-measured by NIRF spectroscopy.
Seven day static toxicity tests with A. bahia (mysid) and A. abdita (amphipod) followed the methods described by Ho et al. [47]. Ten individuals of each organism type were added to test chambers containing 20 g wet sediment and 60 mL of 30‰ reconstituted seawater (RSW) prepared by diluting 100‰ brine with deionized water. Chambers were static and aerated in a 20°C incubator with a light cycle of 16h:8h light:dark. Long Island Sound (LIS) reference sediment (1.8% organic carbon, 92% silt and clay-sized particles) was amended with SWNT [48]. Four SWNT were tested (SG65, SG76, CG100, and OECD) at three nominal concentrations each (0.1, 1, and 10 µg SWNT/g dry sediment), and toxicity in amended sediments was compared to toxicity in reference sediment and carrier controls (2% w/v SDC). Measured concentrations of SWNT in the sediment are reported (Supplemental Information, Table S1). Each treatment consisted of four replicate chambers. Three replicates per treatment were saved for NIRF analysis and one replicate for microscopy of the mysids and amphipods. Details of the sediment composition, SWNT amendment of sediment and food, and sample extraction methods are described in the Supplemental Information. Mysids were fed approximately 100 mL of 0.15 g wet Artemia salina/mL RSW per chamber daily. On day 7, organisms were sieved (0.5 mm) and counted to determine survival, missing organisms were considered mortalities. Dissolved oxygen (DO), pH, salinity and temperature were monitored throughout the test.
Seven day bioaccumulation studies were conducted as described for the toxicity tests, but utilized only the SG65 SWNT-amended LIS sediment. Treatments for the present study included (i) unamended sediment, (ii) 2% w/v SDC-amended sediment, (iii) SWNT-amended sediment with unamended food, (iv) unamended sediment with SWNT-amended food, and (v) SWNT-amended sediment with SWNT-amended food. Each treatment consisted of six chambers per organism type for the unamended treatment (three each for depuration and non-depuration) while all other treatments consisted of eight chambers per organism type (three each for depuration and non-depuration NIRF spectroscopy analysis and one each for depuration and non-depuration microscopy analysis). Percent survival included data from all chambers per treatment for each organism type (six for unamended sediment and eight for all other treatments). The amended food sources for the amphipods and mysids were Cyclotella meneghiniana (algae) and A. salina (brine shrimp), respectively. Nominal concentrations were 10 µg SWNT/g dry sediment, 10 µg SWNT/mL algae culture, or 10 µg SWNT/g wet brine shrimp. Measured concentrations of SWNT in the sediment and food sources are reported (Supplemental Information, Table S2). Upon termination, the non-depurated replicates were sieved (0.5 mm), counted to determine survival, and saved for analysis. The replicates saved for depuration were sieved (0.5 mm), counted to determine survival, transferred to control reference sediment and water, fed control food, and left for 24 hours. After this depuration period, the organisms were sieved (0.5 mm) and saved for analysis. Sediment and food amendment methods as well as sample extraction are described in the Supplemental Information.
L. plumulosus was exposed to [14C]SWNT-amended Isochrysis galbana and/or reference sediment in a static-renewal design for 28 days. The reference sediment was from Bread and Butter Creek (BBC) in the North Inlet Estuarine Research Reserve near Georgetown, SC, USA (median grain size of 4 µm, 98% silt/clay, 2–4% organic carbon [49]). Each chamber contained 20 adult amphipods and 50 juvenile amphipods in 35 mL of sediment slurry (0.1106 g dry/mL slurry) and 150 mL of 15‰ aerated RSW held in a 25°C incubator with a light cycle of 16h:8h light:dark. The treatments were as follows (i) unamended sediment and algae, (ii) unamended sediment and amended algae at 10 or 100 µg [14C]SWNT/g dry algae, (iii) unamended algae and amended sediment at 10 or 100 µg [14C]SWNT/g dry sediment, and (iv) amended sediment and amended algae at 10 or 100 µg [14C]SWNT/g dry sediment or g dry algae. Each treatment included six chambers (three for depurated organisms and three for non-depurated organisms). Measured concentrations of [14C]SWNT in the sediment at day 0 and day 28 as well as the average in the amended algae are reported (Supplemental Information, Table S3). Amphipods were fed three times per week after a 50% water renewal (15‰ seawater filtered through a 0.2 µm filter). Physical measurements (i.e., DO, pH, salinity, and temperature) were monitored twice per week.
Upon termination of the test, half of the replicates were depurated and the other half was analyzed. Depuration was performed by transferring the organisms to control sediment and water where they were fed unamended algae for 24 hours before analysis. Overlying water, sediment, fecal pellets, and organisms were collected for analysis. Fecal pellets and organisms were collected by sieving the sediment of each replicate over a 25
Conclusions:
We have successfully developed, validated, and applied a new method for detecting SWNT in the aquatic environment, based on near infrared fluorescence spectroscopy. The high fidelity of NIRF spectroscopy in assessing the quantitative and qualitative properties of semiconducting SWNT in complex estuarine sediment and biota underscores the value of developing methods for analysis of engineered nanoparticles in the aquatic environment. Our NIRF method is capable of characterization of any and all semiconducting SWNT present in environmental samples and achieves reliable detection without previous knowledge about chirality/diameter distribution. Quantitative information can be obtained using authentic standards. The developed method represents the most sensitive and selective technique currently available for analyzing SWNT in environmental samples, even in highly complex, particle-rich samples (e.g., sediment). It is clear that the high selectivity of the NIRF technique, based on SWNT-specific electronic transitions coupled to the generally low NIR fluorescent background and consequent high sensitivity in most biological and environmental samples allows this technique to serve as a useful method for conducting both laboratory- and field-based fate, transport, and effects research on SWNT in the environment. However, challenges remain in reducing potential optical matrix effects (e.g. internal filter effects) and novel instrumentation combinations such as NIRF with upstream asymmetric flow field flow fractionation (AF4) may yield even more powerful analytical solutions for assessing the fate of SWNT in the aquatic environment.
Application of both the newly developed NIRF spectroscopy technique as well as custom-synthesized radiolabeled SWNT materials to assess the toxicity, fate, and biological uptake of SWNT in the aquatic environment revealed that SWNT associate strongly with particulate phases after entering aquatic systems. Benthic communities appear to be the most important sink for SWNT after entry into the aquatic environment, as these materials become incorporated in bedded sediment. Detailed studies with meiobenthic and macrobenthic estuarine invertebrates revealed that SWNT are practically non-toxic to these organisms and that they may be ingested but not bioaccumulated in tissues to a high level. There also was little or no evidence of SWNT trophic transfer within simple benthic food webs as assessed by feeding studies. Taken together, our results indicate that SWNT may persist in the aquatic environment (particularly in sediments) but that they may pose minimal risk to aquatic life.
References:
- Alpatova, A.L., et al., Single-walled carbon nanotubes dispersed in aqueous media via non-covalent functionalization: Effect of dispersant on the stability, cytotoxicity, and epigenetic toxicity of nanotube suspensions. Water Research, 2010. 44(2): p. 505-520.
- Bauer, B.J., et al., Measurement of single-wall nanotube dispersion by size exclusion chromatography. Journal of Physical Chemistry C, 2007. 111(48): p. 17914-17918.
- Jaisi, D.P., et al., Transport of Single-Walled Carbon Nanotubes in Porous Media: Filtration Mechanisms and Reversibility. Environmental Science & Technology, 2008. 42(22): p. 8317-8323.
- Plata, D.L., C.M. Reddy, and P.M. Gschwend, Thermogravimetry–Mass Spectrometry for Carbon Nanotube Detection in Complex Mixtures. Environmental Science & Technology, 2012.
- Ferguson, P.L., et al., Influence of sediment-amendment with single-walled carbon nanotubes and diesel soot on bioaccumulation of hydrophobic organic contaminants by benthic invertebrates. Environmental Science & Technology, 2008. 42(10): p. 3879-3885.
- Petersen, E.J., et al., Biological Uptake and Depuration of Carbon Nano-tubes by Daphnia magna. Environmental Science & Technology, 2009. 43(8): p. 2969-2975.
- Hyung, H., et al., Natural organic matter stabilizes carbon nanotubes in the aqueous phase. Environmental Science & Technology, 2007. 41(1): p. 179-184.
- Sano, M., J. Okamura, and S. Shinkai, Colloidal nature of single-walled carbon nanotubes in electrolyte solution: The Schulze-Hardy rule. Langmuir, 2001. 17(22): p. 7172-7173.
- Petersen, E.J., et al., Effects of Polyethyleneimine-Mediated Functionalization of Multi-Walled Carbon Nanotubes on Earthworm Bioaccumulation and Sorption by Soils. Environmental Science & Technology, 2011. 45(8): p. 3718-3724.
- Esquivel, E.V. and L.E. Murr, A TENT analysis of nanoparticulates in a Polar ice core. Materials Characterization, 2004. 52(1): p. 15-25.
- Sobek, A. and T.D. Bucheli, Testing the resistance of single- and multi-walled carbon nanotubes to chemothermal oxidation used to isolate soots from environmental samples. Environmental Pollution, 2009. 157(4): p. 1065-1071.
- Ziolkowski, L.A. and E.R.M. Druffel, The feasibility of isolation and detection of fullerenes and carbon nanotubes using the benzene polycarboxylic acid method. Marine Pollution Bulletin, 2009. 59(4-7): p. 213-218.
- Weisman, R., Fluorimetric characterization of single-walled carbon nanotubes. Analytical and Bioanalytical Chemistry, 2010. 396(3): p. 1015-1023.
- Weisman, B., ed. Optical Spectroscopy of Single-Walled Carbon Nanotubes. Contemporary Concepts of Condensed Matter Science. Carbon Nanotubes: Quantum Cylinders of Graphene, ed. S. Saito and A. Zettl. Vol. 3. 2008. 109-133.
- Bonaccorso, F., et al., Density Gradient Ultracentrifugation of Nanotubes: Interplay of Bundling and Surfactants Encapsulation. The Journal of Physical Chemistry C, 2010. 114(41): p. 17267-17285.
- Green, A., M. Duch, and M. Hersam, Isolation of single-walled carbon nanotube enantiomers by density differentiation. Nano Research, 2009. 2(1): p. 69-77.
- O'Connell, M.J., et al., Band Gap Fluorescence from Individual Single-Walled Carbon Nanotubes. Science, 2002. 297(5581): p. 593-596.
- Weisman, R.B., S.M. Bachilo, and D. Tsyboulski, Fluorescence spectroscopy of single-walled carbon nanotubes in aqueous suspension. Applied Physics a-Materials Science & Processing, 2004. 78(8): p. 1111-1116.
- Moore, V.C., et al., Individually suspended single-walled carbon nanotubes in various surfactants. Nano Letters, 2003. 3(10): p. 1379-1382.
- Yamamoto, Y., et al., Fundamental properties of oligo double-stranded DNA/single-walled carbon nanotube nanobiohybrids. Nanoscale, 2010. 2(9): p. 1767-72.
- Noguchi, Y., et al., Single-walled carbon nanotubes/DNA hybrids in water are highly stable. Chemical Physics Letters, 2008. 455(4-6): p. 249-251.
- Yi, W.H., et al., Wrapping of single-walled carbon nanotubes by a pi-conjugated polymer: The role of polymer conformation-controlled size selectivity. Journal of Physical Chemistry B, 2008. 112(39): p. 12263-12269.
- Decker, J.E., et al., Sample preparation protocols for realization of reproducible characterization of single-wall carbon nanotubes. Metrologia, 2009. 46(6): p. 682-692.
- Cherukuri, P., et al., Near-infrared fluorescence microscopy of single-walled carbon nanotubes in phagocytic cells. Journal of the American Chemical Society, 2004. 126(48): p. 15638-15639.
- Irurzun, V.M., Y.Q. Tan, and D.E. Resasco, Sol-Gel Synthesis and Characterization of Co-Mo/Silica Catalysts for Single-Walled Carbon Nanotube Production. Chemistry of Materials, 2009. 21(11): p. 2238-2246.
- Xu, X.Y., et al., Electrophoretic analysis and purification of fluorescent single-walled carbon nanotube fragments. Journal of the American Chemical Society, 2004. 126(40): p. 12736-12737.
- Burgess, R.M., S.A. Ryba, and M.G. Cantwell, Importance of organic carbon quantity on the variation Koc in marine sediments. Toxicological & Environmental Chemistry, 2000. 77(1): p. 9 - 29.
- Burgess, R.M., et al., Sorption of 2,4 '-dichlorobiphenyl and fluoranthene to a marine sediment amended with different types of black carbon. Environmental Toxicology and Chemistry, 2004. 23(11): p. 2534-2544.
- Weisman, R.B. and S.M. Bachilo, Dependence of optical transition energies on structure for single-walled carbon nanotubes in aqueous suspension: An empirical Kataura plot. Nano Letters, 2003. 3(9): p. 1235-1238.
- Fagan, J.A., et al., Length-Dependent Optical Effects in Single-Wall Carbon Nanotubes. Journal of the American Chemical Society, 2007. 129(34): p. 10607-10612.
- Tsyboulski, D.A., et al., Translational and rotational dynamics of individual single-walled carbon nanotubes in aqueous suspension. Acs Nano, 2008. 2(9): p. 1770-1776.
- Tsyboulski, D.A., et al., Structure-dependent fluorescence efficiencies of individual single-walled carbon nanotubes (vol 7, pg 3084, 2007). Nano Letters, 2008. 8(4): p. 1270-1270.
- Lin, S. and D. Blankschtein, Role of the Bile Salt Surfactant Sodium Cholate in Enhancing the Aqueous Dispersion Stability of Single-Walled Carbon Nanotubes: A Molecular Dynamics Simulation Study. The Journal of Physical Chemistry B, 2010. 114(47): p. 15616-15625.
- Zhang, L.W., et al., Interactions of C-14-labeled multi-walled carbon nanotubes with soil minerals in water. Environmental Pollution, 2012. 166: p. 75-81.
- Jaisi, D.P. and M. Elimelech, Single-Walled Carbon Nanotubes Exhibit Limited Transport in Soil Columns. Environmental Science & Technology, 2009. 43(24): p. 9161-9166.
- Gottschalk, F., et al., Modeled Environmental Concentrations of Engineered Nanomaterials (TiO2, ZnO, Ag, CNT, Fullerenes) for Different Regions. Environmental Science & Technology, 2009. 43(24): p. 9216-9222.
- Kennedy, A.J., et al., Factors influencing the partitioning and toxicity of nanotubes in the aquatic environment. Environmental Toxicology and Chemistry, 2008. 27(9): p. 1932-1941.
- Hyung, H., et al., Natural Organic Matter Stabilizes Carbon Nanotubes in the Aqueous Phase. Environmental Science & Technology, 2006. 41(1): p. 179-184.
- Resasco, D.E., et al., A Scalable Process for Production of Single-walled Carbon Nanotubes (SWNTs) by Catalytic Disproportionation of CO on a Solid Catalyst. Journal of Nanoparticle Research, 2002. 4(1): p. 131-136.
- Alvarez, W.E., et al., Characterization of Single-Walled Carbon Nanotubes (SWNTs) Produced by CO Disproportionation on Co−Mo Catalysts. Chemistry of Materials, 2002. 14(4): p. 1853-1858.
- Herrera, J.E., et al., Raman Characterization of Single-Walled Nanotubes of Various Diameters Obtained by Catalytic Disproportionation of CO. Journal of Nanoscience and Nanotechnology, 2003. 3: p. 133-138.
- Bachilo, S.M., et al., Narrow (n,m)-Distribution of Single-Walled Carbon Nanotubes Grown Using a Solid Supported Catalyst. Journal of the American Chemical Society, 2003. 125(37): p. 11186-11187.
- Ferguson, P.L., et al., Influence of Sediment−Amendment with Single-walled Carbon Nanotubes and Diesel Soot on Bioaccumulation of Hydrophobic Organic Contaminants by Benthic Invertebrates. Environmental Science & Technology, 2008. 42(10): p. 3879-3885.
- Xu, X., et al., Electrophoretic Analysis and Purification of Fluorescent Single-Walled Carbon Nanotube Fragments. Journal of the American Chemical Society, 2004. 126(40): p. 12736-12737.
- Templeton, R.C., et al., Life-cycle effects of single-walled carbon nanotubes (SWNTs) on an estuarine meiobenthic copepod. Environmental Science & Technology, 2006. 40(23): p. 7387-7393.
- Schierz, A., et al., Characterization and Quantitative Analysis of Single-Walled Carbon Nanotubes in the Aquatic Environment Using Near-Infrared Fluorescence Spectroscopy. Environmental Science & Technology, 2012. Article ASAP.
- Ho, K.T., et al., Sediment toxicity assessment: comparison of standard and new testing designs. Archives of Environmental Contamination and Toxicology, 2000. 39: p. 462-468.
- Rust, A.J., et al., Relationship between metabolism and bioaccumulation of benzo[α]pyrene in benthic invertebrates. Environmental Toxicology and Chemistry, 2004. 23(11): p. 2587-2593.
- Chandler, G.T. and A.S. Green, in New Techniques in Aquatic Toxicology, G.K. Ostrander, Editor 1996, Lewis Publishers, Inc. p. 23-39.
- Kennedy, A.J., et al., Influence of nanotube preparation in aquatic bioassays. Environmental Toxicology and Chemistry, 2009. 28(9): p. 1930-1938.
- Petersen, E.J., et al., Potential Release Pathways, Environmental Fate, And Ecological Risks of Carbon Nanotubes. Environmental Science & Technology, 2011. 45(23): p. 9837-9856.
- Roberts, A.P., et al., In vivo biomodification of lipid-coated carbon nanotubes by Daphnia magna. Environmental Science & Technology, 2007. 41(8): p. 3025-3029.
- Petersen, E.J., Q. Huang, and J.W.J. Weber, Ecological Uptake and Depuration of Carbon Nanotubes by Lumbriculus variegatus. Environ Health Perspect, 2008. 116(4): p. 496-500.
- Galloway, T., et al., Sublethal toxicity of nano-titanium dioxide and carbon nanotubes in a sediment dwelling marine polychaete. Environmental Pollution, 2010. 158(5): p. 1748-1755.
- Fraser, T.W.K., et al., Dietary toxicity of single-walled carbon nanotubes and fullerenes (C60) in rainbow trout ( Oncorhynchus mykiss). Nanotoxicology, 2011. 5(1): p. 98-108.
- Schlekat, C.E., A.W. Decho, and G.T. Chandler, Bioavailability of Particle-Associated Silver, Cadmium, and Zinc to the Estuarine Amphipod Leptocheirus plumulosus through Dietary Ingestion. Limnology and Oceanography, 2000. 45(1): p. 11-21.
- Emery, V.L., et al., Development of a chronic sublethal sediment bioassay using the estuarine amphipod Leptocheirus plumulosus (Shoemaker). Environmental Toxicology and Chemistry, 1997. 16(9): p. 1912-1920.
- McGee, B.L., et al., Assessment of sediment contamination, acute toxicity, and population viability of the estuarine amphipod Leptocheirus plumulosus in baltimore harbor, maryland, USA. Environmental Toxicology and Chemistry, 1999. 18(10): p. 2151-2160.
- Tervonen, K., et al., Analysis of fullerene-C60 and kinetic measurements for its accumulation and depuration in Daphnia magna. Environmental Toxicology and Chemistry, 2010. 29(5): p. 1072-1078.
- Reichenberg, F. and P. Mayer, Two complementary sides of bioavailability: Accessibility and chemical activity of organic contaminants in sediments and soils. Environmental Toxicology and Chemistry, 2006. 25(5): p. 1239-1245.
- Mayer, L.M., et al., Digestive environments of benthic macroinvertebrate guts: Enzymes, surfactants and dissolved organic matter. Journal of Marine Research, 1997. 55(4): p. 785-812.
- Voparil, I.M. and L.M. Mayer, Dissolution of Sedimentary Polycyclic Aromatic Hydrocarbons into the Lugworm's (Arenicola marina) Digestive Fluids. Environmental Science & Technology, 2000. 34(7): p. 1221-1228.
Journal Articles on this Report : 5 Displayed | Download in RIS Format
| Other project views: | All 21 publications | 5 publications in selected types | All 5 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Kashiwada S, Ariza ME, Kawaguchi T, Nakagame Y, Jayasinghe BS, Gartner K, Nakamura H, Kagami Y, Sabo-Attwood T, Ferguson PL, Chandler GT. Silver nanocolloids disrupt medaka embryogenesis through vital gene expressions. Environmental Science & Technology 2012;46(11):6278-6287. |
R833859 (Final) R833858 (2009) |
Exit Exit Exit |
|
|
Parks AN, Portis LM, Schierz PA, Washburn KM, Perron MM, Burgess RM, Ho KT, Chandler GT, Ferguson PL. Bioaccumulation and toxicity of single-walled carbon nanotubes to benthic organisms at the base of the marine food chain. Environmental Toxicology and Chemistry 2013;32(6):1270-1277. |
R833859 (Final) |
Exit |
|
|
Schierz A, Parks AN, Washburn KM, Chandler TG, Ferguson PL. Characterization and quantitative analysis of single-walled carbon nanotubes in the aquatic environment using near-infrared fluorescence spectroscopy. Environmental Science & Technology 2012;46(22):12262–12271. |
R833859 (Final) |
Exit |
|
|
von der Kammer F, Ferguson PL, Holden PA, Masion A, Rogers KR, Klaine SJ, Koelmans AA, Horne N, Unrine JM. Analysis of engineered nanomaterials in complex matrices (environment and biota): general considerations and conceptual case studies. Environmental Toxicology and Chemistry 2012;31(1):32-49. |
R833859 (Final) R834574 (2011) R834574 (2012) R834574 (Final) R834857 (2011) R834857 (2012) |
Exit Exit |
|
|
Yang S, Parks AN, Saba SA, Ferguson PL, Liu J. Photoluminescence from inner walls in double-walled carbon nanotubes: some do, some do not. Nano Letters 2011;11(10):4405-4410. |
R833859 (Final) |
Exit |
Supplemental Keywords:
Nanoparticles, nanomaterials, pollution, wastewater, bioavailability, analytical methods, mass spectrometry;, Health, Scientific Discipline, PHYSICAL ASPECTS, Health Risk Assessment, Physical Processes, Risk Assessments, Biochemistry, bioavailability, nanomaterials, carcinogenic, biological pathways, CNT, exposure, nanoparticle toxicity, nanotechnology, human health risk, toxicologic assessmentProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.