Grantee Research Project Results
2002 Progress Report: Bioaccumulation and Toxicity of Dietborne Particulate Metals to Benthic Invertebrates
EPA Grant Number: R829500C007Subproject: this is subproject number 007 , established and managed by the Center Director under grant R829500
(EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
Center: HSRC (1989) - Great Plains/Rocky Mountain HSRC
Center Director: Erickson, Larry E.
Title: Bioaccumulation and Toxicity of Dietborne Particulate Metals to Benthic Invertebrates
Investigators: Meyer, J. S.
Institution: University of Wyoming
EPA Project Officer: Aja, Hayley
Project Period: April 1, 2002 through March 31, 2005
Project Period Covered by this Report: April 1, 2002 through March 31,2003
RFA: Targeted Research Center (2002) Recipients Lists
Research Category: Hazardous Waste/Remediation , Targeted Research
Objective:
The objective of this research project is to investigate the ability of the Biotic Ligand Model (BLM) to predict the acute toxicity of Cu and Zn to fish when water quality parameters vary temporally. We will identify water quality parameters that vary diurnally in receiving waters, and conduct preliminary experiments needed to prepare for toxicity tests under time-varying exposure conditions.
Progress Summary:
We conducted acute toxicity tests with larval fathead minnows (FHM; Pimephales promelas) exposed to zinc (Zn) in the presence of elevated concentrations of Ca(NO3)2, CaCl2, Mg(NO3)2, MgCl2, and NaNO3. These tests demonstrate that the paired anion with which a major cation is added to an exposure water can affect the toxicity of Zn to larval FHM. This has important implications for testing the ability of the BLM to predict acute toxicity of metals to fish.
The results of these flow-through Zn toxicity tests suggested that either: (1) Ca2+ is not as protective as traditionally thought, and Mg2+ and Na+ interact synergistically with Zn; (2) NO3- interacts synergistically with Zn; or (3) NO3- interferes with the protective effects of Ca2+, Mg2+, and Na+. However, we could not exclude the possibility that the ACS Grade of these nitrate salts contained an unidentified contaminant, despite analyses of the Ag, Cd, Cu, Ni, Pb, and Zn contents of those salts in our laboratory that showed no concentrations of trace metals high enough to be of toxicological concern. Therefore, we conducted two separate sets of four side-by-side static-renewal Zn toxicity tests to determine if the purity of the added nitrate salt and/or the anion paired with the cation affected the Zn toxicity. The first set of four side-by-side tests was conducted in basewater, basewater containing elevated Ca(NO3)2 (ACS Grade), basewater containing elevated Ca(NO3)2 (ReagentPlus Grade—a higher purity than ACS Grade), and basewater containing elevated CaCl2 (ACS Grade). The second set of four side-by-side tests was conducted in basewater, basewater containing elevated Mg(NO3)2 (ACS Grade), basewater containing elevated Mg(NO3)2 (ReagentPlus Grade), and basewater containing elevated MgCl2 (ACS Grade).
The purity of the nitrate salts did not affect Zn toxicity for either Ca(NO3)2 or Mg(NO3)2. In fact, the 96-hour LC50s for the two purities of Ca(NO3)2 differed by only 2 percent; and the 96-hour LC50s for the two purities of Mg(NO3)2 differed by only 4 percent.
Ca2+ and Mg2+ differed, however, in their protective effects against Zn toxicity. When Ca2+ was added as 2.2 mM Ca(NO3)2, the 96-hour Zn LC50 was 2.4-fold greater than in the basewater; whereas when Mg2+ was added as 2.0 or 2.3 mM Mg(NO3)2, the 96-hour Zn LC50 did not differ from the LC50 in the basewater. When Ca2+ was added as 2.2 mM CaCl2, the 96-hour Zn LC50 was 4.3-fold greater than in the basewater; whereas when Mg2+ was added as 2.3 mM MgCl2, the 96-hour Zn LC50 was only 2.0-fold greater than in the basewater.
Moreover, the anion paired with the cation affected Zn toxicity. For Ca2+ and Mg2+, the 96-hour Zn LC50 almost doubled when the cation was paired with Cl- compared to NO3-. Unfortunately, based on these results alone, we cannot determine whether: (1) Cl- protects against Zn toxicity and NO3- has no effect; (2) NO3- interacts synergistically with Zn to increase the Zn toxicity and Cl- has no effect; or (3) some combination of the previous possibilities occurs. Direct toxicity of NO3- probably is not a contributor, because we observed no mortalities or outwardly apparent signs of stress in FHM larvae at 10 mM NaNO3 during a NaNO3 toxicity test that we conducted in November 2002. To test for indirect interactions of NO3- with Zn, we plan to conduct an analogous set of side-by-side static-renewal Zn toxicity tests in basewater, basewater containing elevated KNO3, and basewater containing elevated KCl—assuming that K+ neither protects against Zn toxicity nor interacts synergistically with Zn. in its review of the plans for the Metals Action Plan.
In February 2003, the Center held an international workshop on the Unit World Model Workshop at the University of Delaware to discuss technical and regulatory issues. Attendees included University researchers, U.S. EPA scientists and program managers, representatives from the metal industry, and many of the Center’s scientists.
Future Activities:
In the next year, the BLM will be tested using fish exposed to time-variable concentrations of copper and zinc.
Additional funding was appropriated for fiscal year 2002 that has allowed expansion of the research program in the future. The Science Advisory Committee (SAC) of the Center reviewed a series of proposals for research projects. Nine projects were approved for funding. Planning has been conducted with the SAC of the Center to allow the development of the Unit World Model (UWM) to be conducted in a 3-year program. The investigators met with the SAC in May 2002 to review project plans. The following are the projects that were approved.
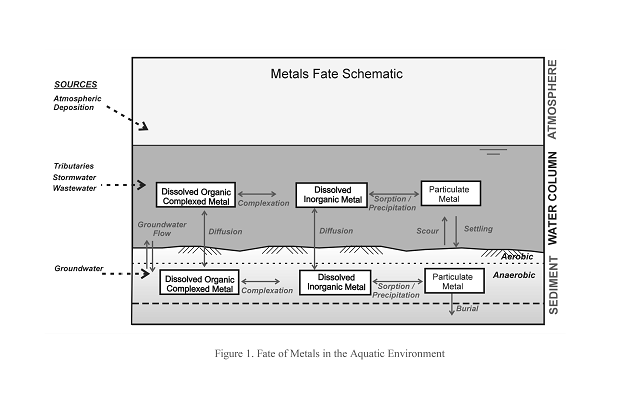
Most of the projects for Year 2 of the project are focused on a central theme related to the development of the UWM. This new modeling framework that will allow prediction of the distribution and, more importantly, the toxicity of metals in aquatic systems, is shown in Figure 1. The lack of a model that accurately predicts metal distribution and toxicity significantly hampers the implementation of scientifically credible total maximum daily loads and other important U.S. Environmental Protection Agency (EPA) programs. The importance of the technical factors being incorporated in the UWM has been pointed out by the U.S. EPA Science Advisory Board in its review of the plans for the Metals Action Plan.
Journal Articles on this Report : 3 Displayed | Download in RIS Format
| Other subproject views: | All 3 publications | 3 publications in selected types | All 3 journal articles |
|---|---|---|---|
| Other center views: | All 4 publications | 4 publications in selected types | All 4 journal articles |
| Type | Citation | ||
|---|---|---|---|
|
|
Field LJ, Macdonald DD, Norton SB, Ingersoll CG, Severn CG, Smorong D, Lindskoog R. Predicting amphipod toxicity from sediment chemistry using logistic regression models. Environmental Toxicology and Chemistry 2002;21(9):1993-2005. |
R829500 (2002) R829500C001 (2002) R829500C002 (2002) R829500C003 (2002) R829500C004 (2002) R829500C005 (2002) R829500C006 (2002) R829500C007 (2002) |
|
|
|
Paquin PR, Gorsuch JW, Apte S, Batley GE, Bowles KC, Campbell PGC, Delos CG, Di Toro DM, Dwyer RL, Galvez F, Gensemer RW, Goss GG, Hogstrand C, Janssen CR, McGeer JC, Naddy RB, Playle RC, Santore RC, Schneider U, Stubblefield WA, Wood CM, Wu KB. The biotic ligand model: a historical overview. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 2002;133(1-2):3-35. |
R829500 (2002) R829500C001 (2002) R829500C002 (2002) R829500C003 (2002) R829500C004 (2002) R829500C005 (2002) R829500C006 (2002) R829500C007 (2002) |
Exit Exit |
|
|
Santore RC, Mathew R, Paquin PR, DiToro DM. Application of the biotic ligand model to predicting zinc toxicity to rainbow trout, fathead minnow, and Daphnia magna. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 2002;133(1-2):271-285. |
R829500 (2002) R829500C001 (2002) R829500C002 (2002) R829500C003 (2002) R829500C004 (2002) R829500C005 (2002) R829500C006 (2002) R829500C007 (2002) |
Exit Exit |
Supplemental Keywords:
bioaccumulation, metals, toxicity, particulate metals, benthic invertebrates, water quality, water quality parameters, trace metals, basewater, Biotic Ligand Model, BLM, copper, Cu, zinc, Zn, fish, larval fathead minnows,, RFA, Scientific Discipline, INTERNATIONAL COOPERATION, Waste, Hazardous, Ecological Risk Assessment, Hazardous Waste, Geochemistry, Environmental Monitoring, dietary exposure, mobility of contaminants, metal contamination, fate and transport , mining wastes, remediation, bioaccumulation, extraction of metals, aquatic ecosystemsRelevant Websites:
http://www.ce.udel.edu/CSME/Index.html Exit
Progress and Final Reports:
Original AbstractMain Center Abstract and Reports:
R829500 HSRC (1989) - Great Plains/Rocky Mountain HSRC Subprojects under this Center: (EPA does not fund or establish subprojects; EPA awards and manages the overall grant for this center).
R829500C001 Role of Dietary Exposure for Bioaccumulation and Toxicity of Metals in Aquatic Ecosystems Affected by Mining
R829500C002 The Role of Organic Matter and Metal Oxides in the Retention of Trace Metals by Soil and Suspended Particles
R829500C003 Developing a Model to Predict the Persistence of Metals in Aquatic Environments
R829500C004 Effects of Dietary Metal Exposure on Fish and Aquatic Invertebrates
R829500C005 Aquatic Toxicity and Exposure Assessment
R829500C006 Development of a Model to Predict the Bioavailability of Metals to Soil Invertebrates
R829500C007 Bioaccumulation and Toxicity of Dietborne Particulate Metals to Benthic Invertebrates
The perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
3 journal articles for this subproject
Main Center: R829500
4 publications for this center
4 journal articles for this center