Grantee Research Project Results
Final Report: Fate and Transport of Carbon Nanomaterials in Unsaturated and Saturated Soils
EPA Grant Number: R832535Title: Fate and Transport of Carbon Nanomaterials in Unsaturated and Saturated Soils
Investigators: Pennell, Kurt D. , Abriola, Linda M. , Hughes, Joseph
Institution: Georgia Institute of Technology , Tufts University
EPA Project Officer: Hahn, Intaek
Project Period: October 1, 2005 through September 30, 2008
Project Amount: $395,300
RFA: Exploratory Research: Nanotechnology Research Grants Investigating Environmental and Human Health Effects of Manufactured Nanomaterials: A Joint Research Solicitation - EPA, NSF, NIOSH (2005) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
Widespread production and application of novel materials at the nanoscale is expected to increase dramatically over the next decade. Such an increase in production and dissemination will inevitably lead to the release of nanoscale compounds into the environment, either from industrial sources or through disposal in municipal waste streams. The current understanding of nanomaterial fate and transport in subsurface environments is quite limited. For example, it is not known how such compounds will interact with soil matrices, whether they can be modeled as colloidal particles using classic particle filtration theory, and how unsaturated soil conditions will impact transport and retention. The overall goal of this project is to expand our knowledge of carbon-based nanoparticle fate and transport in natural soils. Detailed laboratory experiments were conducted to explore the fate and transport of these engineered nanomaterials as a function of soil properties in one-dimensional systems. The research was specifically designed to couple detailed experimental studies with the development and evaluation of mathematical models. Thus, in addition to expanding our fundamental knowledge of nanomaterial fate and transport in the environment, one of the key benefits of this project was the development of a nanoparticle transport simulator. The project was structured around the three tasks, which are summarized below:
Task 1. Investigate the fate and transport of carbon-based nanomaterials in water-saturated soils. The effects of flow velocity, grain size, ionic strength and species on nanoscale fullerene (nC60) aggregate and multi-walled nanotube (MWNT) transport and retention behavior was evaluated in one-dimensional columns packed with quartz sands or natural soils.
Task 2. Investigate the effects of stabilizing agents on the transport of (C60) nanomaterials in porous media. The effects of natural (humic and fulvic acids) and anthropogenic (surfactants) stabilizing agents on the transport and retention of fullerene (nC60) aggregates in quartz sands was evaluated in a series of column experiments.
Task 3. Develop and evaluate a numerical simulator to describe C60 nanomaterial transport, retention and release in porous media. Mathematical models were developed and implemented in a one dimensional transport simulator, to predict nanomaterial transport and retention in the experimental systems. The numerical simulator was validated and refined based on comparisons between model predictions and experimental data.
Summary/Accomplishments (Outputs/Outcomes):
Task 1: Research activities initially focused on experimental measurement of nanoscale fullerene (nC60) aggregate transport and retention in water-saturated quartz sands as a function of flow velocity, grain size, ionic strength, and ionic species. For all experiments, aqueous suspensions of nC60 aggregates were prepared from fullerene powder that was dissolved in tetrahydrofuran (THF). The C60-saturated THF solution was mixed with an equal volume of water and evaporated at 75oC repeatedly to remove residual THF. The resulting aqueous stock suspension contained approximately 6 mg/L of nC60 aggregates in purified de-ionized (DI) water. The mean diameter of nC60 aggregates in this suspension was approximately 100 nm, determined by dynamic light scattering (DLS). To prepare aqueous suspensions for use in batch and column experiments, approximately 100 mL of the nC60 stock suspension were added to an equal volume of DI water containing either NaCl or CaCl2. The final concentration of background electrolyte in the aqueous nC60 suspensions ranged from 1 to 100 mM, which was adjusted to pH 7.0 with NaHCO3. For the transport experiments, glass columns (2.5 cm diam. X 10 or 15 cm length) were packed with either 20-30, 40-50, 80-100 or 100-140 mesh Ottawa sand and then completely saturated with water. Non-reactive tracer studies were performed to assess water flow characteristics and to determine the hydrodynamic dispersion coefficient. A pulse of nC60 suspension was then introduced into each column using a syringe pump, followed by nC60-free solution at the same flow rate. Effluent samples were collected continuously in 15 mL sterile tubes and analyzed for nC60 content by ultraviolet spectroscopy. At the conclusion of each transport experiment, the columns were dissected into 1.5 cm increments and extracted to obtain nC60 retention profiles.
As the mean grain size of Ottawa sand decreased from 0.71 to 0.13 mm, the time required for nC60 appearance in the column effluent increased, while the total amount of nC60 transported through the column decreased. Similarly, when the pore-water velocity was decreased from ca. 8 to 1 m/day, breakthrough of nC60 aggregates was delayed, and was only observed in 20-30 and 40-50 mesh Ottawa sand. Thus, depending upon the grain size and flow rate, either nearly complete breakthrough of nC60 aggregates, or conversely, nearly complete retention of nC60 aggregates was observed. In column experiments conducted with 40-50 mesh Ottawa sand and 1.0 mM CaCl2, nC60 aggregates appeared in the column effluent after approximately 1.4 pore volumes (PVs), and gradually increased to a maximum relative concentration of approximately 0.9 after introduction of 2.5 to 3.0 PVs of the nC60 suspension. Following reintroduction of nC60-free solution, the effluent concentration of nC60 declined sharply, approaching a value of zero at approximately 6.5 PVs. The asymmetric shape of the effluent breakthrough curves (BTCs) suggests that deposited nC60 aggregates were not readily released from the Ottawa sand. When either the ionic strength was increased from 3.05 to 30.05 mM or the sand grain size was decreased from 0.335 to 0.125 mm, virtually all of nC60 introduced into the column was retained. When NaCl was employed as the background electrolyte, much greater transport of nC60 aggregates was observed through Ottawa sand compared similar experimental conditions with CaCl2.
Derjaguin-Landau-Verwey-Overbeek (DLVO) theory was used to calculate electrostatic interaction energy profiles for quartz sand and nC60 aggregates as a function of electrolyte species and concentration. The resulting interaction energy profile for CaCl2 at the lower ionic strength (3.05 mM) indicates a net repulsive force near the sand surface, which should prevent deposition of nC60 aggregates. However, divalent cations are known to form complexes with negatively charged surface sites, thereby reducing the repulsive surface charge, which is not accounted for by DLVO theory. In addition, a small secondary attractive region (-0.5 kT) existed approximately 33 nm from the surface, which could also contribute to nC60 aggregate retention. At the higher ionic strength (30.05 mM), a primary attractive force existed near the surface, consistent with the strong tendency for nC60 aggregates to be retained by Ottawa sand. In the presence of NaCl at 3.05 mM, a large primary repulsive force (45 kT) existed near the surface, with a small secondary minimum (-0.25 kT) approximately 38 nm from surface. This repulsive force is consistent with the minimal retention of nC60 aggregate observed in column experiments conducted with NaCl. At the higher ionic strength (30.05 mM), the primary repulsive force was reduced to approximately 25 kT, while the secondary attractive region became stronger (-9 kT), consistent the greater retention of nC60 aggregates.
To further investigate the aggregate detachment process, a series of recovery experiments were conducted in which nC60 aggregates were deposited in columns packed with 100-140 mesh Ottawa sand with 1.0 mM CaCl2 as the background electrolyte. This was followed by changes in flow rate, ionic strength or solution pH, addition of cosolvent or surfactant. Approximately 1% of the deposited nC60 aggregates were removed when the column was flushed with DI water (IS ≈ 0) or 50% ethanol, while doubling the flow rate or flushing with 0.1% Tween 80 resulted in negligible recovery of deposited nC60 aggregates. In contrast, injection of DI water (3 PV) adjusted to pH 10.0 and 12.0 resulted in substantial increases in nC60 effluent concentration, corresponding to the recovery of 17.2 and 27.6% of deposited mass, respectively. The effect of tube length (0.5-2, 10-30, and 100 um) on MWNT transport in Ottawa sand was evaluated in separate column studies. At the length of 0.5-2 um, MWNT transport mimicked the behavior of a non-reactive tracer. When the length was increased to either 10-30 um or 100 um, however, retention of MWNTs in sand increased to ca. 17% and 40% of total input mass, respectively.
To investigate the transport and retention of nC60 under more realistic conditions, column studies were conducted with Webster soil, a silty clay loam (3.3% OC) and Appling soil, a loamy coarse sand (0.75% OC). When a pulse (100 mL) of nC60 suspension was introduced, the spatial distribution of nC60 retained in Appling and Webster soils was similar, with a maximum concentration of 40 µg/g near the column inlet. As the amount of nC60 suspension applied to Appling soil was increased to 32.5 and 65 PVs, the nC60 retentions profile exhibited a peak concentration of ca. 110 and 130 μg/g, respectively. The existence of concentration plateau and the increasing penetration of nC60 with increasing number of PVs applied suggest that Appling soil possesses a maximum nC60 retention capacity of ca. 140 μg/g under these conditions.
Task 2: A series of column studies was conducted to investigate the effects of stabilizing agents on nC60 transport and retention behavior. Influent nC60 suspensions were prepared by mixing with concentrated stock solutions containing either polyoxyethylene (20) sorbitan monooleate (Tween 80), Suwannee River humic acid (SRHA) or Suwannee River fulvic acid (SRFA) to achieve the desired final concentration of Tween 80 (1,000 mg/L), SRHA (20 mg C/L) or SRFA (20 mg C/L). Due to concerns over the potential effects of residual THF on nC60 transport, an additional influent nC60 suspension was prepared as described above, followed by spiking with 10 mg of THF to yield a THF concentration of ca. 50 mg/L.
In the presence of SRHA or SRFA, breakthrough of nC60 in the column effluent occurred at approximately 1 PV, earlier than observed in similar nC60-alone experiments. Once nC60 appeared in the column effluent, relative concentrations increased rapidly to a plateau value greater than 0.98, followed by sharp descent, with relative concentrations approaching zero at the end of pulse injection. The amount of nC60 retained in the columns was less than 2.9% of the injected mass, which is approximately one tenth of the nC60 mass retained in absence of either SRHA or SRFA. Retention of nC60 decreased gradually along the travel distance, with the maximum solid phase concentration of 0.18 µg/g near the column inlet.
When the nC60 suspension was premixed with Tween 80, nC60 BTCs were identical to those obtained with a non-reactive tracer (bromide), with breakthrough at approximately 1 PV followed by a rapid increase to a relative concentration (C/C0) plateau value of 1.0. When nC60 was introduced into an Ottawa sand column that had been pre-treated with Tween 80, the relative concentration of nC60 reached a maximum value of 0.9. In both the nC60-Tween 80 mixed and pretreated experiments, a similar sharp ascent and descent in the nC60 BTCs was observed. No measurable amounts of retained nC60 were observed when 1,000 mg/L Tween 80 was mixed with the nC60 input suspensions. In contrast, when a surfactant-free nC60 suspension was delivered to columns pretreated with Tween 80, 6.3 and 7.1% of the introduced nC60 mass was retained in the columns. While a large fraction of the introduced nC60 mass was transported through Tween 80-pretreated columns (>93%), retention of nC60 near the column inlet was substantial, with more than 77% of total retained nC60 mass occurring in the first 5 cm from column inlet.
Task 3: Over the initial two years of the project, a mathematical model was developed to describe the transport of nC60 aggregates through water-saturated porous media using a modified form of the advection-dispersion reactive (ADR) transport equation. Assuming conditions of homogeneity, laminar flow, and the absence of particle-particle interactions on the surface, the one-dimensional ADR transport equation for a stable nC60 suspension can be written as:
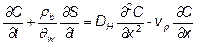
(1)
where, C is the nC60 aggregate concentration in the aqueous phase, t is time, ρb is the solid bulk density, θw is the volumetric water content, S is the attached or solid-phase concentration of nC60, DH is the hydrodynamic dispersion coefficient, x is the travel distance, and vp is the interstitial or pore-water velocity. Following deposition, nC60 aggregates may remain attached to the surface or may detach and re-enter the mobile aqueous phase. A first-order kinetic expression was used to describe the attachment and detachment of nC60 aggregates in water-saturated quartz sands:
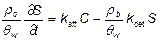
(2)
where, katt and kdet are the first-order attachment and detachment rate coefficients, respectively.
Results obtained from our early column studies suggested that the release of nC60 was negligible, and therefore, the detachment rate coefficient, kdet, was assumed to approach zero. In addition, the relatively constant or flat nC60 retention profiles observed in those column studies suggested that nC60 aggregate attachment approached a limiting or maximum value. Thus, equation 2 was rewritten as:

(3)
where k’att is the attachment rate coefficient at low surface coverage and Ψ is a blocking function that is related to the maximum particle attachment capacity Smax:
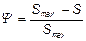
(4)
A numerical simulator, based upon the mathematical model described by (1), (3) and (4), successfully captured the initial gradual increase and subsequent sharp decline observed in measured nC60 BTCs and relatively flat retention profiles observed for different size fractions of Ottawa sand and background electrolyte conditions. In marked contrast, a model based on the traditional filtration theory (equation (2)) failed to accurately fit either the nC60 effluent BTCs or retention profiles. Both the maximum retention capacity (Smax) and the attachment rate (katt) were found to decrease with increasing sand grain size. For example, the value of Smax decreased from 13.8 ug/g sand to 0.8 ug /g sand as the mean grain size increased from 0.125 mm to 0.71 mm at a flow velocity of 8.0 m/day, and from 7.9 ug/g sand to 0.5 ug/g sand at the slower flow velocity (1.0 m/day). Also, for a particular size fraction of Ottawa sand, Smax increased with a decrease in flow velocity, consistent with the observed delay in initial breakthrough at the lower flow rate. For example, in columns packed with 40-50 mesh Ottawa sand, initial breakthrough occurred at ca. 1.3 PVs at pore-water velocity of 7.88 m/d; whereas, nC60 breakthrough did not occur until ca. 4.4 PVs at a velocity of 1.03 m/d.
In the last two years of the project, mathematical modeling efforts focused on describing the transport and retention of nC60 in the presence of stabilizing agents (i.e., SRHA and Tween 80). The hyper-exponential decay profile obtained for nC60 has been reported in colloid transport studies and attributed to a variable particle attachment rate (katt) along the travel distance. Therefore, a lognormal distribution of katt was selected and coupled to the clean bed filtration model. Although predictions from this model matched observations of both the measured BTCs and retention profiles, the selection of a distribution function for the attachment rate is arbitrary. Careful evaluation of the measured retention profiles revealed that the maximum nC60 concentration retained by the solid phase was consistent with the observed retention capacity, indicating that the maximum retention capacity of nC60 had been reached near the column inlet. Thus, we hypothesized that the observed hyper-exponential decay retention profile obtained for nC60 for the Tween 80-coated Ottawa sands resulted from an increase in the available surface area due to desorption of Tween 80. Desorbed Tween 80 in water may then adsorb onto nC60 aggregate surfaces, preventing nC60 from further attachment. Thus, a mathematical model was developed which coupled Tween 80 reactive (adsorption-desorption) transport with the deposition of nC60 on available surfaces. Consistent with previous laboratory studies, the model assumed that Tween 80 sorption conforms to a Langmuir isotherm model. In applying this system of equations, the value of DH was obtained from fitting the non-reactive tracer BTCs, katt and S2max were obtained from prior modeling efforts, and the Langmuir parameters were obtained from batch isotherms for Tween 80 adsorption to Ottawa sand. Predictions obtained using the new model demonstrated the ability of the model to capture the gradual decrease in the maximum concentration of nC60 and the hyper-exponential decrease in nC60 retention with distance from the column inlet.
Conclusions:
- In water-saturated quartz sands, nC60 aggregate transport decreases and retention increases as mean grain size and flow rate decrease.
- Transport and retention of nC60 aggregates in water-saturated porous media is strongly influenced by electrolyte species and concentration. At low ionic strength (1 mM CaCl2), nC60 aggregates are readily transported through quartz sands, while at higher ionic strength (10-100 mM CaCl2) nC60 aggregates are strongly retained by quartz sands. This effect may be due to bridging to adsorbed Ca2+ ions in the primary energy minimum.
- Once deposited, nC60 aggregate retention is largely irreversible, with minimal detachment observed even after flushing with low ionic strength solutions (DI water) or solutions containing surfactant (1% Tween 80) or cosolvent (50% ethanol). Introduction of pH 12 water was required to detach substantial quantities of retained nC60 aggregates.
- A mathematical model that incorporates a rate-limited attachment coefficient and a limiting retention capacity term was able to accurately simulate both nC60 transport and deposition in water-saturated porous media.
- Mixing nC60 suspensions with stabilizing agents, including Tween 80, SRHA and SRFA, enhanced the transport, and reduced the deposition, of nC60 aggregates in water-saturated 40-50 mesh Ottawa sand.
- When 40-50 mesh Ottawa sand was pretreated (coated) with Tween 80, adsorption of Tween 80 onto quartz sand surfaces reduced the retention and facilitated the transport of nC60 aggregates.
- A mathematical model that simultaneously accounts for the adsorption-desorption of Tween 80 and nC60 attachment was able to accurately predict nC60 transport behavior in the presence of Tween 80.
- In two natural soils, Appling and Webster, containing appreciable amounts of clay minerals and organic matter, complete retention of nC60 was observed even after introducing up to 65 PVs of nC60 suspension. The Smax nanoparticle transport model was able to capture this behavior.
- Dispersed MWNTs were readily transport through 40-50 mesh Ottawa sand. Retention of MWNTs increased with the tube length, however, even at the manufacture reported tube length of 100 µm, more than 50% of MWNTs passed through the column.
Journal Articles on this Report : 5 Displayed | Download in RIS Format
| Other project views: | All 22 publications | 7 publications in selected types | All 5 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Li Y, Wang Y, Pennell KD, Abriola LM. Investigation of the transport and deposition of fullerene (C60) nanoparticles in quartz sands under varying flow conditions. Environmental Science & Technology 2008;42(19):7174-7180. |
R832535 (Final) |
Exit |
|
|
Wang Y, Li Y, Fortner JD, Hughes JB, Abriola LM, Pennell KD. Transport and retention of nanoscale C60 aggregates in water-saturated porous media. Environmental Science & Technology 2008;42(10):3588-3594. |
R832535 (Final) |
Exit |
|
|
Wang Y, Li Y, Pennell KD. Influence of electrolyte species and concentration on the aggregation and transport of fullerene nanoparticles in quartz sands. Environmental Toxicology and Chemistry 2008;27(9):1860-1867. |
R832535 (Final) |
Exit |
|
|
Wang Y, Li Y, Kim H, Walker SL, Abriola LM, Pennell KD. Transport and retention of fullerene nanoparticles in natural soils. Journal of Environmental Quality 2010;39(6):1925-1933. |
R832535 (Final) |
Exit Exit |
|
|
Wang Y, Kim J-H, Baek J-B, Miller GW, Pennell KD. Transport behavior of functionalized multi-wall carbon nanotubes in water-saturated quartz sand as a function of tube length. Water Research 2012;46(14):4521-4531. |
R832535 (Final) |
Exit Exit Exit |
Supplemental Keywords:
groundwater, emerging contaminants, Health, Scientific Discipline, ENVIRONMENTAL MANAGEMENT, Health Risk Assessment, Risk Assessments, Biochemistry, Risk Assessment, bioavailability, nanomaterials, soil pollution, ecological risk assessment, contaminated sediments, nanochemistry, nanotechnology, fate and transport, carbon fullerene, ecotoxicogenomics, biochemical researchRelevant Websites:
Integrated Multipurpose Environmental Systems Laboratory Exit
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
