Grantee Research Project Results
Final Report: Secondary Aerosol Formation from Gas and Particle Phase Reactions of Aromatic Hydrocarbons
EPA Grant Number: R831084Title: Secondary Aerosol Formation from Gas and Particle Phase Reactions of Aromatic Hydrocarbons
Investigators: Kamens, Richard M.
Institution: University of North Carolina at Chapel Hill
EPA Project Officer: Chung, Serena
Project Period: July 28, 2003 through July 27, 2006 (Extended to July 27, 2008)
Project Amount: $400,000
RFA: Measurement, Modeling, and Analysis Methods for Airborne Carbonaceous Fine Particulate Matter (PM2.5) (2003) RFA Text | Recipients Lists
Research Category: Particulate Matter , Air Quality and Air Toxics , Air
Objective:
This project focuses on the elucidation of the fundamental chemistry that brings about the secondary organic aerosol formation (SOA) from aromatic reactions in the atmosphere. The overall goal is to develop a “new generation” aromatic chemical mechanism that can integrate newly discovered particle phase heterogeneous processes with the known gas phase chemistry, as a unified, multi-phase, chemical reaction mechanism, which will ultimately permit the prediction of SOA formation in the aromatic system.Summary/Accomplishments (Outputs/Outcomes):
Model description, key assumptions: The toluene mechanism developed under this project was constructed to explicitly represent the formation of first generation gas-phase products from the photooxidation of toluene and the further reactions of these products with atmospheric oxidants. We followed the general approach outlined by Calvert et al. (2002) in their review of aromatic atmospheric chemistry. First generation products are defined as those resulting from initial oxidation of toluene by the hydroxyl (OH) or nitrate (NO3) radical, and include 1,4-butenedial, 4-oxo-2-pentenal, 2-methylbutenedial, 2-methyl-2,4-hexadienedial, methyl glyoxal, glyoxal, cresol and benzaldehyde. A simple illustration for the formation of 1st generation toluene gas phase products is shown in Figure 1. Semi-explicit mechanisms for these 1st generation products were developed and evaluated against outdoor chamber data for 1,4-butenedial, 4-oxo-2-pentenal and cresol (Liu et al., 1999; Johnson et al., 2004). Many of the second generation products from the photo-degradation of these initial products have relatively low vapor pressures (10-2 to 10-7 torr), and thus can partition between the gas and aerosol phases. A kinetic partitioning approach suggested by Kamens et al. (1999; 2001) was used to describe the time-dependent phase distribution of about 70 compounds during the reaction. Of significance is that selected heterogeneous reactions of semi-volatile carbonyl compounds that lead to the formation of large molecules have also been represented either via calculations of uptake coefficients or by reductions in desorption rate coefficients. This will be discussed in more details later. Rate constants were developed by reference to the available literature (Calvert et al., 2002; Liggio et al., 2005b) and experiment data. Where mechanistic or rate constant information were not available, a structure activity relationship (SAR) technique was applied to calculate the gas-phase kinetic parameters (Kwok and Atkinson, 1995), or the mechanistic pathways and rate coefficients from similar compound structures were used (Jenkin et al., 1997; Saunders et al., 2003). The detailed mechanism is available in the supplemental material in Hu and Kamens et al (2007) . In addition to the toluene mechanism, a Carbon Bond 4 (2002) mechanism is also included which incorporates the reactions of inorganic species (Voicu, 2003). A chamber dependent auxiliary wall mechanism that describes the background radical sources and sinks of NOx was also used (Jeffries et al., 1999; Voicu, 2003).
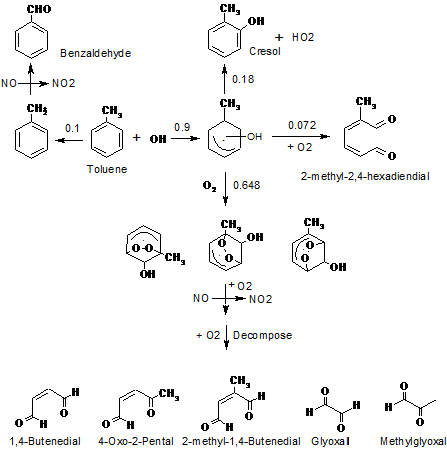
Figure 1. Illustration for the formation of 1st generation toluene gas phase products
|
|
Initial Concentration (ppm)
|
|
|
|
||
|
Experiment Date
|
[Tol]0
|
[NO]0
|
[NO2]0
|
[Tol]0/[NO]0
|
Temp (K)
|
Dew Point (K)
|
|
052204Sa
|
0.992
|
0.445
|
0.088
|
2.23
|
303-314
|
294-295
|
|
092304Nb
|
0.500
|
0.204
|
0.014
|
2.45
|
296-307
|
292-294
|
|
092304S
|
0.980
|
0.280
|
0.026
|
3.50
|
296-307
|
291-295
|
|
101604N
|
1.023
|
0.096
|
0.014
|
10.66
|
288-300
|
277-282
|
|
101604S
|
0.540
|
0.370
|
0.027
|
1.46
|
288-300
|
277-282
|
|
111504N
|
0.864
|
0.272
|
0.055
|
3.18
|
284-294
|
274-276
|
|
072705N
|
0.186
|
0.091
|
0.039
|
2.04
|
300-310
|
276-281
|
|
072705S
|
0.095
|
0.091
|
0.042
|
1.04
|
300-310
|
278-283
|
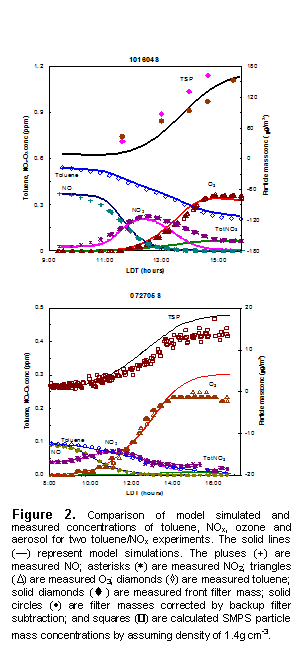 suggests that there may be other reaction pathways that can also remove excess NO2 in the afternoon in the toluene/NOx system.
suggests that there may be other reaction pathways that can also remove excess NO2 in the afternoon in the toluene/NOx system.|
Namea
|
Structure
|
Namea
|
Structure
|
|
GLYPOLY
|
|
RgDOHNO3
|
 |
|
|
|
RgTOHNO3
|
 |
|
MGLYPOLY
|
|
ROHALDNO3
|
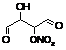 |
|
|
|
|
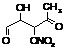 |
|
RgOHDINO2
|
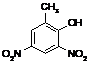 |
|
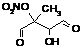 |
|
RgDIOHNO2
|
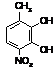 |
ROHALDACIDNO3
|
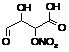 |
|
RgOHNO2
|
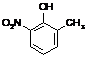 |
|
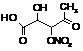 |
|
CROHOOH
|
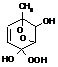 |
|
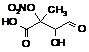 |
|
RgTOHNO2OOH
|
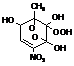 |
BUDACIDONO3
|
|
|
TOLOOH
|
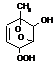 |
MBUDACIDONO3
|
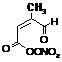 |
|
SEED1
|
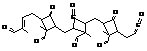 |
C6OHNO2ACID
|
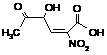 |
Conclusions:
Accomplishments:
Over the course of this project we developed a chemical mechanism for toluene which reasonably predicts the gas phase chemistry and also predicts the SOA formation from toluene oxidation (Figure 1). Two papers have appeared in the peer reviewed literature on this mechanism. Our model includes aerosol phase chemistry that includes nucleation, gas-particle partitioning and particle phase reactions as well as the gas-phase chemistry of toluene and its degradation products are represented. A series of experiments that cover a wide range of temperature, solar condition and initial reactant concentrations, were carried out in the UNC 270 m3 dual outdoor aerosol smog chambers. Data obtained from these experiments were used to develop and test the mechanism. The model adequately simulates the decay of toluene, the NO to NO2 conversion, and ozone formation. Although it provides a reasonable prediction of SOA production under different conditions that range from 15 to 300 mg m-3, the model tends to underestimate the initial particle burst for almost all high toluene concentration experiments.
The main contribution that this study makes to science is that describes an atmospheric aromatic mechanism that: 1. simultaneously considers gas phase reactions, trace gas phase-particle phase partitioning, and subsequent particle phase reactions; 2. it proposes a simple chemical mechanism for particle phase nucleation; 3. permits one to distinguish between gas-particle partitioning of SOA, and heterogeneous SOA formation; 4. demonstrates the relative importance of organic nitrate formation at high and low toluene concentrations; and 5. successfully simulates gas phase toluene oxidation in smog chamber systems. To date, this has not been accomplished by any of the existing aromatic mechanisms.
The dominant particle phase species predicted by the mechanism are glyoxal oligomers (organic nitrates, methyl nitro-phenol analogues, C7 organic peroxides, acylperoxy nitrates and, for the low concentration experiments, unsaturated hydroxyl nitro-acids. The relative amounts of these products vary depending on initial experimental conditions. In general, with decreasing toluene/NO ratios, the relative amount of total organic nitrates and acylperoxy nitrates in the particle phase increases, the mass fraction of total oligomers and organic peroxides decreases. It is also important to note that the relative amount of different SOA species dramatically changes with time. The model also well predicts the SOA mass concentrations observed from the European Photoreactor (EUPHORE) and smog chambers at the California Institute of Technology (Caltech). But to implement the developed mechanism into the regional airshed model, it would be desirable to reduce the reaction steps and number of represented species. It is recommended that future studies should focus on: 1. reaction mechanisms that contribute to rapid particle formation in toluene/NOx system, 2. identification of particle phase oligomers, 3. measurement of organic nitrates and acylperoxy nitrates, 4. chamber experiments with reactant concentrations at ambient levels, and 5. combining aromatics and monoterpenes mechanisms into one unified mechanism.
References
Calvert, J.G., Atkinson, R., Becker, K.H., Kamens, R.M., Seinfeld, J.H., Wallington, T.J., and Yarwood, G., 2002. The Mechanisms of Atmospheric Oxidation of Aromatic Hydrocarbons. Oxford University Press, Inc., Glasstone, S., Laidler, K. J., and Eyring, H., 1941. The Theory of Rate Processes: The Kinetics of Chemical reactions, Viscosity, Diffusion and Electrochemical Phenomena.
Jang, M. and Kamens, R. M., 1998. A Thermodynamic Approach for Modeling Partitioning of Semivolatile Organic Compounds on Atmospheric Particulate Matter: Humidity Effects. Environmental Science and Technology 32(9), 1237-1243.
Jang, M. and Kamens, R. M., 2001a. Atmospheric Secondary Aerosol Formation by Heterogeneous Reactions of Aldehydes in the Presence of a Sulfuric Acid Aerosol Catalyst. Environmental Science and Technology 35(24), 4758-4766.
Jang, M. and Kamens, R. M., 2001b. Characterization of Secondary Aerosol from the Photooxidation of Toluene in the Presence of NOx and 1-Propene. Environmental Science and Technology 35(18), 3626-3639.
Jang, M., Czoschke, N. M., Lee, S., and Kamens, R. M., 2002. Heterogeneous Atmospheric Aerosol Production by Acid- Catalyzed Particle-Phase Reactions. Science (
Jang, M., Lee, S., and Kamens, R. M., 2003. Organic aerosol growth by acid-catalyzed heterogeneous reactions of octanal in a flow reactor. Atmospheric Environment 37(15), 2125-2138.
Jeffries, H., Sexton, K., and Adelman, Z., 1999. Auxiliary mechanisms (wall models) for UNC outdoor chamber. EPA/600/R-00/076.
Jeffries, H., Kessler, M., and Gery, M., 2003. MComp/MEval: The Morphecule Photochemical Reaction Mechanism.
Joback, K.G. and Reid, R.C., 1987. Estimation of pure-component properties from group contributions. Chemical engineering Communications 57, 233-243. Johnson, D.,
Kalberer, M., Paulsen, D., Sax, M., Steinbacher, M., dommen, J., Prevot, A.S.H., Fisseha, R., Weingartner, E., Frankevich, V., Zenobi, R., and Baltensperger, U., 2004. Identification of polymers as major components of atmospheric organic aerosols. Science 303(5664), 1659-1662.
Kamens, R. M. and Jaoui, M., 2001. Modeling aerosol formation from -Pinene + NOx in the presence of natural sunlight using gas-phase kinetics and gas-particle partitioning theory. Environmental Science and Technology 35(7), 1394-1405.
Kamens, R., Jang, M., Chien, C.-J., and Leach, K., 1999. Aerosol Formation from the Reaction of -Pinene and Ozone Using a Gas-Phase Kinetics-Aerosol Partitioning Model. Environmental Science and Technology 33(9), 1430-1438.
Klotz, B., Barnes,
Kroll, J.H., Ng, N.L., Murphy, S.M., Varutbangkul, V., Flagan, R.C., and Seinfeld, J.H., 2005. Chamber studies of secondary organic aerosol growth by reactive uptake of simple carbonyl compounds. Journal of Geophysics Research 110, D23207.
Kwok, E.S.C. and Atkinson, R., 1995. Estimation of hydroxyl radical reaction rate constants for gas-phase organic compounds using a structure-reactivity relationship: an update. Atmospheric Environment 29, 1685-1695.
Kwok, E.S.C., Aschmann, S.M., Atkinson, R., and Arey, J., 1997. Products of the gas-phase reactions of o-, m-, and p-xylene with the OH radical in the presence and absence of NOx. Journal of the Chemical Society, Faraday Transactions 93, 2847-2854.
Leungsakul, S., Jeffries, H. E., and Kamens, R. M., 2005a. A kinetic mechanism for predicting secondary aerosol formation from the reactions of d-limonene in the presence of oxides of nitrogen and natural sunlight. Atmospheric Environment 39(37), 7063-7082.
Leungsakul, S., Jaoui, M., and Kamens, R. M. , 2005b. Kinetic Mechanism for Predicting Secondary Organic Aerosol Formation from the Reaction of d-Limonene with Ozone. Environmental Science and Technology 39(24), 9583-9594.
Liggio, J., Li, S.-M., and McLaren, R., 2005a. Heterogeneous Reactions of Glyoxal on Particulate Matter: Identification of Acetals and Sulfate Esters. Environmental Science and Technology 39(6), 1532-1541.
Liggio, J., Li, S.-M., and McLaren, R., 2005b. Reactive uptake of glyoxal by particulate matter. Journal of Geophysical Research 110, D10304.
Liu, X., Jeffries, H. E., and Sexton, K. G., 1999. Atmospheric Photochemical Degradation of 1,4-Unsaturated Dicarbonyls. Environmental Science and Technology 33(23), 4212-4220.
Mackay, D.B.A., Chan, D.W., and Shiu, W.Y., 1982. Vapor pressure correlations for low-volatility environmental chemicals. Environmental Science and Technology 16, 645-649.
Matsumoto, M., Yasuoka, K., and kataoka, Y., 1994. Evaporation and condensation at a liquid surface. II. Methanol.
Nielsen, T., Platz, J., Offenberg, J.H., Kleindienst, T.E., Jaoui, M., Lewandowski, M., and Edney, E.O., 2006. Thermal Properties of Secondary Organic Aerosols. Geophysical Research Letters 33, L03816.
Pankow, J.F., 1994. An absorption model of gas/particle partitioning of organic comounds in the atmosphere. Atmospheric Environment 28, 185-188.
Saunders, S.M.,
Stein, S.E. and Brown, R.L., 1994. Esitimation of normal boiling points from group contributions. Journal of Chemical Information and Computer Sciences 34, 581-587.
Tolocka, M. P., Jang, M., Ginter, J. M., Cox, F. J., Kamens, R. M., and Johnston, M. V., 2004. Formation of Oligomers in Secondary Organic Aerosol. Environmental Science and Technology 38(5), 1428-1434.
Voicu,
Yasuoka, K., Matsumoto, M., and Kataoka, Y., 1994. Evaporation and condensation at a liquid surface. I. Argon. The Journal of Chemical Physics 101, 7904-7911.
Zhao, L., Li, P., and Yalkowsky, S.H., 1999. Predicting the Entropy of Boiling for Organic Compounds. Journal of Chemical Information and Computer Sciences 39, 1112-1116.
Journal Articles on this Report : 5 Displayed | Download in RIS Format
| Other project views: | All 8 publications | 6 publications in selected types | All 6 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Hu D, Tolocka M, Li Q, Kamens RM. A kinetic mechanism for predicting secondary organic aerosol formation from toluene oxidation in the presence of NOx and natural sunlight. Atmospheric Environment 2007;41(31):6478-6496. |
R831084 (2006) R831084 (2007) R831084 (Final) |
Exit Exit Exit |
|
|
Hu D, Kamens RM. Evaluation of the UNC toluene-SOA mechanism with respect to other chamber studies and key model parameters. Atmospheric Environment 2007;41(31):6465-6477. |
R831084 (Final) |
Exit Exit Exit |
|
|
Lee S, Kamens RM. Particle nucleation from the reaction of α-pinene and O3. Atmospheric Environment 2005;39(36):6822-6832. |
R831084 (2005) R831084 (2006) R831084 (2007) R831084 (Final) |
Exit Exit Exit |
|
|
Leungsakul S, Jeffries HE, Kamens RM. A kinetic mechanism for predicting secondary aerosol formation from the reactions of d-limonene in the presence of oxides of nitrogen and natural sunlight. Atmospheric Environment 2005;39(37):7063-7082. |
R831084 (2005) R831084 (2006) R831084 (2007) R831084 (Final) R828176 (Final) |
Exit Exit Exit |
|
|
Li Q, Hu D, Leungsakul S, Kamens RM. Large outdoor chamber experiments and computer simulations: (I) secondary organic aerosol formation from the oxidation of a mixture of d-limonene and α-pinene. Atmospheric Environment 2007;41(40):9341-9352. |
R831084 (2007) R831084 (Final) |
Exit Exit Exit |
Supplemental Keywords:
Secondary organic aerosol formation, aromatics, modeling, organic particle formation
, RFA, Scientific Discipline, Ecosystem Protection/Environmental Exposure & Risk, Environmental Chemistry, Monitoring/Modeling, Atmospheric Sciences, Environmental Monitoring, air quality, secondary organic aerosol, particle phase reactions, gas phase chemistry, environmental measurement, Toluene, organic chemistry, aerosol analyzersRelevant Websites:
Progress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
Project Research Results
- 2007 Progress Report
- 2006 Progress Report
- 2005 Progress Report
- 2004 Progress Report
- Original Abstract
6 journal articles for this project
