Grantee Research Project Results
2009 Progress Report: Compound Specific Imprinted Nanospheres for Optical Sensing
EPA Grant Number: R830911Title: Compound Specific Imprinted Nanospheres for Optical Sensing
Investigators: Lavine, Barry K. , Seitz, William Rudolf , Fendler, Janos
Institution: Oklahoma State University , Clarkson University , University of New Hampshire
EPA Project Officer: Hahn, Intaek
Project Period: August 24, 2003 through August 23, 2006 (Extended to August 31, 2010)
Project Period Covered by this Report: August 24, 2008 through August 23,2009
Project Amount: $323,000
RFA: Environmental Futures Research in Nanoscale Science Engineering and Technology (2002) RFA Text | Recipients Lists
Research Category: Nanotechnology , Safer Chemicals
Objective:
The objective of this research project is to investigate the use of molecularly imprinted polymers as the basis of a sensitive and selective method for the detection of pharmaceuticals and other emerging organic contaminants at parts per billion (ppb) levels in aquatic environments. Moderately cross-linked, molecularly imprinted polymeric nanospheres (ranging from 100 nm to 1,000 nm in diameter), which are designed to swell and shrink as a function of analyte concentration in aqueous media, are being prepared. The nanospheres are spin coated onto a gold substrate. Chemical sensing is based on changes in the optical properties of the spin coated particles that accompany swelling of the molecularly imprinted nanospheres. Two effects contribute to this change. One is an increase in the size of the nanospheres, resulting in an increase in the angle of minimum reflectance. The other is a change in refractive index. Because swelling leads to an increase in the percentage of water in the polymer, the refractive index decreases as the nanospheres swell. This brings them closer to the refractive index of water. For the systems that we are studying, the change in refractive index is the dominant effect. This change can be measured by a quartz crystal microbalance (QCM) or surface plasmon resonance (SPR) spectroscopy. Using SPR, our prototype sensor is capable of detecting pollutants and hazardous materials selectively at ppb levels in the environment.Progress Summary:
Theophylline-imprinted polymer particles (approximately 0.3 microns in diameter) suitable for SPR have been prepared by dispersion polymerization at 650C and deposited onto a gold SPR slide by spin coating a methanol suspension of the particles onto a gold SPR slide. Ten drops of the methanol suspension are placed in the center of the sample slide, which is spun at 500 rpm for 5 seconds and 3000 rpm for 25 seconds. Two or three additional applications of two drops of the polymer suspension were added to build a nearly uniform polymer layer as determined by visual inspection using a Leitz Orthoplane microscope. The slide is then allowed to dry for two days in a desiccator before rehydration. The particles are held on the slide by electrostatic attraction. The polymer particles form a layer that is both sensitive and specific. The addition of as little as 1.0x10-6 M theophylline is sufficient to cause a change in the refractive index, which can be detected by SPR. Higher concentrations of theophylline produced larger changes in the refractive index (see Figure 1). In contrast, the particles showed no response to distilled water or 1.0x10-2 M caffeine or theobromine. (Caffeine, theobromine, and theophylline differ by only a single methyl group.) The full-scale response of the imprinted particles to the template occurs in less than 10 minutes. Swelling is reversible, and replicate precision is less than 10-4 refractive index units. A unique aspect of the prepared particles is the use of light crosslinking rather than heavy cross-linking.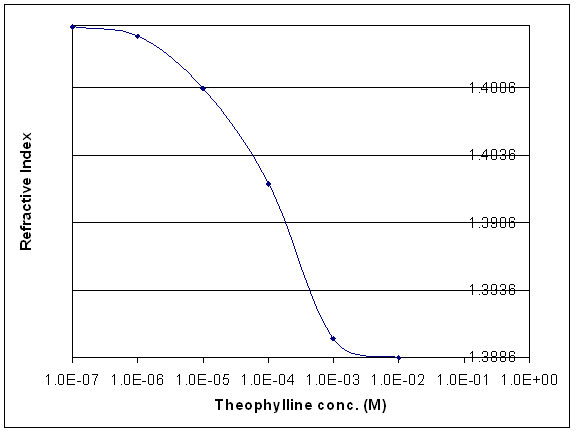
Figure 1. Refractive index of the spin coated imprinted polymer particles as a function of theophylline concentration. The refractive index values of the polymer particles were determined by fitting the SPR spectra to a five layer model: the first layer is the prism, the second layer is chromium (1 nm layer), the third layer is gold (50 nm layer), the fourth layer is the polyNNPA particles, and the fifth layer is the solution in contact with the polymer.
Swellable polymer particles that respond to pH have also been prepared by dispersion polymerization at ambient temperature using NIPA and 650C using NNPA. When these polymer particles are dispersed in a hydrogel, there are large changes in turbimetry (absorbance) as the pH of the solution in contact with the membrane is varied. Changes of approximately one-half of an absorbance unit have been observed in the physiological pH range at both ambient and physiological temperatures (see Figure 2). Swelling is relatively independent of ionic strength, it is nearly reversible, and the polymer particles show a larger response over a narrower pH range than predicted by the Henderson-Hasselbach equation. The pKa of the particles can be tuned by varying the degree of crosslinking or the pKa of the pH sensitive comonomer used. One potential application of the pH sensitive polymer particles developed in our research is monitoring the progress of open-heart surgery, where pH serves as a measure of tissue ischemia. Gastric pH sensing is yet another possible application. Monitoring the pH of rivers and streams is a third application.
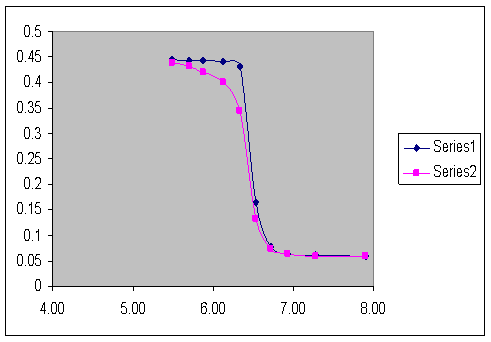
Figure 2. pH Titration Curve (0.1 M Ionic Strength) for M-120t Particles at 370C. The apparent pKa of the particles is 6.45.
Future Activities:
The plot of refractive index versus concentration of theophylline shown in Figure 1 suggests a detection limit of 10-6M for the template. Although the detection limit for the template is lower than what has been previously reported in the literature, our goal is to detect theophylline at 4 ppb, which corresponds to 2x10-8M, the detection limit for theophylline by solid phase extraction-liquid chromatography/mass spectrometry (SPE-LC/MS). To further reduce the detection limit by a factor of 50, it will be necessary to further improve the quality of our formulation. For this reason, we will obtain the components used in the formulation from Waco Chemicals. It is crucial for both the recognition monomer and crosslinker to be free of impurities. Our previous supplier, Aldrich, cannot provide us with monomers of the necessary purity. We will also continue to investigate the use of more hydrophobic recognition monomers, in particular ethacrylic acid. In aqueous media, hydrophobicity is an important force controlling the interactions between recognition sites in the polymer and the template, which is in the solution.Particle adhesion to the gold surface has been problematic, contributing to higher detection limits. The two-step spin coating procedure that has been developed in our laboratory has helped to obviate this problem. Recently, we have been able to place a polyvinyl alcohol hydrogel membrane on top of the spin coated particles, ensuring particle adhesion and improving the quality of the SPR spectra obtained by ensuring a more uniform polymer film distribution along the gold surface. We plan to further investigate this procedure during the final reporting period.
Experiments will also be undertaken to assess the advantages of using the quartz crystal microbalance (QCM) to monitor polymer swelling. Preliminary studies undertaken in our laboratory indicate that delamination of the polymer film with swelling and shrinking is a less serious problem. Sensitivity, however, is an open question that must be addressed to assess the viability of QCM.
Journal Articles on this Report : 4 Displayed | Download in RIS Format
| Other project views: | All 19 publications | 5 publications in selected types | All 5 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Lavine B, Mwangi G, Kim M. Characterization of Swellable Molecularly Imprinted Polymer Particles by Surface Plasmon Resonance Spectroscopy. APPLIED SPECTROCSOPY 2012;66(4) |
R830911 (2009) |
Exit |
|
|
Lavine B, Oxenford L, Kim M, Kaval N, Benjamin M, Seitz W. Novel turbidimetric method to study polymer swelling. MICROCHEMICAL JOURNAL 2012;103:97-104 |
R830911 (2009) |
Exit |
|
|
Metzler D, Erdem A, Huang C. Influence of Algae Age and Population on the Response to TiO2 Nanoparticles. INTERNATIONAL JOURNAL OF ENVIRONMENTAL RESEARCH AND PUBLIC HEALTH 2018;15(4):585. |
R830911 (2009) R831710 (Final) |
Exit Exit |
|
|
Stoian R, Lavine B, Rosenberger A. pH sensing using whispering gallery modes of a silica hollow bottle resonator. TALANTA 2019;194:585-590 |
R830911 (2009) |
Exit |
Supplemental Keywords:
Sustainable Industry/Business, RFA, Ecosystem Protection/Environmental Exposure & Risk, Scientific Discipline, Water, Technology for Sustainable Environment, Sustainable Environment, Environmental Chemistry, Engineering, Chemistry, & Physics, Monitoring/Modeling, Environmental Monitoring, membranes, membrane technology, environmental sustainability, nanospheres, aquatic ecosystem, chemical detection techniques, nanoporous membranes, nanotechnology, surface plasma resonance spectroscopy, nanosensors, analytical chemistry, hydrogel membranes, aquatic toxins, aqueous impurities, chemical sensorsProgress and Final Reports:
Original AbstractThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.
