Grantee Research Project Results
Final Report: Solving the Arsenic Problem in Rural California
EPA Grant Number: SU839960Title: Solving the Arsenic Problem in Rural California
Investigators: Gadgil, Ashok , Tseng, Winston , Kumar, Arkadeep , Nahata, Mohit , Hernandez, Dana , Bandaru, Siva , Duffy, Lucas , Majmudar, Jay
Institution: University of California - Berkeley
EPA Project Officer: Callan, Richard
Phase: I
Project Period: October 1, 2019 through September 30, 2020 (Extended to September 30, 2021)
Project Amount: $25,000
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2019) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
The goals of the project have not significantly changed from the original application, but some changes have been made in the experimental design. The experiments in objective 1 were not conducted in a continuous flow through system, but rather in a batch system. This change was due to the improved safety of working with a smaller amount of high concentrations of arsenic and a reactor less likely to leak. We also tested the arsenic removal performance and efficiency for a much higher initial As(III) concentration of 1,500 ppb, rather than 250 ppb, in order to demonstrate the high efficacy of the technology. Moreover, due to the complete shutdown of lab operation on March 14, 2020 due to the COVID-19 pandemic, and reopening with a major curtailment in mid August 2020, the flow through technology in objective 2 was not tested for a 6 month duration in the lab during this phase, but rather for a 2-week period during our no cost extension period.
Summary/Accomplishments (Outputs/Outcomes):
Objective 1: Demonstrate arsenic removal from very high initial concentrations of 1,500 ppb of As(III), which is the more difficult species of inorganic arsenic to remove, to below the EPA-MCL in a synthetic groundwater matrix mimicking the composition of contaminated aquifers in California.
We demonstrated that high initial concentrations of 1,500 ppb of As(III) are effectively removed in a synthetic groundwater matrix. For a total charge dosage of 600 C/L, or 3.1 mM of iron, the residual arsenic levels in ACAIE experiments were below 4 ppb for a large span of coulombic dosage rates (of 1.5, 6, 60, 100, and 600 C/L/min). These rates correspond to electrolysis times of 400 min to 1 min, respectively. Dosage rates of 100 and 600 C/L/min are considered high and make ACAIE (Air Cathode Assisted Iron Electrocoagulation) competitive in achieving desirably large throughputs of arsenic-safe drinking water. The samples were analyzed using ICP-OES and ICP-MS. This work has been published in January 2020 in Environmental Science & Technology(Bandaru et al., 2020). While conducting these arsenic removal experiments we also collected relevant voltage, current, conductivity and pH data to obtain realistic estimates on operating costs.
Long-term studies of electrode performance in the ACAIE reactor system are on-going and in progress. These studies will allow us to better quantify long-term cathodic and anodic performance for better operation and maintenance in the field. By measuring the faradaic efficiencies of Fe(II) and H 2O 2 production over time, we will be able to predict consumption of the iron anode, and its replacement schedule. With these studies, we are also examining air cathode degradation over time as it relates to surface fouling by iron oxides and other constituents present in the groundwater. Future work will focus on how to mitigate the negative side-effects of electrode (specifically, the air cathode) degradation. These studies will provide insight into how the larger system should be designed for lower maintenance costs and robust performance at field scale.
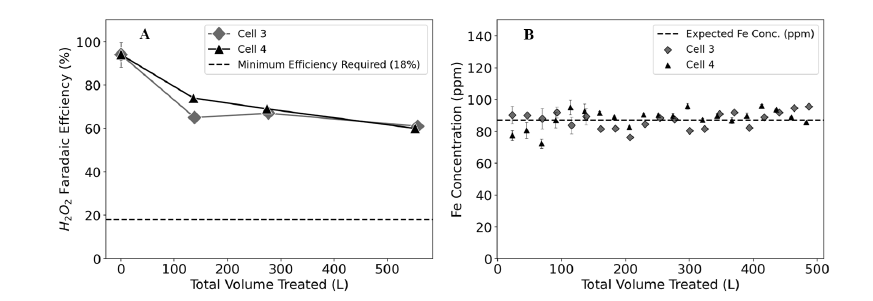
The approach for the long term studies of ACAIE performance currently involves two reactor cells (named “Cell 3” and “Cell 4” in Figures 2A and 2B legends), running in parallel under the same conditions. The operating conditions chosen for this study are summarized below, and consistent with published work (Glade et al., 2021) in terms of minimum charge dosage required to remove arsenic below EPA-MCL of 10 μg/L, while current density is nearly 10 times higher than what we expect to use in the field with the intention of reaching a failure point much sooner than we would in the field. The electrolyte composition used in these experiments is representative of the groundwater sampled in Allensworth, CA (Glade et al., 2021). Operating conditions of the test cells (reactor volume: 0.5 L, active surface area of cathode: 64 cm 2) are as follows– charge dosage: 300 C/L; charge dosage rate: 384 C/L/min; current density: 50 mA/cm2 .
Figure 1. Experimental set-up of benchscale flow-through, long-term testing of ACAIE cells
under high current density conditions.
Faradaic efficiency of H 2O 2 production is an important performance metric for the air cathodes as it dictates the chemical kinetics of Fe(II) oxidation and thus arsenic removal in the ACAIE system. Under these operating conditions, the concentration of H 2O 2 produced is expected to be around 1.55 mM. Cathode performance is monitored over time under these conditions by comparing the actual concentration of H2 O 2 produced with the expected value, and tracking that comparison over time. Figure 2A, below, shows how the faradaic efficiency of H2 O 2 production is reduced to about 60% after treating close to 600 liters of water using a single cathode 0.5 L ACAIE reactor cell under high current density conditions. The minimum faradaic efficiency of H2 O 2 production required for satisfactory ACAIE performance was estimated to be around 18% based on the kinetics of Fe(II) oxidation by O 2 and H 2O 2 for 300 C/L charge dosage conditions, which yields sufficient iron dose for removing arsenic in Allensworth groundwater to levels
below the EPA standard.
Figure 2. A) Faradaic efficiency of H 2 O2 production over time (as shown by total volume of water treated). B) Total iron concentration in Cell 3 and Cell 4 over time (as shown by total volume of water treated).
Iron anodes were replaced periodically when observed voltages began to exceed safe operating limits. The theoretical concentration of iron produced under these conditions is expected to be around 87 mg/L (ppm as total Fe) based on Faraday’s law. The observed iron concentrations were consistent with expected concentrations (Figure 2B), confirming that the ACAIE reactor performance was not limited by anode performance at any time during these experiments, and any decrease in performance by the ACAIE cells is due to cathode degradation and/or fouling.
Objective 2: Design and optimize a compact, stacked continuous flow system that produces 600 LPH of arsenic-safe water.
During this project period we faced challenges with water leakages in the previous design of the continuous flow-through ACAIE reactor. To solve this problem, we created a new design that exposed to the atmosphere, which overcomes the high pressurization and leakage at corners or edges along the air cathode, and also avoids the accumulation of iron oxides within the reactor. Addressing the above-mentioned challenge of water-leakage from the reactors, and subsequent testing of various design iterations, our objective of identifying an optimal design for the continuous-flow mode ACAIE reactor with corresponding operating parameters was delayed.
Despite long delays, our team has achieved this objective of designing and building a 600 liter per hour (LPH) ACAIE flow-through system (Figure 3). This 600 LPH treatment system was successfully operated over a 2 week period under realistic operating conditions to treat nearly 2500 liters of synthetic California groundwater. Careful testing and sample collection occurred over a 1.25 hr operation period, where 800 liters of water was treated and analyzed for meeting drinking water standards. The findings of these experiments are discussed below.
Figure 3. A) Digital image of ACAIE 600 LPH treatment system used for full scale testing. B) Closeup view of five upright, modular ACAIE reactors s tacked in parallel.
The performance of the ACAIE 600 LPH treatment system was evaluated by treating nearly 800 liters of synthetic California groundwater (without arsenic) in a continuous flow mode. The recipe for the synthetic California groundwater (“SCAGW”) was determined from groundwater data (GAMA database, 2019) of various drinking water wells with arsenic concentrations greater
than 10 μg/L or 10 ppb. The composition of SCAGW is as follows: - : 1 mM; HCO 3 - : 1.4 mM; NO 3 - : 0.1 mM; SO 4 2- : 0.3 mM; HPO 4 2- : 4 µM; Ca 2+ : 0.6 mM; Mg 2+ : 0.3 mM; SiO 3 2- : 0.6 mM. A range of initial pH values were selected (pH 6.4 in Experiment 1 and pH 8.4 in Experiment 2) to investigate the performance of this system under the expected range of pH conditions in various groundwater matrices. Initial pH was adjusted by adding concentrated HCl to the influent tank while the electrolyte was mixed vigorously using a recirculation pump (1 HP). The average initial dissolved oxygen and conductivity of the influent water are 8.9 ± 0.6 mg/L and 782.1 ± 2 μS/cm, respectively.
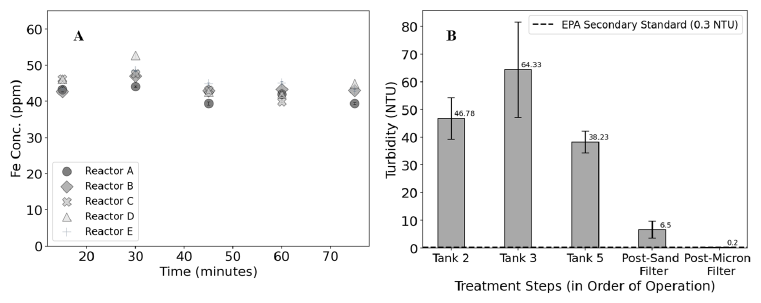
This system was operated at a flow rate of 600 liters per hour (retention time in each ACAIE reactor = 1.5 minutes) in continuous flow mode. The treatment steps are sequential from left to right in Figure 3A, where the ACAIE 600 LPH treatment system comprise Fe-electrolysis, coagulation and flocculation, gravitational settling, rapid sand filtration followed by an additional filtration with a pleated filter (20 inch Big Blue Filter, 5 micron size). A peristaltic pump is used to dose alum as a coagulant ( 4.2 ± 1.4 mg/L as Al) prior to the in-line static mixer. Water quality parameters (pH, dissolved oxygen, conductivity and turbidity), bulk-solution parameters (total iron production and residual H 2O 2 ) were monitored at various stages of treatment to understand the system performance in terms of total Fe production (via monitoring electrolysis) and particle removal (via monitoring turbidity). The average total iron concentrations during electrolysis in the water emerging from reactors A, B, C, D and E, are 41.6 ± 2.2 mg/L Fe, 43.8 ± 1.8 mg/L Fe, 44.2 ± 3.4 mg/L Fe, 45.7 ± 4.3 mg/L Fe and 45.0 ± 2.1 mg/L Fe respectively (Figure 4A). These values are consistent with the expected total iron concentration of 43.4 mg/L Fe in each reactor according to Faraday's law. The treatment system consistently delivers treated water in which concentrations of relevant added ions (such as Fe and Al), and the turbidity, are below their allowable limits for drinking water
(Figure 4B and Table 1).
Figure 4. A) Total iron concentrations in ACAIE reactors at various treatment times. B) Average turbidity measurements of the bulk solution at various treatment steps – Tank 2 (electrolysed water post-ACAIE reactors), Tank 3 (coagulated/flocculated water) and Tank 5 (settled water). See the image in Figure 3A for a visual of the treatment train order.
| P (mg/L) | Ca (mg/L) | Mg (mg/L) | Si (mg/L) | Total Fe (mg/L) | Total Al (mg/L) | |
| Before treatment | 0.4 ± 0.0 | 36.4 ± 1.8 | 9.5 ± 0.9 | 27.4 ± 4.2 | Below detect | Below detect |
| After treatment | 0.1 ± 0.0 | 32.8 ± 0.6 | 9.4 ± 0.2 | 16.9 ± 0.6 | 0.3 ± 0.1 | Below detect |
Table 1. Average concentrations of relevant constituents of interest in the influent water and
treated water.
Future work involves long-term testing of the ACAIE 600 LPH system in the field with arsenic contaminated groundwater. We will monitor operational (voltage, current, flow rates and overall energy consumption) and water quality parameters (pH, dissolved oxygen, conductivity and turbidity). The aforementioned parameters will be monitored, as well as subsequent potential fouling on the cathode while we validate the residence time that is necessary for removing arsenic to below EPA MCL. Lastly, we will measure H 2O 2 concentrations spatially and over time to study how this distribution affects Fe(II) oxidation kinetics.
No EPA funds were used to conduct community engagement work. During the extension period from January 2021 until September 2021, our team began engaging virtually with a community in Allensworth, with a different communication strategy than planned earlier. We submitted our IRB approval to the EPA Human Subjects Research Review Official (HSRRO) for final approval. The IRB approvals were sent to the Project Officer on Feb 2, 2021. Our original IRB application was approved on June 27, 2019. We made an amendment to our IRB in 2020 that was approved on May 20, 2020. Our community engagement efforts needed to be conducted mostly virtually during this project period except for two in-person meetings in June 2021 and December 2021. We travelled to Allensworth on June 1, 2021 to meet in-person for the first time with local community leaders, including leaders of the Allensworth Progressive Association (APA) to present and discuss about the ACAIE field trial plans and seek guidance from the Allensworth community leaders about the local logistics in planning this field trial. The local community leaders expressed strong interest in partnering with us on this field trial and also took us to visit a private farm on which they suggested would be a good site for conducting the field trial of ACAIE in early 2022. We also discussed their interests in seeking our partnership on their community education initiatives
in Central Valley such as the 2021 sustainable agriculture Work-based Learning (WBL) program hosted annually during the summer for local high school students in Alpaugh and Allensworth that was co-organized by the Allensworth Progressive Association (APA). We were invited to present educational modules virtually on Zoom for the 2021 WBL summer program, which we accepted. APA also expressed interest and discussed having us provide additional STEM education modules about water safety and health in Fall 2021 to their local K-8 students.
Figure 5. A) At the private farm in Allensworth with our community partners. This is the identified location of the field trial, to take place in 2022. B) Picture of a field in Allensworth from our site visit in June 2021.
For the 2021 WBL program, we delivered a set of education sessions about water quality, electrocoagulation, and air quality. As part of the 2021 WBL summer program, we also conducted a post-workshop evaluation using an anonymous online survey. Through the survey we discovered and documented that almost all participants fully agreed that these sessions increased their understanding of environmental issues and that they were excited to share this information they learned with their family and friends. Due to the success of this summer workshop, the APA invited us to partner with them on an EPA grant proposal to continue to work
with us and ensure support for the WBL program in Summer 2022. Whereas the 2021 WBL program was held virtually, the 2022 WBL program is planned to be held in-person.
During Fall 2021, we also partnered with the Allensworth Elementary School (K-8) and provided instruction virtually on a set of STEM education modules about water safety and health to the Allensworth 4th/5th grade and 7th/8th grade students to support their goals for improving STEM education at the school. We are looking forward to continuing to work with Allensworth Elementary School in the next Phase of the project.
Objective 4: Conduct an economic analysis to estimate the total cost of removing arsenic from the water at a community-scale plant in California. We conducted cost estimates for a 10,000 liter per day ACAIE plant. The estimates include the ACAIE reactors as well as the post-electrolysis flocculation and filtration steps. We also included waste management, housing and automation, and commissioning costs. These components yielded a total capital cost of $49,000, with a 125 square feet physical space requirement. For operating costs, we estimated $0.20 per 1,000 liters of treated water. The operating cost components include the consumables (e.g., iron plates, coagulation chemicals, and electricity). Labor costs are minimized to 2 hours per week due to the plans for almost completely automating the plant.
Conclusions:
Quality assurance requirements were, and are continuing to be, met by ensuring that the data collected is of the correct type, quantity, and quality to meet our project objectives. The system in place assures that data of high quality with integrity, objectivity, and utility, are being produced through the peer-review process of manuscript submission to academic journals. We report the specific parameters that we are consistently measuring in our experiments, and use controls to ensure the accuracy of results. Inductively Coupled Plasma –Optical Emission and –Mass Spectrometry (ICP-OES and ICP-MS) are used to measure arsenic concentrations in water samples to within 1 and 0.1 ppb, respectively. To improve the accuracy and precision of our ICP-OES analytical results, an internal standard is used which accounts for factors such as instrument drift and water matrix effects that would otherwise degrade the results of our
analytical method. Success of the novel technology is validated by consistent delivery of final arsenic concentrations to below EPA-MCL. We have carried out and will continue to conduct experiments with multiple replicates to ensure that arsenic levels remain below the EPA MCL with tests of statistical significance.
Journal Articles on this Report : 1 Displayed | Download in RIS Format
| Other project views: | All 8 publications | 1 publications in selected types | All 1 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Bandaru SR, van Genuchten CM, Kumar A, Glade S, Hernandez D, Nahata M, Gadgil A. Rapid and Efficient Arsenic Removal by Iron Electrocoagulation Enabled with in Situ Generation of Hydrogen Peroxide. Environmental science & technology 2020;54(10):6094-103. |
SU839960 (2020) SU839960 (Final) |
Exit |
Progress and Final Reports:
Original AbstractP3 Phase II:
Solving the Arsenic Problem in Rural California | 2022 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.