Grantee Research Project Results
2020 Progress Report: Low-cost Household Water Filter for PFAS Removal
EPA Grant Number: SU839810Title: Low-cost Household Water Filter for PFAS Removal
Investigators: Wu, Tingting , Vogler, Bernhard , Thanh Vu, Chi
Institution: The University of Alabama in Huntsville
EPA Project Officer: Page, Angela
Phase: I
Project Period: November 1, 2019 through October 30, 2020 (Extended to December 31, 2022)
Project Period Covered by this Report: November 1, 2019 through October 31,2020
Project Amount: $24,997
RFA: P3 Awards: A National Student Design Competition Focusing on People, Prosperity and the Planet (2019) RFA Text | Recipients Lists
Research Category: P3 Awards , P3 Challenge Area - Safe and Sustainable Water Resources
Objective:
This project aims to develop novel low-cost bio-hybrid adsorbents for point-of-use (POU) treatment of both long-chain and short-chain PFAS at homes/offices, and thus helps promote safe drinking water supply. Given the widespread contamination of drinking water sources by PFAS and the challenges to implementing comprehensive and uniform regulatory strategy, POU treatment can represent an inexpensive and immediate solution, which may be particularly appealing to small, rural, tribal, and disadvantaged communities.
Progress Summary:
During the reporting period, the research team focused on synthesis and testing of supported bio-hybrid adsorbents. We worked on selection of bio-hybrid adsorbent materials, screening tests, and optimization of synthesis procedure. In particular, we have successfully developed novel protein-functionalized adsorbents with a much enhanced adsorption capacity towards PFAS especially the challenging short chain PFAS. These results will lay a solid groundwork for phase II where a prototype point-of-use filter to be installed in home/office will be developed for PFAS removal using novel bio-hybrid adsorbents.
1. Protein & substrate selection
Four proteins, including Lysozyme (LYZ), Bovine Serum Albumin (BSA), Bovine Gamma Globulin (BGG), and Alpha Amylase (AAM) with isoelectric points of 11.4, 4.7, 6.7, and 9.1, respectively, were selected for the synthesis of bio-hybrid adsorbents. The wide span of these proteins’ isoelectric points (4.5 – 11) may provide opportunities of tuning the electrostatic interactions between the adsorbent and adsorbate, which together with hydrophobic interaction are hypothesized to be the main removal mechanisms of PFAS adsorption using bio-hybrid adsorbents.1,2
Three different substrates, i.e. graphene oxide (GO), layered double hydroxides (LDH), and granulated activated carbon (GAC), were tested for the synthesis of bio-hybrid adsorbents. These three materials have different characteristics. For example, GO and GAC have high surface area and many oxygen-containing functional groups, while LDH possesses high anion exchange capacity. All these properties may be appealing for PFAS adsorption.
2. Substrate synthesis
GO was prepared with Hummers’ method following our previous publication.3 Synthesis of Fe3O4@MgAl–LDH followed the co-precipitation method reported in our previous publication.4 GAC (coconut type, AquaCarb® 1240C Carbon, Evoqua) was also used as the substrate for bio-hybrid adsorbents.
3. Preparation of bio-hybrid adsorbents
Bovine serum albumin (BSA) loading on GO was conducted following the procedure reported in the literature.5 Briefly, a total of 20 mg of GO, 60 mg of BSA, 35 μL of Py, and
EDCI/HOBt/DMAP (molar composition = 1.5:1:0.5) were mixed and dissolved in 6 mL of a de-ionized (DI) water/DMF reaction mixture (DI/DMF = 4 mL/2 mL) for 5 h at room temperature. Then the mixture was washed with a mass ratio of ethanol/DI water = 3/1. Finally, the solution mixture was either freeze-dried for 3 days or oven-dried (at 30oC) for 3 hrs. Lysozyme (LYZ) was loaded on GO following a reported proceudre.6 1.5 mL (0.4 mg/mL in DI water) of LYZ was mixed with 0.5 mL of GO (0.5 mg/mL) for 24 h (25 oC; 60 rpm; pH 4). Then the sample was centrifuged and washed several times. The LYZ–GO was lyophilized and kept at -10 °C.
BSA was loaded on LDH as follows. BSA (3 mg/mL) was dissolved in 5ml of 0.1 M phosphate buffer containing 0.6 M NaCl at pH 5 and 25 mg of Fe3O4@MgAl–LDH was added, then the mixture was continuously shaken at 30 ◦C for 4 h, which was then separated by a magnet. LYZ–LDH nanocomposites was prepared following a reported method.7 The intercalation reaction was performed by adding Fe3O4@MgAl–LDH (0.25 g) into 20 mL LYZ (0.1 g) aqueous solution followed by shaking for 24 h (25 oC; 60 rpm; pH 11.9). Then the sample was centrifuged and washed several times. The LYZ–LDH was lyophilized and kept at -10 °C.
BSA-GAC composites was prepared as reported previously.8 100 mL (0.5 mg/mL in DI water) of BSA was mixed with GAC (100 mg) for 24 h (25 oC; 60 rpm; pH 4.5). Then the sample was centrifuged and washed several times. The BSA–GAC was lyophilized and kept at 4 °C. LYZ-GAC composites were prepared following the literature. 9 100 mL (1 mg/mL in 50 mM Tris-HCl) of LYZ was mixed with GAC (100 mg) for 24 h (25 oC; 60 rpm; pH 11). Then the sample was centrifuged and washed several times. The LYZ–GAC was lyophilized and kept at -10 °C. Bovine gamma globulin (BGG) was loaded on GAC as follows. 100 mL (0.5 mg/mL in DI water) of BGG was mixed with GAC (100 mg) for 24 h (25 oC; 60 rpm; pH 6). Then the sample was centrifuged and washed several times. The BGG–GAC was lyophilized and kept at -10 °C. Similarly, Alpha Amylase (AAM) was loaded on GAC as follows. 100 mL (0.5 mg/mL in DI water) of AAM was mixed with GAC (100 mg) for 24 h (25 oC; 60 rpm; pH 9). Then the sample was centrifuged and washed several times. The AAM–GAC was lyophilized and kept at -10 °C.
4. Screening tests of Bio-hybrid adsorbents
Fig. 1 PFAS removal using bio-hybrid adsorbents and corresponding substrates [5 mg of adsorbent was mixed with 40 mL PFAS solution (PFBA, PFBS, KPFOS or PFPeA; initial conc. 1 μg/L; pH 6; 60 rpm; 25 oC; 24 h)]. The error bars represent standard deviations of duplicate experiments.
The synthesized bio-hybrid adsorbents and corresponding substrates were first tested for PFAS adsorption in batch experiment. At the end of each run, the adsorbents were separated by centrifugation and aliquots of the solution were analyzed for PFAS by HPLC-Orbitrap-MS following a reported method following. 10 As shown in Fig. 1, for GO and LDH based adsorbents, protein functionalization did not always improve the performance, particularly for BSA. Here we tested two proteins with much different isoelectric points (BSA: 4.7; LYZ: 11.4). In general, LYZ-GO and LYZ-LDH performed better than the BSA loaded ones, especially for short chain PFAS (PFBA and PFBS), which may indicate electrostatic interaction mainly governed the PFAS adsorption. On the other hand, all protein functionalized GAC exhibited better adsorption capacity, and the enhancement was even more pronounced for PFAS of a shorter chain. Since GAC has long been used in water treatment and are commercially available, we selected GAC as the substrate in the following study. In addition to BSA and LYZ, we also tested BGG (isoelectric points 6.7) and AAM (isoelectric points 9.1) on GAC. As can be seen in Fig. 2, the performance of PFAS removal using GAC functionalized with different proteins did not always follow the order of increasing isoelectric points of the proteins. This implies electrostatic and hydrophobic interactions, as well as the characteristics of the specific protein play a role in PFAS adsorption with GAC-based bio-hybrid adsorbents. Further investigation is needed to better understand the underlying mechanisms. Moreover, it is worth mentioning that protein functionalization has improved PFAS adsorption 3~4 times for the challenging short chain PFAS, demonstrating the great potential of bio-hybrid adsorbents for such applications.
Fig. 2 PFAS removal of GAC-based bio-hybrid adsorbents [5 mg of adsorbent was mixed with 40 mL PFASs solution (PFBA, PFBS, KPFOS or PFPeA; initial conc. 1 μg/L; pH 6; 60 rpm; 25 oC; 24 h)]. The error bars represent standard deviations of duplicate experiments.
5. Synthesis optimization
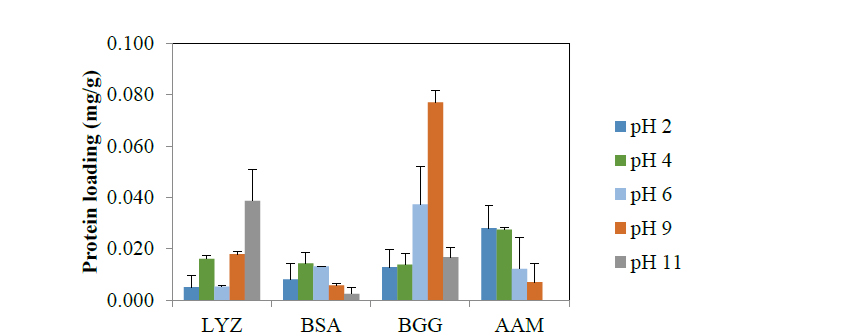
Following the initial screening of bio-hybrid adsorbents, we examined the effects of pH and ionic strength conditions on protein loading on GAC. As shown in Fig. 3, the optimum pH for protein loading was LYZ at 11, BSA at 4, BGG at 9, and AAM at 2. At the optimum pH for each protein, we further examined the effects of ionic strength (Fig. 4). While no significant effect of ionic strength on LYZ loading was observed, loading of BGG apparently favored low ionic strength. For BSA and AAM, a medium ionic strength resulted in enhanced protein loading
Fig. 3. Effects on pH on protein loading on GC [10 mg of GAC was mixed with 10 mL solution of proteins (LYZ, BSA, BGG
or AAM; initial conc. 0.5 mg/mL; 60 rpm; 25 oC; 24 hrs)]. The error bars represent standard deviations of duplicate experiments.
Fig. 4 Effects of ionic strength on protein loading on GAC [10 mg of GAC was mixed with 10 mL solution of proteins (LYZ, BSA, BGG or AAM; initial conc. 0.5 mg/mL; 60 rpm; 25 oC; 24 hrs; pH: LYZ 11, BSA 4, BGG 9, AAM 2)]. The error bars represent standard deviations of duplicate experiments.
In addition to chemical conditions, we also examined the kinetics of protein loading on GAC so the optimize the synthesis procedure. As shown in Fig. 5, for all the four proteins, uptake of protein by GAC reached equilibium after ~3 hrs. Moreover, the protein-loaded GAC samples collected at different time intervals here will be then tested for PFAS adsorption. The objective of these tests is to test the hypothesis that the amount of protein loaded on GAC is positively related with the adsorption capacity of the bio-hybrid adsorbents. Once these data are collected, we will finalize the synthesis procedure and proceed with Task 2.
The results obtained so far have not only demonstrated the great potential of bio-hybrid adsorbents for adsorptive removal of PFAS of different chain lengths, but also laid solid groundwork for the development of novel point-of-use filters to address the water quality challenge associated with these challenging pollutants.
Fig. 5 Kinetics of protein loading on GAC [10 mg of GAC was mixed with 10 mL solution of proteins (LYZ, BSA, BGG or AAM; initial conc. 0.5 mg/mL; 60 rpm; 25 oC)]. The error bars represent standard deviations of duplicate experiments.
Future Activities:
Future activities will focus on evaluation of PFAS removal from drinking water using bio-hybrid adsorbents. Specifically, we will evaluate the effects of water chemistry parameters on PFAS adsorption in series of batch kinetics experiments, followed by PFAS removal tests in environmental matrices (e.g. surface water, groundwater). We will also explore possible regeneration methods for the bio-hybrid adsorbents.
We will focus on Task 2, i.e. evaluation of PFAS removal from drinking water using bio-hybrid adsorbents. Specifically, we will evaluate the effects of water chemistry parameters on PFAS adsorption in series of batch kinetics experiments, followed by PFAS removal tests in environmental matrices (e.g. surface water, groundwater). We will also explore regeneration methods for the bio-hybrid adsorbents. It is expected that these results will lay a solid groundwork for phase II where a prototype point-of-use filter to be installed in home/office will be developed for PFAS removal using novel bio-hybrid adsorbents.
Journal Articles on this Report : 2 Displayed | Download in RIS Format
| Other project views: | All 3 publications | 3 publications in selected types | All 3 journal articles |
|---|
| Type | Citation | ||
|---|---|---|---|
|
|
Vu CT, Wu T. Adsorption of short-chained perfluoroalkyl acids (PFAAs) from water/wastewater. Environmental Science:Water Research & Technology 2020;6:2958-2972. |
SU839810 (2020) SU839810 (2021) |
Exit |
|
|
Vu CT, Wu T. Recent progress in adsorptive removal of perfluoroalkyl and polyfluoroalkyl substances (PFAS) from water/wastewater. Critical Reviews in Environmental Science and Technology. 2020. |
SU839810 (2020) SU839810 (2021) |
Exit |
Supplemental Keywords:
drinking water treatment technology, water filtration, human health, point of use, cost-effectiveProgress and Final Reports:
Original AbstractP3 Phase II:
Low-cost Household Water Filter for PFAS RemovalThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.