Grantee Research Project Results
2017 Progress Report: Beneficial Reuse of PCBs
EPA Grant Number: SU836782Title: Beneficial Reuse of PCBs
Investigators: Lu, Mingming , Zhang, Junsong , Khopade, Akshay , Dong, Son , McNeeley, Adam , Vu, Trang
Current Investigators: Lu, Mingming
Institution: University of Cincinnati
EPA Project Officer: Page, Angela
Phase: I
Project Period: August 1, 2016 through October 31, 2017
Project Period Covered by this Report: August 1, 2016 through July 31,2017
Project Amount: $14,985
RFA: P3 Awards: A National Student Design Competition for Sustainability Focusing on People, Prosperity and the Planet (2016) RFA Text | Recipients Lists
Research Category: Sustainable and Healthy Communities , P3 Awards , P3 Challenge Area - Chemical Safety
Objective:
Even though PCBs (poly-chlorinated biphenyls) production has ceased due to their high toxicity and bio-accumulation, PCB contamination remains a challenging problem to solve worldwide.
There has been a lot of progress made over the years in PCB treatment, via chemical, biological and thermal desorption, etc.. However, there are challenges in adopting these technologies due to the following reasons: high temperature, longer duration, and high initial cost, etc. Therefore, there is the constant need to seek low cost and yet effective technologies.
The overall objective of the research was to develop a low cost technology for PCB decontamination. Phase I tested the feasibility of catalytic hydrodechlorination by converting select PCBs into non-chlorine containing chemicals. Parametric study was carried out to understand what factors, and how the factors affect the reaction. Reaction kinetics will also be studied with the time remained to prepare for future scale up. Phase I (this proposal) is a proof of concept of the proposed technology. In Phase II, the team planned to test with more complex PCBs including some commonly used in the past, such as Aroclor 1254, 1248 and Askarel. With the help of their partners, they will modify the design based on test results as well as the real world oil or sediment matrixes. If Phase II is successful, they will pursue SBIR or contract opportunities to do a pilot test with the actual site conditions. The possibility of patenting the technology will also be pursued, to obtain license opportunities from companies.
Progress Summary:
The Phase I goal was to develop an all in one system for both dechlorination and hydrogenation. Specific aims of the project included the following: 1) Study the optimum conditions for catalytic dechlorination. 2) Research on the optimum conditions for bipehneyl conversion, 3) public outreach: disseminate research results at various venues to educate the public about waste reuse in environmentally friendly ways.
For specific aim 1, studying the optimum conditions for catalytic dechlorination has been carried out, and is in healthy progress that may result in a journal publication. Detailed kinetic studies will be carried out, and the resultant parameters will be helpful with future scale up.
New findings include a better solvent than reported from literature, much lower temperatures and much shorter time used. Reaction kinetics, will also be estimated to help with the scale up.
A few tests were performed on specific aim 2. But after discussion with the ETSC (Engineering Technology Support Center) of the US EPA and one of the executive secretaries of the PEN (PCB Elimination Network), we decided to stop this goal since it is “not practical” based on the feedback from the experts above. We are glad to stop right here before more investment in time and money are wasted.
For specific aim 3, student mentoring and outreach is being carried out and is on a healthy pace. An interdisciplinary team of students has been formed, which consists of two graduate students (environmental and materials engineering) and three undergraduate students (one in Chemical Engineering and two in Chemistry). Also a visiting faculty mentor has been coleading this project, who is an expert in environmental catalysis. The team developed best practices through working together. They made a few binders to document experimental conditions, one for instrumental analysis and a third one for literature on sustainability and technology related publications. They also established a shared Google Drive for literature and results. Students developed effective management skills for documentation/research.
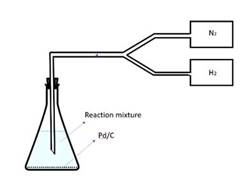
The experimental setup is a batch process carried out in a flask (Fig. 1). 50 mg of reactants
(AccuStandard), the catalyst Pd/C (with Pd content varying from 0.5% to 10% by weight), and Et3N (1.2 mole equivalent of chlorine in the PCB) and 50 ml toluene were placed in a flask. This flask is sealed with a cork with a carrier gas tube passing through. Before reaction, N2 gas was passed through the flask for 15 minutes to get rid of air inside the flask.
Fig. 1 Experimental Setup
Then a mixture of N2 and H2 (with a H2 partial pressure varying from 0.41 to 0.5) was passed through the suspension at room temperature ( 25°C ) and atmospheric pressure. The carrier gas supplied the hydrogen and also mixes the suspension. After reaction, the products were washed with magnetic stirs, filtered using vacuum filtration, and a naliquot of the filtered liquid was diluted 50 times for GC-MS (gas chromatography and mass spectroscopy) analysis. Both 3 chloronbiphenyl (3-PCB) and 2,3-dichloronbiphenyl (2,3-PCB) were tested. The reaction time was varied from 0.5hrs to 6hrs.Other temperatures (50°C and 80 °C) were also tested to obtain kinetics parameters, such as the activation energy, and rate coefficient.
The following has been achieved for specific aim 1. Improvement: improved biphenyl recovery and also used a new solvent. Much shorter reaction time than reported (Kume et al, 2008).
Prior to the experiments, the experimental setup was modified, and improvements were made to prevent leakage.
Most of the dechlorination studies ended at PCB's dis-appearance, without further discussion of expected product/products. Methanol has been redeemed effective, however, in our study, we found toluene as a better solvent, by having higher product recovery (not much difference with dechlorination), and also more sensitive for instrumentation (peak areas in toluene are 6.08 times larger based on our studies).
10% Pd/C is more effective than 5%, or 0.5% Pd/C under the same dosage. SEM-EDS (Energy Dispersive X-ray Spectroscopy) studies indicated that the surface Pd rate on 10% Pd/C is much higher than the other two: Pd weight were 0.1%, 2.1% to 7.0% for 0.5%, 5% and 10% Pd/C. Given the hydrodechlorination (HDC) is heterogeneous surface reaction, catalyst amount on the surface is essential. Therefore, 10% Pd/C is considered the best catalyst condition.
| Raction | Dechlorinaation rate, % | Biphenyl Recovery, % |
|---|---|---|
| R23(6hr) | 100 | 78.49 |
| R37(4 hr) | 99.97 | 78.51 |
| R38(2 hr) | 99.13 | 81.76 |
| R43(6 hr) | 100 | 75.32 |
| R44(4 hr) | 100 | 71.79 |
| r23(2 hr) | 100 | 69.07 |
Reaction time determination. Various reaction times were studied to find the dechlorination rate and biphenyl recovery rate. Table 1 indicated that these two rates did not change for 3-PCBs. Therefore, the effective reaction time for 3 PCBs is determined as 2 hours. Similarly, the reaction time for 2,3 PCBs is determined as 6 hours. Biphenyl (the major product) recovery rate has been lower than dechlorination, which may be caused by activated carbon absorption (even after 2hrs of sample washing in toluene).
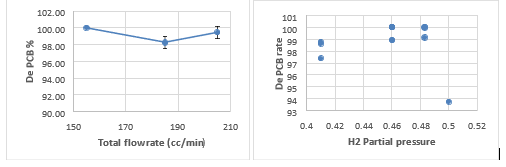
Fig. 2 The dechloriantion rate of 3 PCB vs. hydrogen partial pressure and total gas flow rate
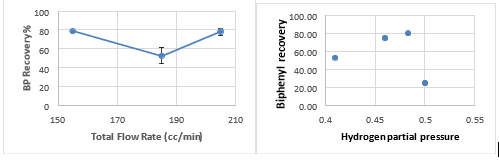
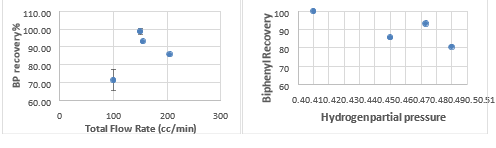
Fig. 3 Biphenyl recovery rate of 3 PCB vs. hydrogen partial pressure and total gas flow rate
For 3 PCBs, the hydrogen partial pressure (only one point at 0.5, and needs repeating) slightly increased the dechlorination rate (all of them are very high, 97% and above). The total flow rate at 150 cc/min (3 point all 99-100%) (hydrogen partial pressure of 0.483) seems to be the optimum condition. This is also supported by the highest biphenyl recovery rate (average of 79.6%, 3 repeats) at the same condition (Fig. 3).
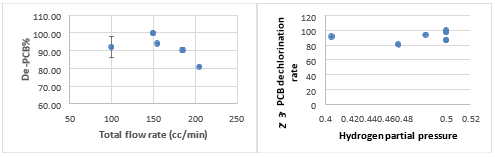
Higher hydrogen mole fraction (partial pressure) seems to increase dechlorination rate of 2,3 PCB (Fig. 4-5). The points at 0.405 and 0.46 H2 partial pressure will be repeated before further conclusion. Stirring intensity (indicated by gas mixture flow rate) can facilitate gasliquid transport when first increased, but then decrease dechlorination rate. The total flow rate at 150 cc/min and hydrogen partial pressure of 0.5 seems to be the optimum condition, but it will be repeated since only two points are shown.
Fig. 4 The dechloriantion rate of 2,3 PCB vs. hydrogen partial pressure and total gas flow rate
Fig. 5 Biphenyl recovery rate of 2,3 PCB vs. hydrogen partial pressure and total gas flow rate
Since Pd is an expensive catalyst, we tried to use spent coffee ground derived activated carbon coated in Zn. But the dechlorination rate was much lower (50-70%) for 3PCB after 68 hours. So this was not further pursued, although it seems a much cheaper option.
Specific aim 2 failed fast but with valuable lessons learned.
Due to hydrogen storage capacity of the Pd/C system through the formation of metal hydrides (Paál and Menon, 1988), we found that single catalyst, instead of bimetal, can also covert small amount of biphenyl to CHP at 180°C. This biphenyl has to be “freshly made” from PCB, no CHP was formed from pure biphenyl compound at 180°C. In order to have more CHPs, higher temperatures will be needed, which reduces the cost effectiveness.
Specific aim 3 is successful as indicated previously. The team of 2 faculty mentors and 3 students (other two graduated) will attend the Tech Connect Conference, and a platform presentation (Lu et al, 2017) has been accepted by the Annual Conference of the Air and Waste Management Association in June. In addition, a Master’s thesis will be produced together with a journal publication.
The PI worked with a consulting company, Progress in Energy and Environment, PE2, which has rich experience in waste reuse with thermal technologies, such as reuse of petroleum sedimentation as fuels (Sasivongpakdi et al., 2012). The company provided connection of the EPA’s ETSC(Engineering Technology Support Center) of the US EPA), whose main goal is the remediation of contaminated sites. Through the EPA Traineeship program, Adam McNeeley, went to work with the ETSC, and helped to find similar (but more expensive) applications, which indirectly supported our catalytic hydrodechlorination pathway.
Future Activities:
The experiments are on the right track, and will be completed by the time the project ends. A few more repeats will be performed in the next step to ensure repeatability. The reaction order (with regard to PCB and hydrogen) will be determined, and also the activation energy (varying the reaction temperature). These are all important for scale up.
Since we are still in the experiment stage, and did not have enough information for cost estimate yet. Cost estimate will be tried in the next step.
Supplemental Keywords:
PCBs (poly-chlorinated biphenyls), site remediation, catalytic hydrodechlorinationProgress and Final Reports:
Original AbstractP3 Phase II:
Beneficial reuse of PCBs (poly-chlorinated biphenyls) as new materials through a low cost process | 2018 Progress Report | 2019 Progress Report | Final ReportThe perspectives, information and conclusions conveyed in research project abstracts, progress reports, final reports, journal abstracts and journal publications convey the viewpoints of the principal investigator and may not represent the views and policies of ORD and EPA. Conclusions drawn by the principal investigators have not been reviewed by the Agency.